Valsartan Lawsuit Overview
Valsartan is a prescription drug approved by the U.S. Food and Drug Administration (FDA) in 1996 to treat high blood pressure, hypertension, and heart conditions. It was initially marketed by Novartis under the brand name Diovan.
It is believed that hundreds of batches of the prescription drug were contaminated with NDMA, a cancer-causing agent. The discovery of this contamination led to global recalls, including by the FDA and European health officials. NDMA issues found in batches of the drug are believed to be similar to those in Zantac lawsuits.
Recalls of valsartan, which is available under multiple names, began in 2018, especially with the generic form of the drug. The widespread use of valsartan-containing drugs has led to a significant number of lawsuits. It is believed that millions may be affected. To date over 1200 lawsuits have been consolidated in a Multidistrict Litigation (MDL) in New Jersey. Affected individuals are strongly encouraged to seek legal representation.
Valsartan Lawsuit – 2026 Updates
October 20, 2025: Plaintiffs File Motion for Class Action Settlement in Irbesartan Portion of Valsartan Lawsuit
The plaintiffs have reached a potential settlement in the lawsuit against Aurobindo and other manufacturers of irbesartan and valsartan. The settlement may resolve many of the claims made by irbesartan patients who took this drug and developed cancer because of the NDEA in it. Aurobindo has said it is still denying liability in the valsartan claims. However, the fact that a settlement was reached in the irbesartan portion of this massive group case is promising news for valsartan patients. More valsartan patients can file claims if they were diagnosed with cancer and believe a contaminated batch of valsartan is the reason.
October 3, 2025: Experts Testify at Expert Testimony Hearing in Valsartan Case
Several medical experts will testify at an upcoming Daubert hearing in the Valsartan case to determine which of them presents reliable, logical, and scientifically sound information. The plaintiffs’ attorneys intend to have several medical professionals give their formal opinions about the link between the NDMA in Valsartan and the plaintiffs’ cancer. The judge will hear evidence and testimony from the defense and plaintiff pool of experts and decide whether their opinions can be presented to the jury during the bellwether (test) trials. More consumers can join in this active group litigation by contacting an attorney for help filing their own Valsartan claims.
September 17, 2025: Valsartan Lawsuit Remains Stable, Parties Prepare for Upcoming Trial
There are 1,348 total cases pending in the active litigation against Valsartan’s manufacturers. Hundreds of consumers developed cancer after they took Valsartan contaminated with NDMA. Valsartan was recalled and taken off shelves across the globe starting in 2018; however, many patients are still developing cancer because they took the medication in the past. People who are filing lawsuits believe Valsartan’s manufacturer knew about the NDMA contamination and did not do enough to warn the public of the dangers.
September 2, 2025: As Valsartan Trials Approach, Defendants Try to Keep Evidence Out of Court
Trial preparations are in full swing in the Valsartan group case as parties continue to interview experts, request information, and file motions. The defendants have filed a host of court documents in an attempt to keep plaintiff-friendly evidence out of the courtroom. The plaintiffs’ attorneys have filed objections to these efforts, and the judge will hear arguments and issue a decision soon. Victims can still file claims if they took Valsartan and developed cancer.
August 19, 2025: Plaintiffs Call on Judge to Sanction Valsartan Manufacturer
Plaintiffs have asked the judge to impose sanctions on Zhejiang Huahai Pharmaceutical for destroying requested discovery documents before giving them to the plaintiffs’ counsel. These documents include certificates of analysis and safety data about Valsartan. The plaintiffs say these are materials that should have been preserved and sent to them. However, the defendant says these were destroyed as part of ordinary business practices. The judge should review the arguments and issue a decision soon.
August 1, 2025: Deadlines Extended for Test Trials in Valsartan Lawsuits
The Honorable Renée Marie Bumb has extended deadlines in the Valsartan, Losartan, and Irbesartan Products Liability Litigation. The second round of bellwether trials (test trials) are underway, and Judge Bumb gave attorneys on both sides more time to gather information for their expert reports. Both sides will also get an additional 21 days for rebuttal reports, depositions, and dispositive motions. People who developed cancer after taking contaminated Valsartan may still be able to join the group lawsuit.
July 18, 2025: More People File Valsartan Lawsuits in July
Between June and July, about 20 people filed new lawsuits as part of the Valsartan, Losartan, and Irbesartan Products Liability Litigation-MDL 2875. There are now 1,342 active lawsuits against companies who made contaminated Valsartan and similar drugs. People who have developed cancers of the colon, stomach, liver, esophagus, prostate, bladder, lung, and blood have lawsuits claiming the contaminant NDMA in Valsartan caused their cancer. Injured people may still be able to qualify for a lawsuit against Valsartan’s manufacturers.
July 1, 2025: Court Sets New Schedule for Second Wave of Bellwether Trials in Valsartan MDL
Judge Renee Marie Bumb has entered an order that finalizes the preparation schedule for Wave 2 of the Valsartan cases that are on track for their Bellwether trials. For example, under the order, legal counsel for the victims can interview experts between July 18, and August 20, 2025. Then, the victims’ attorneys can use the evidence gathered during these discussions to craft compelling arguments against the Valsartan manufacturers. People who developed certain cancers after taking Valsartan may still be able to join the group lawsuit.
June 17, 2025: More People File Valsartan Lawsuits in June
There is multidistrict (group) litigation for people harmed by contaminated Valsartan (MDL -2875 IN RE: Valsartan, Losartan, and Irbesartan Products Liability Litigation). Between May and June, more people joined the group lawsuit, bringing the total to 1,325 active plaintiffs. As more people develop cancer after taking Valsartan contaminated with the carcinogen NDMA, more people will file lawsuits against the drug’s manufacturers.
June 3, 2025: Test Trials Are Approaching in Group Valsartan Lawsuit
The judge in the group lawsuit for people who developed cancer after taking contaminated Valsartan has set key trial dates. The first round of bellwether (test) trials in this group litigation is slated for October 2025. U.S. District Judge Renee M. Bumb has also asked attorneys to start preparing four lawsuits for a second round of test trials. In group trials, like this multidistrict litigation, it is not uncommon for settlements to occur prior to these bellwether trials. A second round of these trials could motivate some Valsartan manufacturers to settle lawsuits from people harmed by their drug.
May 2, 2025: Valsartan Cancer Lawsuit Grows As More People Join the Federal MDL
The lawsuit involving the medication Valsartan grew by 12 cases to 1,322 as of May 1, 2025. Valsartan, also known by the brand name Diovan, was used to treat high blood pressure and cardiac issues. It was recalled after product testing found the drug contained the carcinogen, NDMA. Hundreds of people have filed legal claims to recover compensation after discovering their or their loved one’s cancer may be due to contaminated Valsartan.
April 16, 2025: Defendant in Valsartan Lawsuit Struggles to Produce Documentation
ZHP, a Valsartan manufacturer, has been asked to turn over multiple documents for the multidistrict litigation involving harm caused by their drug. ZHP, a Chinese manufacturer, has not produced all of the documents. These documents are related to certificates of analysis, material safety data sheets, document retention policies, and the Chinese “standard policies” for certificates of analysis. ZHP received a seven-day extension to April 15, but plaintiffs’ attorneys still have not received the documentation, which will be important in building their case.
April 2, 2025: Seven More Valsartan Cases Filed in Federal Multidistrict Litigation
The total pending federal lawsuits against the companies that manufactured and sold Valsartan rose to 1,310 as of April 1, 2025. This is an increase of 7 cases since the March numbers were released. The parties are prepping for the September 2025 test trials, with important discovery deadlines approaching. Consumers are suing because they unknowingly took Valsartan that was contaminated with a substance that causes cancer. Once the test trials are underway, it is likely many more plaintiffs will join the Valsartan litigation.
March 26, 2025: Valsartan Judge Sets Trial Preparation Deadline Ahead of September Bellwether Trial
The judge overseeing the Valsartan MDL has entered an order setting key deadlines to help the parties prepare for the trials set to occur in September 2025. These include giving the parties between April 11 through May 8, 2025, to question expert witnesses. Some experts who may be questioned include those on plaintiffs’ medical teams and scientists involved in making and testing this now-recalled heart and blood pressure medication. These trial preparations are important to people who have filed and who will file lawsuits related to cancers caused by Valsartan.
March 4, 2025: Fifteen More Valsartan Lawsuits Added as Preparations Continue for First Bellwether Trial
As of March 3, 2025, there are 1,303 pending claims in the Valsartan multidistrict litigation against Zhejiang Huahai Pharmaceutical Co. and other drug companies. Plaintiffs claim Valsartan and drugs like it contained NDEA and NDMA, which are known probable carcinogens. The parties are preparing for one of the first Bellwether trials in this litigation, which began in 2019. Affected patients and surviving family members can join in this lawsuit.
February 25, 2025: Documents Turned Over By Valsartan Manufacturer May Help Plaintiffs’ Lawyers Strengthen Lawsuit
In a letter drafted by attorneys representing people harmed by contaminated Valsartan, lawyers say ZHP turned over evidence that solvents used in its manufacturing process may have led to contamination. One theory is that ZHP and other drug makers used cheaper ingredients and low-quality processes that led to the presence of NDMA and NDEA in Valsartan. These contaminants may have led to people who used Valsartan to develop cancer. The documents provided by manufacturers will be used as evidence in the trial.
February 6, 2025: Valsartan MDL Judge Sets Scheduling Order Ahead of Test Trials
On February 3, 2025, Judge Renee Bumb entered an order scheduling times to complete and address key legal issues ahead of the test trials in the Valsartan multidistrict litigation (MDL). Plaintiffs believe this heart medication that the FDA approved in the late 1990s was contaminated with a cancer-causing substance, NDMA. The federal court’s order seeks to resolve pending matters, such as questioning experts and submitting their reports. This litigation is still active and people who were diagnosed with various cancers, non-Hodgkin’s lymphoma, and multiple myeloma after taking Valsartan can still join the lawsuit.
January 16, 2025: 1,288 Lawsuits Pending in the Valsartan Federal Litigation
Many people have joined the consolidated lawsuits against Valsartan manufacturers (MDL 1875). Attorneys in these cases are preparing to hold bellwether (test) trials. Heading into these trials, the total pending lawsuits against the makers of Valsartan—a heart and blood pressure medication—reached 1,288 as of January 2, 2025. The plaintiffs claim many batches of Valsartan were contaminated with N-Nitrosodimethylamine (NDMA). Lab studies reveal that NDMA may cause cancer and hepatic (liver) injuries. Plaintiffs are encouraged to contact a lawyer if they take Valsartan and suffer serious harm.
December 20, 2024: Federal Valsartan MDL Judge Approves 5 Cases for Second Set of Test Trials
On December 16, 2024, the federal judge in the lawsuit against manufacturers of Valsartan entered an order that confirmed the selection of five lawsuits for the second round of bellwether trials. Bellwether trials are a select group of cases that serve as test trials for other cases in a multidistrict litigation (MDL). Many people who were harmed by Valsartan have joined MDL 2875, which represents people harmed by NDMA contamination in Valsartan and other medications. These five test trials can predict how other cases in the MDL might be decided, so these cases are very important to all plaintiffs in the MDL.
December 10, 2024: Valsartan Judge in Federal Lawsuit Sets Important Deadlines to Advance Case
On December 4, 2024, Judge Renee Bumb—the Judge managing the Valsartan MDL—entered an order setting key deadlines to help advance the lawsuit to the next stage. For example, the order gives a deadline of February 14, 2025, for the parties to finish fact discovery for individual cases. Additionally, the plaintiffs have until February 28, 2025, to pick experts they will use to support their legal arguments.
December 4, 2024: Parties Discuss Concerns Before Status Hearing in Valsartan Lawsuit
On December 3, 2024, the plaintiffs and defendants submitted letters to the judge in the Valsartan multidistrict lawsuit (MDL 2875). In the letters, each side gives their opinions on unresolved issues in the case. For example, they discuss whether the plaintiffs should dismiss claims against defendants who have not been served with paperwork yet. Additionally, they discuss waivers and what cases should proceed to the Bellwether (test) trial stage of the lawsuit.
November 19, 2024: Plaintiffs Send Letter Proposing Schedule for Expert Reports in Valsartan Multidistrict Litigation
On November 19, 2024, the plaintiffs sent a letter to the judge in MDL 2875, explaining their positions about the timing of expert reports and questioning. The plaintiffs think the defendants should file their expert reports before the plaintiffs question their experts. The plaintiffs believe this is fair, aligns with customary practice, and allows them to question their experts about matters the defendants’ experts discuss in their reports.
November 15, 2024: Parties Agree to Deadlines in Valsartan MDL Case Set for Bellwether Trial
On November 12, 2024, the defendants in the Valsartan MDL case sent a case scheduling letter to the Judge. In it, the defendants outline the agreed-upon deadlines for milestones—like the case-specific discovery—in one of the lawsuits with an upcoming Bellwether trial. The letter notes that the parties disagree on the appropriate deadlines for questioning experts. The parties will discuss this more on November 19, 2024.
November 8, 2024: Plaintiffs’ Attorneys in Valsartan MDL Request More Time to Respond
In an October 29, 2024, letter to the Honorable Renee Marie Bumb, attorneys for the plaintiffs in the Valsartan multidistrict lawsuit MDL 2875) asked for an extension to submit information. The defendants, who represent Valsartan manufacturers, drafted a letter that plaintiffs had to respond to. The previous day, the defendants had sent a letter to the Judge recommending new deadlines for the Bellwether trial (i.e., test trial) preparations and start dates. The plaintiffs asked for an extension past the November 12, 2024, response deadline. Judge Bumb granted the plaintiffs’ extension request during the hearing on October 29, 2024.
November 1, 2024: Lawyers Suggest Schedules for Bellwether Trial Preparation in Valsartan Litigation
On October 29, 2024, the plaintiffs and defendants in the Valsartan MDL sent letters to the judge to suggest schedules for the Bellwether trial preparations. Previously, the judge said these six trials would begin in early November. Now, the first trial won’t start until September 2, 2025. The plaintiffs asked to finalize trial preparations by August 15, 2025. Defendants asked for an August 22, 2025, deadline instead. Bellwether trials are a small group of test trials that help determine how future, similar cases will play. They are used to establish common evidence and predict success for future Plaintiffs.
October 15, 2024: Plaintiffs in Valsartan Litigation Look to Dismiss Defendant’s Expert Witness
In a letter to the Honorable Renée Marie Bumb, the Chief Judge in the Valsartan multidistrict litigation, lawyers for the plaintiffs asked for an expert witness to be dismissed. Dr. Chodosh was an expert witness for the defendants. Plaintiff’s attorneys said Dr. Chodosh should not be allowed to testify before the jury. The attorneys stated that the Valsartan trial is an economic loss trial based on regulatory standards. Dr. Chodosh was an expert witness for general causation, which they argue is not relevant to this trial. Therefore, they say Dr. Chodosh’s expert testimony is not relevant. A final decision on this matter will be made by Judge Bumb.
October 3, 2024: First Valsartan Bellwether Trial Set for November
The first bellwether trial for the Valsartan lawsuit will begin in November. Bellwether trials reflect the larger group of cases and are used to determine how a jury will react to the evidence and arguments presented. They also influence settlement discussions. As of October 2024, there are approximately 1,200 filed cases in this MDL. The court has scheduled oral arguments for October 8th to address unresolved issues regarding discovery and testimony in preparation for the upcoming trial.
October 1, 2024: Son Files Wrongful Death Lawsuit Alleging Valsartan and Irbesartan Caused Mother’s Multiple Myeloma
On September 26, 2024, the son of a deceased Missouri woman filed a lawsuit on behalf of his mother, alleging that her use of Valsartan and Irbesartan caused her to develop multiple myeloma. The complaint against Mylan Laboratories Ltd. and Mylan Pharmaceuticals Inc. claims that the defendants were negligent and failed to warn of the dangerous side effects of the drugs. The lawsuit demands punitive damages and wrongful death.
September 1, 2024: Valsartan Trial Rescheduled for Fall 2024 Under New Judge
The first Valsartan trial is back on track for the fall of 2024. Originally scheduled for earlier this year, the trial was delayed due to the judge’s illness. The case has since been transferred to a new judge, the Honorable Renee Marie Bumb, who has made it clear that the trial will proceed as planned. Jury selection is set for late October, with three to four weeks of testimony beginning in early November.
August 1, 2024: Rescheduling of Valsartan Lawsuit Case Management Conference and Daubert Hearings
Judge Rene Marie Bumb has rescheduled the next Valsartan lawsuit Case Management Conference from August 21, 2024, to Wednesday, September 18, 2024, at 1:00 PM. At this time, the Court will conduct Daubert hearings on the proposed testimony for several experts. “Daubert” hearings are used in complex cases so the judge can assess the relevancy and reliability of expert testimony. The Daubert standard typically uses the following factors: whether the technique or theory in question can be, and has been, tested; whether it has been subjected to publication and peer review; its known or potential error rate; the existence and maintenance of standards controlling its operation; and whether it has attracted widespread acceptance within a relevant scientific community.
The “Daubert Standard” was established in the 1993 case Daubert v. Merrell Dow Pharmaceuticals Inc., 509 U.S. 579 (1993) and revolutionized the way courts evaluate expert testimony.
June 18, 2024: Plaintiffs Seek Sanctions Against ZHP for Failure to Produce CEO in Valsartan Case
An important motion has been made in the Valsartan Lawsuit. The brief, filed by plaintiff lawyers, addresses whether a penalty, also known as a sanction, should be paid by defendant ZHP. ZHP has failed to produce their CEO for a deposition, despite being ordered by the court to do so. The judge previously ordered sanctions against the company. The brief alleges a particular email that admits the company knew the process used to create Valsartan created NDMA. Plaintiff’s counsel further argues that there are other significant gaps in the production of documents by Defendant ZHP. A special master previously ordered that an adverse inference be applied at the time of trial in the Valsartan MDL.
June 9, 2024: Special Master Appointed in Valsartan Lawsuit to Oversee Complex Discovery Issues
Judge Thomas I. Vanaskie (Ret.) is assigned as the Special Master in the Valsartan Lawsuit. The Special Master is usually a retired judge or attorney acting as a court-appointed representative to sort through and guide specific and complex issues of the case, such as discovery. Judge Vanaskie recently scheduled a conference for June 21, 2024, to discuss the scheduling for the lawsuit as well as any other necessary issues related to the claims. Chief Judge Renee Bumb will be meeting with Judge Vanaskie the following week, on June 25, 2024.
June 8, 2024: Details of the Valsartan Class Action Lawsuit – Divisions and Allegations
The Valsartan Class Action Lawsuit is comprised of three parts: first, the personal injury claims; second, medical monitoring claims; and third, the economic loss claims. The lawsuit alleges that several manufacturers produced Valsartan and Losartan containing the carcinogen NDMA. It is believed that the contamination was caused by the manufacturing process used to produce Valsartan and Losartan. One complicating factor is that cancer can develop years after exposure to a toxic substance like NDMA. Everyone who paid for a contaminated drug can request a refund through the class action lawsuit. Individuals with cancer have a separate personal injury claim. Additionally, those who were exposed can also apply for medical monitoring if they have not yet received a diagnosis that might be related to Valsartan exposure.
May 5, 2024: Valsartan Trial Assignment and Plaintiff’s Request for Conference
The reason for the delay in the Valsartan Trial is now clear. The case has been reassigned to another judge. MDL 2875 In Re: Valsartan, Losartan, and Irbesartan Products Liability Litigation is now assigned to the Honorable Renedd Bumb. By a letter dated April 25, 2024, plaintiffs have requested an in-person conference with the new judge to address the status of the litigation and the third-party payor subclass trial that was scheduled for March 18, 2024.
April 9, 2024: No New Trial Date Scheduled for Valsartan Lawsuits
There is still no new trial date scheduled in the Valsartan Lawsuit. There have been several pre-trial motions decided in this bellwether trial. The federal court filing system known as “PACER” shows that a status conference occurred on March 27, 2024, and the transcript was posted under seal the next day. Whatever happened at that conference seems important. We think it is unlikely that the Valsartan trial is going forward this month since there is no trial date officially set at this point.
April 1, 2024: Valsartan Class Action Certification and Personal Injury Claims
The valsartan lawsuit has two distinct parts. First, there is the class action lawsuit. The Valsartan class action was certified by the US District Court for New Jersey in December of 2023. The class action certification does not decide whether any defendant did anything wrong; it simply allows different classes of plaintiffs to bring their cases in an efficient manner. The classes in the Valsartan class action lawsuit are: 1) consumer economic loss class, 2) medical monitoring class, and 3) the Third-Party Payor Class. In addition to the class action lawsuit, there are personal injury claims, which are separate from the class action. The personal injury cases allege that contaminated generic Valsartan caused cancer. Our firm is investigating valsartan cancer cases.
March 26, 2024: Key Decision Issued Ahead of Valsartan Trial by Judge Kugler
Judge Kugler has issued an important decision in advance of the Valsartan bellwether trial that was supposed to start 10 days ago. The Order, issued today, largely denies the request for decisions as a matter of law. Both plaintiffs and the defendants asked Judge Kugler to rule on at least 16 issues based on the documents and deposition testimony that have been produced so far. The Judge did, however, grant one important request made by the Plaintiffs. He ruled that defendants’ affirmations, statements, and labeling of the generic Valsartan they produce was an “express warranty.” Since the judge has already ruled that the statements of the defense were express warranties, they will not be able to challenge whether or not there were express warranties in front of the Jury. There is not yet a new trial date in the Valsartan MDL. We expect a date soon.
March 21, 2024: Study Links NDMA-Contaminated Valsartan to Increased Cancer Risk
Several scientific studies have linked the use of Valsartan contaminated with NDMA to an increased risk of cancer. One study compared 1.4 million people and their cancer rates. The control group consisted of the population that was taking Valsartan before the contamination date, and the other group took the contaminated Valsartan. The study was titled “Nitrosodimethylamine-Contaminated Valsartan and Risk of Cancer: A Nationwide Study of 1.4 Million Valsartan Users.” It was published in the Journal of the American Heart Association on December 20, 2022.
March 20, 2024: Valsartan Lawsuit Status Update
The Valsartan trail has been postponed. There is some indication that a member of the Court has become ill. There are also several pretrial motions that have not been decided yet. It is unclear when the trial will begin. It is possible that the court could grant summary judgment and say that certain manufacturers provided contaminated drugs. Plaintiff’s lawyers argue that Valsartan was clearly contaminated and several government findings confirms that much of the Valsartan sold between 2015 and 2018 was not pure. If summary judgement is granted there may be a damages only trial.
March 2024: To date, 1,231 lawsuits are pending, 1,398 have been filed, and no settlements have been made. Litigation continues.
August 2023: The number of lawsuits pending in the MDL expands to over 1,200.
January 2023: After a court ruling allows cases to move forward using scientific study findings, the number of lawsuits grows to over 1,000.
February 2019: Valsartan lawsuits are consolidated in a Multidistrict Litigation (MDL) in New Jersey.
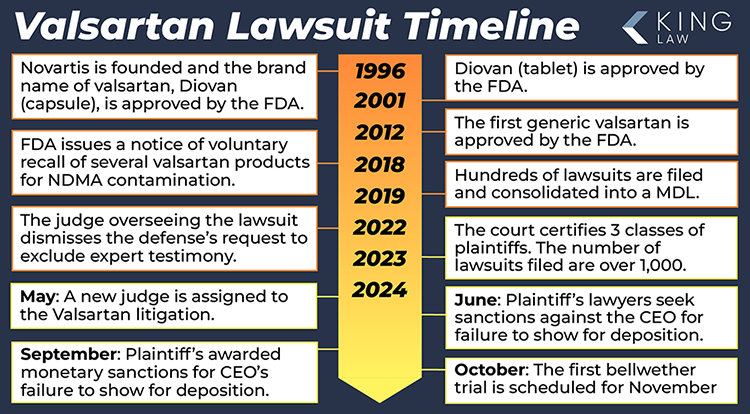
Table of Contents:
What is Valsartan and How Does It Work?
Valsartan Side Effects and Injuries
Valsartan Recalls – FDA List of Recalled Medications
Eligibility to File a Valsartan Lawsuit
Recoverable Damages in the Valsartan Lawsuit
Statute of Limitations for Valsartan Lawsuits
How to File a Valsartan Lawsuit
Valsartan Payout and Settlement Amounts
What is Valsartan and How Does It Work?
Valsartan, an angiotensin II receptor blocker (ARB) marketed by Novartis, was originally approved by the U.S. Food and Drug Administration in 1996 under the brand name Diovan. It is primarily used to treat high blood pressure, heart failure, and diabetic kidney disease. In 2012, after the Novartis’ patent expired, the market saw an expansion of generic valsartan. Most of the generic forms of the drug were produced overseas.
Valsartan-containing drugs relax blood vessels, reducing blood pressure and increasing the blood and oxygen supply to the heart. It works almost immediately, reducing blood pressure around two hours after initial intake. The drug achieves full effectiveness within two to four weeks.
There are nearly two dozen pharmaceutical companies that manufacture valsartan-containing ARBs. By 2019, many of the generic versions of the brand had been recalled by the FDA due to the discovery of unacceptable limits of impurities. This led to the FDA fast-tracking a new generic version of the drug to help mitigate shortages caused by these recalls.
What Is Valsartan Used For?
Valsartan is primarily used to reduce high blood pressure. It inhibits angiotensin II binding to the AT1 receptor, reducing vasoconstriction and aldosterone synthesis. In a short amount of time, valsartan-containing drugs facilitate easier blood flow and reduce the heart’s workload.
Primary uses of valsartan:
- Lower high blood pressure (hypertension)
- Treat heart failure
- Reduce hospitalization risk from heart failure
- Improve post-heart attack survival rates
- Manage diabetic kidney disease
The drug is often prescribed for heart and left ventricular failure after a heart attack. Millions of people have been prescribed valsartan-containing drugs, particularly generic versions of the medication.

Valsartan Side Effects and Injuries
There are a number of known or reported side effects associated with taking valsartan. While some symptoms may be relatively mild, many patients have reported serious health conditions related to prolonged use of the drug.
Our law firm is accepting the following cancers as injuries for the valsartan lawsuit:
- Colorectal/colon/rectal
- Intestinal (including small intestine)
- Stomach/gastric
- Liver
- Esophageal
- Prostate
- Blood (including Non-Hodgkin’s lymphoma, Multiple myeloma, and Leukemia)
- Bladder
- Lung (Will need to know smoking history)
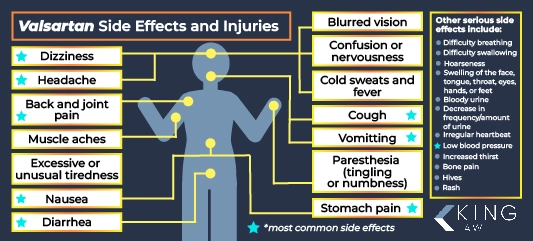
Potential side effects of valsartan:
- Dizziness
- Headache
- Excessive or unusual tiredness
- Diarrhea
- Nausea
- Stomach pain
- Back and joint pain
- Blurred vision
- Paresthesia (tingling or numbness)
- Muscle aches
- Cough
- Cold sweats and fever
- Confusion or nervousness
The drug may also cause serious side effects such as difficulty breathing or swallowing, hoarseness, and swelling of the face, tongue, throat, eyes, hands, or feet.
Patients taking valsartan should seek emergency medical treatment for any adverse health conditions, including bloody urine or a decrease in the frequency or amount of urine, an irregular heartbeat, low blood pressure, or increased thirst. The drug may also cause unexplained weight gain, bone pain, and hives or a rash.
Most Common Side Effects of Valsartan
The most common side effects of valsartan are typically mild and temporary but may result in discomfort, including abdominal pain, dizziness, and changes in blood pressure. Patients affected by serious or persistent side effects related to valsartan usage should consult with their doctor.
Common side effects of valsartan:
- Flu-like symptoms (cough, fatigue, joint pain)
- Abdominal pain
- Back pain
- Diarrhea
- Dizziness
- Headache
- High blood potassium (hyperkalemia)
- Low blood pressure (hypotension)
- Vomiting
Many of these symptoms are transient in nature and do not require stopping the use of valsartan. Symptoms should be discussed with a primary care physician to determine the best treatment plan moving forward.
Brand Names for Valsartan
Valsartan has been marketed under a number of name brands and generic versions of the medication. It was initially approved under the name brand Diovan in 1996. The drug is available in single-ingredient and multi-ingredient formulations.
Brand-name valsartan medications:
- Diovan (single-ingredient)
- Prexxartan (single-ingredient)
- Valturna
- Exforge
- Exforge HCT
- Diovan HCT
- Byvalson
- Vyduo
- Entresto
Currently, over two dozen manufacturers make valsartan-containing drugs in single-ingredient or multi-ingredient formulations. There are several options available for patients throughout the United States and across the globe.
Drug Companies Involved in the Valsartan Lawsuit:
By 2018, it was discovered that potentially hundreds of batches of valsartan had been contaminated with NDMA (N-nitrosodimethylamine), particularly generic versions of the drug manufactured overseas. It is alleged that Zhejiang Huahai (a Chinese pharmaceutical company) was the primary manufacturer of the contaminated lots. The drugs were then distributed by U.S. companies, including Teva Pharmaceuticals and Solco Healthcare.
Pharmaceutical companies involved in the manufacture or distribution of valsartan:
- Zhejiang Huahai (manufacturer)
- Teva Pharmaceuticals Ltd.
- Solco Healthcare
- Actavis
- A-S Medication Solutions
- AvKARE
- Bryant Ranch Prepack
- Camber Pharmaceuticals
- Hetero Labs, Inc.
- HJ Harkins Company
- Major Pharmaceuticals
- Mylan Pharmaceuticals
- Northwind Pharmaceuticals
- NuCare Pharmaceuticals
- Preferred Pharmaceuticals
- Prinston Pharmaceutical
- RemedyRepack Inc.
- Torrent Pharmaceuticals Limited
The contaminant found in valsartan is the same potentially cancer-causing toxin at the center of Zantac lawsuits. Individuals who have been exposed to NMDA as a result of taking valsartan are strongly encouraged to consult with an attorney as soon as possible.
Valsartan Mechanism of Action
Valsartan blocks angiotensin II by selectively binding to the angiotensin receptor 1 (AT1), preventing its vasoconstrictive and aldosterone-secreting effects. It provides comprehensive angiotensin II antagonism, which affects blood vessels, aldosterone production, cardiac function, and sodium reabsorption.
Unlike ACE inhibitors, valsartan prevents the action of angiotensin II regardless of its production pathway, ensuring a complete receptor blockade. It also directly acts on receptors without influencing bradykinin levels.
Ingredients in Valsartan
Diovan, a popular name-brand of the drug, contains 80 mg or 160 mg of valsartan, depending on the dosage. However, the active ingredient dosage depends on the specific brand. Most versions of the drug also contain a number of inactive ingredients that also differ by brand.
Inactive ingredients in Diovan (a name-brand version of valsartan):
- Cellulose compounds
- Crospovidone
- Gelatin
- Iron oxides
- Magnesium stearate
- Povidone
- Sodium lauryl sulfate
- Titanium dioxide
Different versions of the drug contain varying levels of the active ingredient (valsartan) and inactive ingredients.
Valsartan Recalls – FDA List of Recalled Medications
On July 13, 2018, the FDA issued a press release calling for the voluntary recall of several valsartan-containing medications following the detection of an impurity in multiple batches of the drug. The recall affected various manufacturers and was due to the discovery of a potentially carcinogenic toxin known as NDMA. Patients were advised to continue using the medication until they were able to secure a replacement product
Manufacturers involved in the recall include:
- A-S Medication Solutions LLC
- Aurobindo Pharma
- AvKARE Inc.
- Bryant Ranch Prepack Inc.
- Hetero Labs
- Major Pharmaceuticals
- Mylan Pharmaceuticals
- Northwind Pharmaceuticals
- NuCare Pharmaceuticals
- Preferred Pharmaceuticals
- RemedyRepack Inc.
- Rising Pharmaceuticals
- Solco Healthcare LLC
- Teva Pharmaceuticals
- Torrent Pharmaceuticals
Impacted medications include those containing valsartan with amlodipine, hydrochlorothiazide, or both. Valsartan with Hydrochlorothiazide by Alembic was not affected by the recall. Some companies, including Solco Healthcare LLC, who were part of the recall, provide customer reimbursement.
Eligibility to File a Valsartan Lawsuit
As of October 2025, King Law is no longer accepting Valsartan lawsuits.
Individuals must meet certain eligibility requirements to file a personal injury lawsuit. In order to determine whether you meet the criteria, you need to speak with an attorney as soon as possible.
Criteria for a Valsartan lawsuit:
- Proof of at least 6 months of valsartan use between 2015 to 2019 prior to cancer diagnosis (can be in combination with HCTZ and/or Amlodipine)
- Relevant post-2017 cancer diagnosis
- Not currently represented by an attorney
Only certain cancer diagnoses will qualify. Qualifying cancer diagnoses may include colorectal, intestinal, stomach, liver, esophageal, prostate, blood, bladder, or lung.
Evidence to Gather Before Filing a Valsartan Lawsuit:
Valsartan lawsuits are based on evidence. Therefore, it is critical to gather evidence prior to filing a lawsuit.
Evidence you may need to collect includes:
- Medical records
- Proof of Valsartan usage
- Cancer diagnosis details
- Other relevant documentation to support your case
Working with an attorney as early in the process as possible can help ensure you secure all necessary documents and information.

Recoverable Damages in the Valsartan Lawsuit
Damages in a Valsartan lawsuit are based on a number of factors, including the severity of a person’s injuries and financial losses. Compensation in these cases may include coverage of medical expenses, lost wages, and future care and treatment.
Generally, both economic and non-economic damages are available. An attorney can help ensure you receive financial recovery for all of your Valsartan-induced injuries, including your long-term care, physical pain, mental anguish, diminished quality of life, and more. Under certain circumstances, individuals who have lost a loved one may be able to file a wrongful death lawsuit on their behalf.
Statute of Limitations for Valsartan Lawsuits
Under most circumstances, there is a two-year statute of limitations on Valsartan lawsuit cases; however, the deadlines vary by state. The statute of limitations begins to toll once the injury is discovered, such as a valsartan-related cancer diagnosis. Prompt filing of a case ensures the best chance for a successful claim. Since there is no federal statute of limitations, it is imperative to work with an attorney to ensure your claim is filed timely.
How to File a Valsartan Lawsuit
If you were diagnosed with cancer after taking valsartan, you may be eligible to file a lawsuit. The first step in filing a lawsuit is to consult with an attorney. An attorney can help determine whether you have a valid claim for damages. Next, you need to understand the statute of limitations and make sure that your claim is filed timely.
Working with your attorney, you will need to gather evidence, including the necessary documents to prove your case. Finally, your legal representative will assist you in filing the appropriate paperwork to start your claim.

Valsartan Payout and Settlement Amounts
While payouts will vary depending on the severity of the injury, valsartan usage, and type of cancer, anticipated average settlements in Valsartan lawsuits are expected to be between $100,000 and $400,000.
Settlements are generally categorized into three categories based on the individual’s injury severity, drug usage, age, and cancer type. Top-tier settlements could be $400,000 or more. Middle-tier settlements will potentially be between $200,000 and $300,000. Bottom-tier payouts may be $100,000 or less. The estimated average settlement is expected to be between $150,000 and $200,000.
Contact a Valsartan Lawsuit Attorney Today
If you were diagnosed with cancer after taking valsartan, you may be eligible to take legal action. It is important to act fast to ensure compliance with all statutes of limitations and other legal deadlines. Contact King Law today to schedule a free, no-obligation consultation.