Complete the form below to see if you qualify for a Powerport Lawsuit

People are filing Bard PowerPort lawsuits alleging the catheter device caused injuries like device fractures, infections, blood clots, hemorrhage, and heart punctures. The lawsuits claim that Bard Access Systems, Inc., a subsidiary of Becton, Dickinson and Company, failed to properly warn patients of these risks. People who suffered injuries from Bard’s PowerPort catheters can speak with an attorney about filing a lawsuit as part of this litigation.
Bard PowerPort Lawsuit Overview
The Bard PowerPort is a port-a-cath, which is a port catheter device that is implanted under the patient’s skin to allow easy access to a vein. Port catheters are commonly used for medical IV therapy or the delivery of fluids.
The Bard PowerPort was approved by the FDA in 2000 as an implanted port catheter device. However, lawsuits allege that design and manufacturing flaws make the device prone to breakage (fracturing) and migration, causing blood clots, serious injuries, such as cardiac punctures, and severe infections.
Due to injuries allegedly caused by the port-a-cath, Bard is facing product liability lawsuits, known in the legal community as the Bard PowerPort lawsuits. These lawsuits claim that patients who had a Bard PowerPort implant may have a higher risk of complications or injuries due to catheter failure.
In August 2023, the Bard lawsuits were consolidated as a federal “multi-district litigation” before Judge David G. Campbell. The case is MDL 3081 and officially named In re: Bard Implanted Port Catheter Products Liability Litigation (MDL 3081).
If you, your family member, or loved one had a Bard PowerPort catheter device and subsequently suffered injuries or negative health effects due to fracture, migration, or other failures of the implant, you may be eligible for compensation. Contact an experienced product liability lawyer to get your case evaluated and prepared for a port catheter lawsuit.
Bard PowerPort and Port Catheter Lawsuit – 2026 Update
May 8, 2026: First Bellwether Trial in Bard PowerPort MDL Decided in Favor of Defendant
A jury in the U.S. District Court for the District of Arizona found that Becton Dickinson and Company and other defendants were not liable in the first Bard PowerPort lawsuit. In this case, a man alleged that the companies were liable for his injuries and failed to warn him of design defects and associated risks of PowerPort catheters. This trial was the first bellwether trial in the Bard PowerPort product liability MDL. Although this verdict is a setback for plaintiffs, the jury could not reach a verdict in the design defect claim. So, this issue may be tackled in future bellwether cases, which are scheduled for later this year. Meanwhile, plaintiffs’ lawyers will take the knowledge learned from this first trial and apply it to future litigation. Those alleging injuries from their PowerPort catheters can still speak with an attorney about eligibility for filing a claim.
May 1, 2026: Nearly 3,200 Bard PowerPort Lawsuits In MDL as Bellwether Trial Continues
There are 3,187 lawsuits centralized in the federal lawsuit against Bard over its allegedly defective PowerPort catheter. Some patients who had the PowerPort catheter implanted to help their doctors administer chemotherapy treatment say they were not warned about the potential risks of infections and other complications. One of the first trials in the Bard PowerPort MDL began on April 21, 2026. According to reports, the patient’s wife has testified about how their doctor had recommended that they use the PowerPort catheter to administer many of the chemotherapy treatments at home rather than make frequent, lengthy drives to the clinic. However, according to the man’s legal complaint, they were not told that the catheter carried an increased risk of infection.
April 13, 2026: Jury Selection Continues as First Bard PowerPort Bellwether Trial Date Nears
In the Bard PowerPort MDL, the judge has approved a list of about 40 jurors who will be called in for more screening questions on April 21, 2026. The first Bellwether (test) trial for an individual Bard PowerPort lawsuit in the MDL is scheduled to start on April 21, 2026. Jury selection is an important part of the trial preparation process. More Bard PowerPort trials are set to occur later this year. More than 3,000 lawsuits about alleged Bard PowerPort complications have been consolidated into a federal MDL that is still accepting new claims from eligible patients or their surviving family members.
March 31, 2026: Parties Submit Proposed Jury Selection Agenda Ahead of Last Pretrial Conference
The parties (plaintiffs and defendant attorneys) in the Bard PowerPort MDL have submitted a proposed agenda for the upcoming jury selection process. The first individual trial in the Bard PowerPort MDL is scheduled for April 21, 2026, and the parties must pick a jury panel before that date. The agenda gives a list of proposed statements and questions that may be raised when the parties meet with potential jurors. For example, the parties suggest asking potential jurors whether they have any conflicts of interest that could influence their decision if they were to serve as jurors. The proposal has been submitted to the judge for review.
March 2, 2026: Nearly 200 More Lawsuits Join Consolidated Group Claim About Bard’s PowerPort Catheter
The number of lawsuits in the centralized MDL against Bard has increased to 2,828 as of March 2, 2026. Some legal complaints say that patients with a Bard PowerPort may have an increased chance of developing an infection, the device breaking off, and experiencing other complications. Bard is accused of failing to warn patients about some of the increased risks that may be associated with its PowerPort catheter. Patients who believe they were harmed by a PowerPort manufactured by Bard can talk to an attorney about eligibility to file a claim in the MDL.
March 3, 2026: One of the Bard PowerPort Bellwether Trials Is Postponed After Plaintiff Passes Away
The Bard PowerPort MDL has been pending for several years, and the first wave of Bellwether (test) trials is scheduled for 2026 and 2027. One of those Bellwether trial plaintiffs has passed away of cancer, months before their Bard PowerPort case was set to go to trial in July 2026. The plaintiff’s attorneys are working with the surviving family members to determine if they want to continue with the lawsuit.
February 4, 2026: First Bard PowerPort Case Approaches Trial, Over 100 More Lawsuits Join MDL
The parties continue to prepare for the first upcoming Bellwether trial in the Bard PowerPort consolidated case (called an MDL). The trial date has been rescheduled several times, and the currently scheduled date is April 21, 2026. This trial will allow the plaintiff’s attorneys to tell the plaintiff’s story and fight for a fair outcome to compensate him for his injuries. It may also help inform others in the Bard PowerPort MDL about which arguments may be effective during trials in other cases in the MDL. Currently, there are 2,674 active cases in the MDL against Bard PowerPort, an uptick of 129 from the previous month.
January 27, 2026: Man’s Bard PowerPort Breaks Off and Migrates to a Pulmonary Artery Months After It Was Implanted
A man had the Bard PowerPort catheter device implanted so his doctors could give him chemotherapy and other treatments. About six months later, he went to the hospital because of chest pain, a rash, and itching at the PowerPort’s site. His doctors were concerned that he had a PowerPort-related infection, so they removed the device. After the operation, they discovered that a piece of the device had broken off and traveled into his left pulmonary artery (one of the major arteries that goes from the heart to the lungs). His doctors had to go through an artery in his leg to find and remove the missing piece. Other patients have experienced similar complications, and some are suing Bard, alleging the company did not properly warn them about this potential risk.
January 21, 2026: Over 2,500 Bard PowerPort Lawsuits Consolidated in Federal MDL Over Complications and Defects
The number of active lawsuits in the federal group claim against Bard PowerPort has grown to 2,545 as of January 5, 2026, an increase of 82 from December 2025. Bard PowerPort patients and their surviving loved ones are suing Bard after patients developed complications, including infections, device breakage, and device migration. Other patients suffered from blood clots, which required them to be hospitalized and undergo treatment. The people filing the lawsuits believe that Bard was aware of the risks of these complications but did not properly inform patients. As this litigation progresses, bellwether trials are scheduled for 2026 through 2027, and more lawsuits are being added each month.
January 6, 2026: Court Approves Modified Bellwether Trial Schedule in Bard PowerPort MDL
Thousands of Bard PowerPort patients suffered from complications after they had the device implanted to make it easier for their doctors to give them intravenous treatments. Legal complaints indicate that many affected Bard PowerPort patients say they were not told that infections, device migration, device breakage, or other adverse events could occur. Enough Bard PowerPort patients filed lawsuits about these complications that a federal MDL (a type of group case) was formed. The court has approved an updated schedule for upcoming test trials (bellwether trials) for a select number of cases in this MDL. The first Bard PowerPort trial of the MDL will be held on April 21, 2026. Meanwhile, more patients who suffered complications from a Bard PowerPort implant can investigate filing new claims and joining the MDL.
>>> Read Past Bard PowerPort Lawsuit Updates Here
About the Bard PowerPort Lawsuit:
What is Wrong with the Bard PowerPort Device?
Common Bard PowerPort Complications and Injuries
Bard PowerPort Lawsuit Allegations
Manufacturer Recalls of Bard PowerPort Devices
How to File a Bard PowerPort Lawsuit
Eligibility Criteria for Filing a Bard PowerPort Lawsuit
PowerPort Settlement and Payout Amounts
What is Allegedly Wrong with the Bard PowerPort Device?
Patients have experienced various degrees of PowerPort complications from Bard PowerPort usage. Lawsuits claim that the Bard PowerPort has a faulty design sometimes causing the catheter tube to crack, migrate, and/or leak. These defects significantly increase the risk of infections, injuries, or incorrect/insufficient delivery of essential fluids or medication.
PowerPort catheter tubes are made of ChronoFlex, which is made from a mixture of silicone or polyurethane and barium sulfate. Barium sulfate is used to make the device visible on X-rays.
A PowerPort’s materials can degrade after they come in contact with the bloodstream. The barium sulfate used in the ports allegedly degrades polyurethane and silicone after the port is inside the body. The barium sulfate particles do not seem to fully integrate into the catheter polymer and air pockets tend to form, allegedly making the material fragile. Additionally, due to high concentrations of barium sulfate, the material may become less durable. Then, as the patient moves, the catheter may bend and can fracture, subjecting the patient to the risks of thromboembolism and blood clots.
Catheter pieces can cause significant internal organ damage if they get into the bloodstream. Emergency surgery may be required to remove fractured pieces and treat affected organs. The cost of removing a PowerPort can be thousands of dollars.
According to lawsuits, Bard allegedly knew about PowerPort catheter fractures, migrations, and infections. Bard’s failure to inform patients and medical providers and to recall the product in a timely fashion resulted in serious injuries and deaths, and led to patients and family members to file lawsuits hoping for justice.
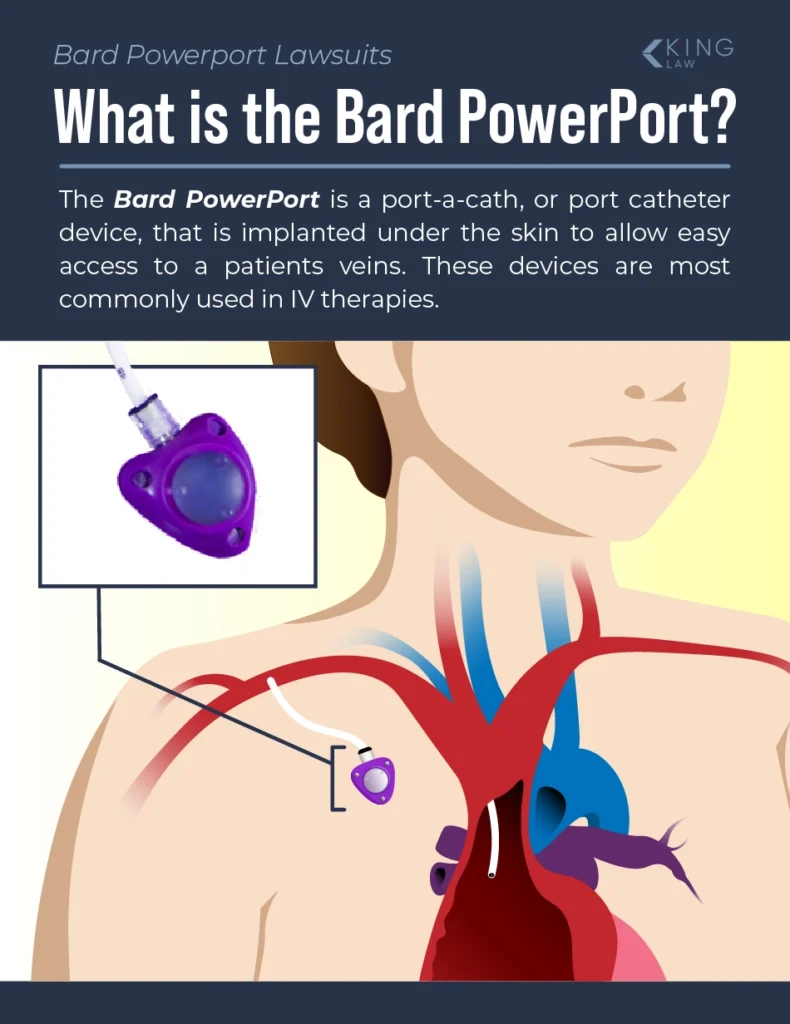
Bard PowerPort Design
Before we discuss injuries associated with Bard PowerPort catheters, it is important to understand their design and the materials used to make them.
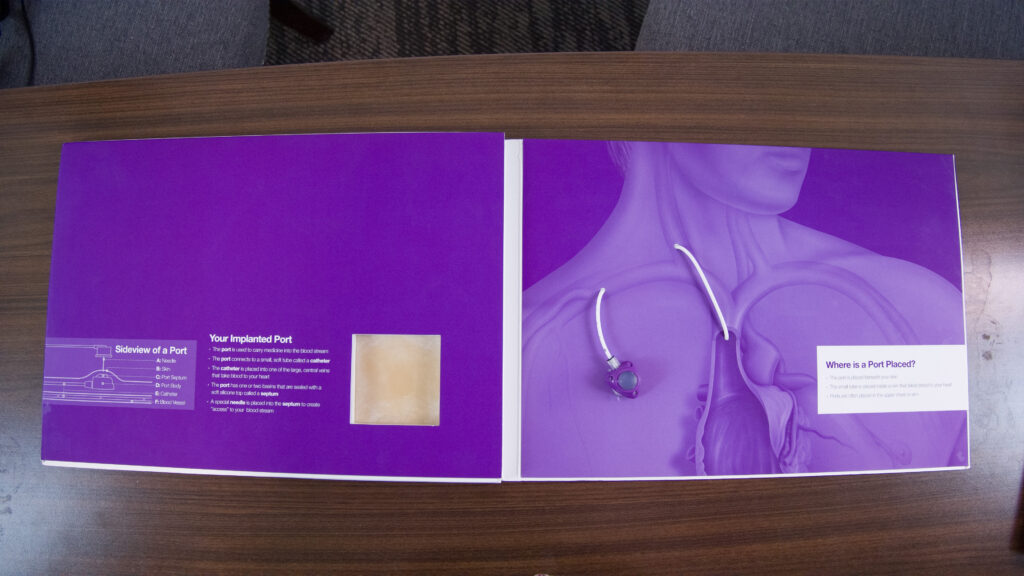
The Bard PowerPort is an implantable catheter that is made up of two main parts:
- The injection port: a small, self-sealing port that is implanted under the skin close to a vein. It has a silicone septum so a needle may pass through the skin and then through the septum to inject medication directly into the reservoir for an IV treatment or blood transfusion.
- The catheter: a flexible tube that connects the injection port to a vein. The body of the catheter is made of polyurethane material and is radiopaque (it is visible on X-rays/MRIs). Some Bard PowerPort catheters are made using ChronoFlex (a family of biocompatible aromatic polycarbonate-based thermoplastic elastomers). ChronoFlex is supposed to be more durable than polyurethane.
| Component | Material |
|---|---|
| Injection port | Silicone |
| Catheter | Polyurethane or ChronoFlex |
Who Uses Bard PowerPort Catheters?
The Bard PowerPort is used to deliver medications, fluids, and blood transfusions. It is often used in cancer patients who are receiving chemotherapy or other cancer therapies. PowerPort is also used in patients with chronic kidney disease, people who suffer from irritable bowel disease (IBD), or patients who need regular blood transfusions.
These catheters allow medical professionals to have direct and repeated access to a vein in the body’s vascular system. People suffering from other medical conditions may benefit from these devices.
These ports can increase a patient’s comfort during medical treatments. The ports also reduce the risk of tissue damage. Once the patient heals from device insertion, they can then swim and bathe normally, without an increased risk of infection.
Although these ports are helpful for many people, port insertion and use can have serious side effects. One study found that 6% of patients who had implanted PowerPorts experienced a post-surgical complication, including infection, catheter pinching, or port extravasation (extra-vascular leaking).
PowerPorts for Chemotherapy
Port catheters are a common way to deliver chemotherapy drugs to cancer patients. Cancer patients often endure multiple blood draws, infusions, and injections. A port catheter allows doctors to administer these treatments and tests. Doctors can also use the ports to provide IV fluids and administer blood products such as platelets and plasma. The ports make administering these treatments easier and provide greater comfort to patients during such treatments. The catheters are supposed to protect a patient’s veins from injuries caused by repeated access.
PowerPorts can also be used to give patients IV contrast for imaging scans. Cancer patients often have MRIs or CT scans, so the ports give medical professionals another way to inject contrast for these tests.
PowerPorts for IBD and Other Conditions
Because PowerPorts access the body’s vascular system, they are used for a variety of reasons. People with bowel disease or severe infections may also be good candidates for port catheters. Because medical professionals can use the ports to administer IV fluids, blood products, and parenteral nutrition (feeding someone through a vein), they are used for a variety of conditions, including IBD.
Common Bard PowerPort Complications and Injuries
Patients have suggested that the PowerPort device is less durable than it should be. A growing number of lawsuits point out a flaw in the design of the ChronoFlex catheters that results in the following Bard PowerPort problems:
- Catheter fracture injuries
- Catheter-related infections
- Catheter migration
Bard Catheter Fracture Injuries
The PowerPort catheter tube is prone to fracturing, which can lead to small pieces of plastic breaking away and getting into the vascular system, potentially leading to life-threatening complications. This means some PowerPort patients could experience the following injuries:
- Cardiac arrhythmia
- Pulmonary embolism
- Cardiac punctures
- Blood clots
- Hematomas
- Lacerations to blood vessels
- Cardiac/pericardial tamponade (pressure caused by an accumulation of fluid in the area around the heart)
- Thromboembolism
- Perforations of tissue, vessels, and organs (e.g., heart or lungs)
Bard catheter failure can also result in severe and persistent pain. Emergency surgery may be needed to remove fractured pieces and treat affected organ systems.
Bard Catheter Infection Injuries
Patients with a Bard catheter may develop infections due to bacteria entering broken areas of the PowerPort. Common symptoms of an infection include:
- Fever
- Chills
- Inflammation, redness, swelling or pain around the catheter site
- Drainage or pus
- Skin discoloration
- Numbness
- Confusion
Complications from catheter infection can be serious and even life-threatening.
Infection around the catheter site can lead to bloodstream infections, severe swelling, and even necrosis of tissues around the PowerPort.
Bard Catheter Migration
Catheter disconnection and migration are rare but serious complications associated with Bard PowerPort. It can lead to side effects such as venous thrombosis and neurologic problems.
Patients experiencing catheter migration can be asymptomatic, which makes diagnosis and treatment more difficult. The most common port-a-cath migration symptoms include:
- Neck pain
- Shoulder pain
- Chest pain
- Ear pain
- Heart palpitations
Entire catheter dislodgement is even more rare, but they are possible and may require immediate surgery.
Catheter migration can lead to obstruction of blood flow, infection and organ damage.
Chemo Port Injuries
Medical literature notes the FDA received more than 250 adverse event reports involving vascular access devices rupturing, when used with power injectors for contrast media. Some lawsuit allege that PowerPorts ruptured when patients are receiving IV contrast for CT scans or MRIs. Shortly after the PowerPort came to market, the FDA received more than 250 adverse event reports about vascular access devices that ruptured when contrast media was injected using power injectors. These reports documented that some power injector ports ruptured when contrast media was given as part of CT or magnetic imaging.
This puts cancer patients and other people who need frequent imaging tests at risk for port rupture and the associated side effects.
Bard PowerPort Lawsuit Allegations
The Bard PowerPort lawsuits claim that the devices are defective, creating possible infections and catheter failures that can lead to thrombosis, organ damage, and even death. The lawsuits allege that Bard Access Systems, Inc., and its parent company, Becton Dickinson and Company, the manufacturers of the PowerPort, knew about the risks associated with the device, but failed to inform patients and medical professionals.
The lawsuits specifically note that the PowerPort is susceptible to fractures and infections. Some of the specific injuries that have been reported in connection with the PowerPort include:
- Thrombosis
- Damage to veins and blood vessels
- Necrosis
- Blood clots
- Cardiac/pericardial tamponade
- Cardiac arrhythmia
- Perforations of vessels, organs, and tissue
- Chronic pain
Manufacturer Recalls of Bard PowerPort Devices
In 2019, Bard initiated a recall for multiple lots of PowerPort devices. The following products were affected:
- PowerPort Implantable Port With Attachable 9.6 F Open-Ended Single-Lumen Venous Catheter REF: 1709600, Air Guard, With suture Plugs, (01)00801741026720;
- PowerPort Implantable Port With Attachable 9.6 F Open-Ended Single- Lumen Venous Catheter, REF: 1709601, Air Guard, Without Suture Plugs, (01)00801741026737;
- PowerPort Implantable Port with Pre-Attached 9.6F Open-Ended Single-Lumen Venous Catheter, REF: 1759600 Air Guard, With Suture Plugs, (01)00801741026850
If you have a Bard PowerPort implant, it is important to consult your doctor immediately to see if your catheter is affected by the recall. You can also check the FDA’s website for a list of all recalls of Bard PowerPort devices.
The FDA listed it as a Class II recall in March of 2020 and terminated the recall in February of 2022. The FDA issued the recall because of an incorrect barb housed within the device, not because of fractures or other defects. If you were affected by the recall, you should contact an attorney to discuss your options.
How to File a Bard PowerPort Lawsuit
If you or someone you know has been injured by a PowerPort, please contact one of our experienced attorneys. We offer free case reviews. We will help you navigate tasks such as:
- Collecting evidence to support your claim, including medical records and witness testimonies
- Determining state-specific deadlines and statute of limitations
- Handling settlement negotiations
- Setting the case for trial if a favorable settlement cannot be reached
How Much Time Do You Have to File a Lawsuit?
Because of statutes of limitations, it’s important to consult with a lawyer promptly. If you have been harmed by a PowerPort catheter, you should contact an attorney as soon as possible after your injury. Each state has its own laws about how much time can pass between an injury and a lawsuit filing. Our attorneys can help you understand which state you can file your case in and states’ filing deadlines. They will help you achieve the best possible outcome.
What to Expect During the Legal Process
Our lawyers will work with you throughout the legal process for filing a Bard PowerPort lawsuit. We recommend working with a lawyer who has experience handling similar cases. Here are the steps involved:
- Consult an attorney. An experienced Bard PowerPort attorney can help you evaluate your case and determine if you are eligible to file a claim. They can also help you understand the legal process and timeline.
- Gather evidence. You will need to gather evidence to support your claim. This usually includes medical records, photographs, witness statements, and medical bills.
- With the help of your lawyer, file a lawsuit. Once you have gathered evidence, you will need to file a lawsuit against Bard Access Systems, Inc., and its parent company, Becton Dickinson and Company.
- Discovery process. Both sides gather evidence and documentation. Your attorney will support you throughout this process and advise what documentation will best support your case, such as medical records, bills, and proof of payment.
- Settlement or trial. You and your attorney may choose to pursue a settlement. If the case doesn’t get settled, it could go to trial. At trial, a jury will decide whether Bard is responsible for your injuries and subsequent suffering/loss of income/loss of consortium/etc. If the jury finds Bard responsible then they will determine the payout amount you are entitled to.
- Appeal. If you are not satisfied with the outcome of the trial, you and your lawyer may appeal the decision.

Eligibility Criteria for Filing a Bard PowerPort Lawsuit
Individuals who have received a diagnosis for an adverse health condition after receiving a PowerPort catheter may be entitled to take legal action.
Eligibility criteria for filing an PowerPort lawsuit include:
- Proof that you had a Bard PowerPort implantable catheter inserted
- A diagnosis of an adverse health condition, such as cardiac arrhythmia; pulmonary embolism; cardiac punctures; blood clots; hematomas; lacerations to blood vessels, cardiac/pericardial tamponade; thromboembolism; or perforations of tissue, vessels, and organs (e.g., heart or lungs)
- An emergency room visit, hospitalization, or similar medical visit related to the condition allegedly caused by the faulty PowerPort device
It is critical to consult with a PowerPort lawyer to determine if you meet the criteria requirements and for state-specific statutes of limitations that may affect your case.
PowerPort Settlement and Payout Amounts
It is difficult to predict the Bard settlement amounts. Further, the amount of compensation you could receive from a PowerPort lawsuit settlement will depend on the severity of your/your loved one’s injuries. It is also important to note that compensation or even favorable outcomes of the trial are not guaranteed.
Possible damages may include:
- The cost of past and future medical treatments
- Legal fees
- Lost wages
- Reduced future earning potential
- Decreased quality of life
- Physical and emotional pain and suffering
- The loss of a loved one

PowerPort Lawsuit Payout Amounts
If your case goes to trial, the specific amount of compensation you receive will depend on the individual circumstances of your case. An attorney will work with you to understand and document the losses you have experienced. For a free case evaluation, consult our team of experienced product liability and personal injury lawyers.
Factors That Determine Settlement Amounts
There are multiple factors that would affect the amount a patient receives from a successful settlement. If you were harmed by a PowerPort device, your individual factors and hardships would be considered. It’s important to consult an experienced attorney who can document the losses you experienced and fight for a fair and full settlement.
Contact a Port Catheter Injury Lawyer
At King Law, our PowerPort lawyers provide dedicated representation for individuals who have suffered adverse health conditions as a result of having a port-a-cath implanted to receive medical treatment. We have years of experience advocating on behalf of those injured by defective medical products.
We are a team of experienced personal injury lawyers with a strong commitment to our clients. Call us todayfor a free case evaluation to see if you qualify for the Bard PowerPort lawsuit.

Other News on Bard PowerPort
In November, a woman in North Carolina filed a legal complaint against Bard, the company that made the PowerPort catheter. Her doctors implanted the PowerPort catheter in 2023, and she suffered an infection and thrombosis (blood clot) afterward. She believes the Bard PowerPort device’s design is to blame for these complications. Dozens of other patients have experienced blood clots, infections, device migration, and other side effects after having the Bard PowerPort implanted. Like other patients in the MDL, she is seeking compensation to hold Bard responsible for not warning her about the risks. Between November and December of 2025, 336 new plaintiffs filed lawsuits against Bard for defects with its PowerPort device. There are 2,463 active lawsuits in this group litigation for PowerPort injuries. People who experienced infection, migration, perforation, and similar malfunctions of their Bard PowerPorts can still seek compensation and file as part of this active litigation. A woman in Minnesota has filed an updated legal complaint against the manufacturers of the Bard PowerPort devices. She had a Bard PowerPort implanted in 2022 and experienced a severe infection because of it. She believes the manufacturers were aware of the increased risk of infections and other complications and should have told people about it. She joins hundreds of other patients and their surviving family members who have filed lawsuits involving port device injuries. Bard’s PowerPort have been known to injure people who have the catheters implanted. People who have been injured have joined a group lawsuit against Bard. The judge overseeing that litigation (Judge Campbell) has asked attorneys to ready jury-selection questionnaires for PowerPort lawsuits. Additionally, he asked attorneys to prepare information for a Status Conference on December 4, 2025. This litigation is moving forward, as lawyers and plaintiffs prepare for test (bellwether) trials and jury selections in 2026. There have been 2,065 lawsuits filed against Bard for its faulty port catheter device. However, as of October 2025, there are 1,973 active cases in the group litigation for people harmed by Bard’s PowerPort Catheters (MDL -3081 IN RE: Bard Implanted Port Catheter Products Liability Litigation). Each of these cases represents someone who allege serious injuries when their device fractured, migrated, caused blood clots or lacerations, or became infected. People who were harmed by Bard’s port catheters may still be able to join the group litigation. Lawyers continue to prepare for the upcoming Bellwether trials in the Bard PowerPort federal lawsuit. PowerPort patients suffered from infections, device migration, and device breakage, and they believe these were known complications that Bard should have warned them about. The discovery process allows the plaintiffs’ attorneys to learn more about the complications that the plaintiffs experienced, the safety data for Bard PowerPort, and other critical evidence. Bard PowerPort patients are continuing to file lawsuits after they endured infections, revision surgeries, and other losses due to these defective devices. Judge David G. Campbell has issued a scheduling order with new deadlines for one of the Bard PowerPort cases set for an upcoming Bellwether trial. Events with new due dates include medical expert disclosures, depositions (interviews conducted under oath), and motions. Adjusting these deadlines will allow the plaintiffs’ legal teams to prepare for the intensive discovery process, which helps them find evidence to prove Bard’s PowerPort was defective. More Bard PowerPort patients who experienced severe complications can join in the federal multidistrict litigation. In the federal Bard PowerPort group litigation, Judge David Campbell approved an amended schedule for when the parties (lawyers) need to disclose the nursing experts they may interview during discovery. Medical experts can provide critical insight into the patients’ condition before and after receiving the PowerPort and how complications were identified and addressed. Hundreds of patients suffered severe adverse events after they had the Bard PowerPort implanted into their bodies. Patients experienced severe infections, device migration, and pieces of the device breaking off and migrating to other places. The lawsuits allege that Bard was aware of the high risk of complications, but it did not design a safer system or warn patients of these dangers. Many people who were harmed by C.R. Bard’s implantable port catheters have joined a group lawsuit called a multidistrict litigation (MDL). As part of the MDL process, several test trials called bellwether trials take place. These initial trials are important, because they often set the tone for the rest of the plaintiffs in the group. The judge overseeing these lawsuits, U.S. District Judge Campbell, has issued an order scheduling these test trials for people harmed by PowerPorts. The first six Bard PowerPort trials will take place from March through December of 2027 (this trial schedule would later be amended). People harmed by Bard’s PowerPort catheters can still join this group litigation and should consult an attorney about eligibility. The judge overseeing the multidistrict litigation against Bard has approved the final selection of six Bard PowerPort lawsuits slated for trial. The judge has also set August 28, 2025, as the deadline for the parties to interview one of the witnesses, Kelly Christian. The defective Bard PowerPort has a history of breaking off in the patient’s body, and more victims can join in this active litigation. The number of lawsuits in the Bard PowerPort multidistrict litigation has increased to 1,763, a rise of over 100 from June. Patients experienced severe and sometimes fatal complications after having the PowerPort implanted to make it easier to administer treatments. More people are encouraged to file a claim if they were hurt or their loved ones were killed by Bard PowerPort complications. More than 1,600 federal lawsuits are pending against Bard for its defective PowerPort device. A Case Management Conference was held on June 3, where the attorneys discussed many topics relevant to litigation. One of the most notable topics was requested adjustments made to deadlines in the litigation. Bard’s attorneys want a 90-day extension, based on multiple factors. Delays of this nature are typical, and the fact that this litigation is moving toward fall 2025 bellwether trials is promising. This means that settlements and resolutions could be near for people harmed by Bard’s port catheters. The multidistrict litigation against Bard has grown to 1,410 cases in May, an increase of about 300 since March. Patients are suing because they experienced complications, such as life-threatening infections, after having Bard’s PowerPort implanted. The litigation is ongoing, and the parties are moving through the trial preparation process. Patients who were harmed or whose loved ones died because of complications can join in this active lawsuit. The deadline for attorneys on both sides of the PowerPort litigation to agree on pre-trial rules was April 7. Attorneys are working to resolve some issues before bellwether (test) trials begin later this year. Before this deadline, attorneys sent a status memo to the judge overseeing the Bard PowerPort MDL to update the judge on unresolved legal issues. For example, the defendants want to prevent some of the plaintiffs’ experts from testifying, claiming the plaintiffs did not give proper notice. The plaintiffs believe they sent in the notice on time. The parties will discuss this issue in court, and the judge will make a decision. These sorts of issues are common in mass litigations. These developments show lawsuits against Bard’s PowerPort device are moving forward. A West Virginia woman passed away just two months after filing a lawsuit against Bard PowerPort. She and her wife, Shannon, had sued Bard because she had a port catheter implanted in 2019 and developed an infection afterward. They filed their complaint on August 8, 2024. If her infection or the PowerPort catheter is responsible for her passing, her loved ones may file a lawsuit against Bard for wrongful death. The Bard PowerPort manufacturer now faces 1112 lawsuits in the massive federal litigation filed against it, an increase of 108 cases from the previous month. Since 2018, nearly 1,200 cases have been transferred to the active, consolidated federal lawsuit. Patients and surviving loved ones are suing Bard because they believe its catheter’s defective design caused patients to experience severe complications with sometimes fatal consequences. There are 1,004 lawsuit pending against Bard Access Systems, Inc., owned by Becton, Dickinson and Company. Plaintiffs claim that the PowerPort catheter’s design is flawed and has caused serious complications, including death, infections, heart punctures, and blood clots. February’s case numbers represent an increase of 125 Bard lawsuits from December. The case count is expected to grow as more people take a stand in this active lawsuit. Judge David G. Campbell entered an important discovery order in the PowerPort lawsuits on January 3, 2025. This document resolves disagreements each side had about handling different discovery matters. Additionally, the court order gives deadlines for each side to exchange information and interview people, such as expert witnesses and treating physicians. The parties can use the information gathered during discovery to help support their legal arguments or discredit the other side’s claims. On December 3, 2024, co-lead attorneys for the plaintiffs in the Bard PowerPort litigation filed a second amended master complaint. This document outlines some claims being made by multiple plaintiffs against Becton Dickinson. For example, some claims include a dangerous product design and failure to warn patients of the risks. Future plaintiffs can join this second amended complaint by filing their own short-form complaint. On December 2, 2024, the plaintiffs and defendants in the Bard PowerPort multidistrict lawsuit (MDL) submitted a joint case memorandum to summarize the status of the lawsuit to date. The memorandum says there are currently 879 individual lawsuits pending within the larger multidistrict litigation against Becton Dickinson, the makers of Bard PowerPort. The document also summarizes each side’s position about important matters like turning over key case documents in preparation for trial. Traci Christine Hicks and Robert Eugene Hicks filed an amended complaint on November 5, 2024. Their original lawsuit, Traci Christine Hicks v. Becton Dickinson and Company, et al., was filed October 3, 3024. In Christine’s original complaint, she said she suffered an infection because of complications with a PowerPort Implantable Port. By amending this complaint, Robert has been added to the case, claiming loss of consortium. Traci and Robert requested a jury trial on their claims against Bard, Becton, Dickinson and Company, and others, including failure to warn and strict liability for the product’s design defects. Christine DeStefano’s estate filed a Complaint on October 28, 2024, in the PowerPort lawsuit against Bard, Beckton Dickinson, and other manufacturers. Christine’s doctors put in a PowerPort Implantable Port in 2016 and again in 2021. The complaint says the person in charge of Christine’s estate believes an infection from the PowerPort may be why Christine died. The defendant companies should file an answer to this complaint in the coming weeks. In the most recent lawsuit filed in the Bard PowerPort litigation, a Chuathbaluk, Alaska man alleges that the PowerPort ClearVUE Slim Implantable Port he was implanted with in February 2020 fractured and migrated, causing serious injury. The plaintiff demands punitive damages due to the defendant’s negligence, failure to warn of the risks associated with the implant, among other complaints. In the most recent Bard PowerPort lawsuit filed in Arizona federal court, the plaintiff, a Tennessee man, was implanted with the Bard PowerPort ISP M.R.I. Implantable Port on December 20, 2020. The claim alleges that the port caused infection and blood clots, resulting in injury. The plaintiff asserts claims against the defendants, including CR Bard, for failure to warn, negligence, manufacturing defect, misrepresentation, and breach of implied warranty. On August 15, 2024, a Louisiana woman filed a Bard PowerPort lawsuit in the Multi-District Litigation in Arizona, alleging design defect, failure to warn, manufacturing defect, among other causes of action. In the case Davidson vs. Becton, Dickinson and Company, et al., the plaintiff was implanted with a PowerPort ClearVUE ISP Implantable Port on January 8, 2018, and subsequently experienced fracturing of the catheter device. A fracturing injury can be particularly dangerous, as the plastic components of the device can break free, migrate through the vein, and potentially reach the heart, leading to serious complications, including death. The complaint filed in Prentice v. Becton, Dickinson and Company; CR Bard Inc., Bard Access Systems provides a detailed overview of the Bard PowerPort port-catheter lawsuit. The twenty-two-page complaint was filed on April 14, 2023. The plaintiff alleges that the vascular access device was implanted on January 10, 2022, with the BardPort for chemotherapy in the treatment of breast cancer at a hospital in Phoenix, Arizona. A CT scan showed the port had fractured and moved. Emergency surgery was performed to retrieve the broken port and implant another one. The complaint alleges that the defendants knew the port might fracture from prior reports of failure. It also claims defective design, manufacture, and negligence. Additionally, the complaint alleges that ports have caused other injuries in other plaintiffs, like hemorrhage, perforation of tissue, cardiac arrhythmia, and pericardial tamponade. As of July 1, 2024, there are 299 pending actions in the PowerPort multidistrict litigation lawsuit. This is a large increase from the 69 pending actions in January of 2024. This means more people are taking action after being injured by faulty PowerPort devices. Hon. David G. Campbell, the judge overseeing the Bard PowerPort Lawsuit, will hold the ninth case management conference on August 16, 2024. The Court has ordered the parties to provide a summary of the topics to be discussed by August 14, 2024. The likely talking points should include: Group 1 cases for discovery, the status of cases filed in state courts, access to discovery (things like documents, emails, notes, etc.) in the possession of the defendants and their custodians (record keepers), and depositions of witnesses. The Bard Implanted Port Catheter MDL’s next status conference is scheduled for July 8th, and we expect to see minor developments as the defendant continues to submit files for the discovery phase of this litigation. We hope to gain a better understanding of the timeline for this lawsuit after the status conference. The parties filed a joint memorandum in the Bard PowerPort Lawsuit on May 22, 2024. The document outlines the current position of the parties. There have been fewer than a million documents produced by the defendants, which is actually a low number in a major medical device lawsuit like this one. The defense argues that there were only 11 lawsuits filed in the several years prior to the attorney advertising related to the multidistrict litigation. The plaintiffs say that the defendants are minimizing serious injuries, including fractures to PowerPorts that lead to permanent injury or death. The defense says it is spending $350,000 per week to review documents to turn over in this case. We anticipate the discovery fights to continue in the PowerPort Lawsuit. The Bard PowerPort Lawsuit is in the height of discovery negotiations. Case Management Order number 23, filed on May 24, 2024, by Judge Campbell, highlights several agreements as well as outstanding discovery issues in the case, which is technically referred to as IN RE: Bard Implanted Port Catheter Products Liability Litigation, MDL 3081. The order is setting up the issues for the eighth Case Management Conference, which is scheduled for July 9, 2024. Discovery is the term to describe how the parties exchange information the other side is entitled to in a lawsuit. There are discovery disputes in virtually every major lawsuit. This is an important part of the case, even though it doesn’t seem exciting. The agreements on discovery will directly affect what evidence the plaintiffs have at the time of trial. Plaintiffs are interested in knowing how much Bard knew about the potential failure of the medical devices. Judge Campbell is keeping a close eye on this litigation and is showing himself as a skilled and effective judge so far in the Bard PowerPort litigation. The Bard PowerPort Lawsuit received a new update on May 13, 2024, in the form of Case Management Order Number 22 from Judge Campbell. The parties are in the process of selecting Bellwether (test) cases. The defendants have raised concerns about several cases that have been filed to date in the PowerPort lawsuit. The biggest complaint is incomplete Plaintiff Profile Forms (PPFs). Bard, the defendant, has also raised concerns about the number of documents requested by the plaintiff. This objection is known as proportionality. In a big lawsuit, plaintiffs can get a lot of information. In a small lawsuit, the plaintiff can’t get as much background information about the company. Generally, a major MDL like the Bard PowerPort Lawsuit entitles the plaintiffs, through the plaintiffs’ lawyers, to get a lot of corporate information. The scope is not limitless, however. The judge scheduled a remote status conference for May 24, 2024, on the issues. On April 23, 2024, United States District Court Judge David Campbell issued an order about how depositions would be handled in the Bard PowerPort Lawsuit. Depositions are the sworn statements of witnesses in cases prior to the time of trial. The most interesting provisions of the order centered around the Judge’s preference for remote depositions of witnesses. In a major federal court lawsuit like the Bard PowerPort litigation, where there are hundreds of millions or billions of dollars at stake, each deposition can be critical to the case. It seems surprising that the parties would be comfortable with depositions that were not in person. Most lawyers would agree that it is easier to examine someone in person and there is something lost with remote depositions. Another interesting provision is the section on cross-noticing of depositions. This means there might be state court cases that proceed on similar issues, and the lawyers will attempt to coordinate with other groups of lawyers with cases outside of federal court. There were two important agreements between plaintiff and defense attorneys in the Bard PowerPort lawsuit. By a stipulated Order dated April 4, 2024, the parties have agreed that the defense will be entitled to certain PowerPort pathology that is in the plaintiffs’ possession and the plaintiffs will also provide HIPAA-compliant authorizations, so the defense can get the plaintiffs’ certified medical records. Another Order filed by the judge indicates how the parties will handle documents that they say are privileged and should not be turned over to the other side. The order refers to privilege logs. Privilege logs tell the other side there is a document, but we don’t think you should be able to see it, usually because it was the subject of attorney-client privilege. The PowerPort lawsuit seems to be moving slowly forward. One of the central issues in the Bard PowerPort lawsuit will be the types of injuries that the manufacturer (Bard) knew the PowerPort might cause. One important study the lawyers will likely argue about is titled “Complications after implantation of subcutaneous central venous ports (PowerPort).” The study, by Takatoshi Nakamura, published in the Annals of Medicine & Surgery in May 2017, indicated that infection was a significant risk of PowerPort use. The study also noted the risk of significant injury associated with rupture or fragment release from the catheter, the risk of thrombus, the risk of sepsis, and “catheter pinch-off syndrome.” Studies like this would appear to place the manufacturer on notice of specific risks associated with the use of the Bard PowerPort. This week Hon David G. Campbell, the judge overseeing the Bard PowerPort lawsuit, approved the Plaintiff Profile Form (PPF). This 30-page document must be completed by the attorney filing a lawsuit on behalf of their client. The information provided is used as reference by both the defendant and the court. The PPF contains 8 sections, including Background Information; Claim Information; Medical Background; Health Insurance Information; Prior Claim Information; Fact Witnesses; Identification of Documents; and Document Requests. The approval of the PPF by the Court is an important step in the litigation. One of the next major milestones will be the approval of a Master Complaint and a Short Form Complaint. In an updated Case Management Order by Judge Campbell, the Court addressed concerns about the slow pace of cases filed and the overall size of the MDL. To date there are approximately 115 lawsuits filed in this case, which is organized in the District of Arizona federal court. With an upcoming date to begin choosing candidates for trials, the defendants expressed concern that the case has not grown to the numbers predicted by the plaintiffs, and with such a small sample to choose from, any cases chosen for trial may not be fully representative of all of the potential plaintiffs. In a previous case involving inferior vena cava (IVC filters), which was also handled by Judge Campbell, the Court noted that litigation also started very slowly but began to pick up significantly, with more than 80% of the cases being filed after 18 months. In light of this, Judge Campbell has decided that the timeline for selecting trial candidates and discovery deadlines will remain as scheduled. The bellwether trial selection process will begin on April 1, 2024. The Bard PowerPort lawsuit has been slow moving. Just over a half-dozen cases were added in February, with just 107 total cases filed in the multi-district litigation case. The PowerPort catheter lawsuit just became official in August of 2023, when all outstanding cases were transferred to federal court in the district of Arizona under Hon. David G. Campbell. With this case being brand new, expect several years of litigation before a full resolution. Consistent with the Court’s case management order, the Court has appointed a Certified Public Accountant to establish an account and record-keeping practices for the Common Benefit Fund and Expenses. In a case such as the Bard PowerPort lawsuit, the attorneys selected to represent the interests of all plaintiffs, are entitled to a portion of an award or settlement for their years of work. Importantly, this percentage comes out of the fee of attorneys representing plaintiffs, it is not paid by the client. This account, managed by an independent third party, will ensure transparency for all expenses incurred by the plaintiffs’ leadership and eliminate commingling of funds with attorney’s accounts. Litigation continues for the Bard PowerPort lawsuit. According to the most recent filing from the Judicial Panel on Multidistrict Litigation (JPML), 99 lawsuits are pending consolidation. The Judicial Panel on Multi-District Litigation (JPML) will soon rule as to whether post-reservoir defects will be added to the Bard PowerPort lawsuit. Their allegations deal with the use of polyoxymethylene, a type of plastic polymer, in the manufacturing of the port reservoir. That material can break down over time leading to splintering or fracturing of the device. This PowerPort defect may cause infections and perforation of tissue and organs. The JPML will rule on this issue on January 25, 2024. If the panel decides to include the Bard PowerPort reservoir defects in the lawsuit, the master complaints will be amended to include it the addition. After a very slow month of December where only a half dozen cases were filed in the lawsuit, we’ll get a progress update from the plaintiffs’ and defendant’s attorneys in a status conference to be held by Judge Campbell on January 8, 2023. With the Bard PowerPort litigation still in the early stages, fewer than 100 cases have been filed in this class action-style port catheter lawsuit. On November 30, 2023, Judge Campbell, who is overseeing the Bard PowerPort Port Catheter “class-action” style lawsuit in Arizona Federal District Court, issued an order approving the short form complaint. This document, which is a condensed version of the long-form complaint, details the allegations made by the plaintiffs that the devices caused serious injuries such as splintering, blood clots, organ perforation, infections and others. The complaint also outlines the specific products that are the basis of the lawsuit. After a plaintiff’s attorney builds the case for their client, they will file the short-form complaint with the court, initiating the lawsuit. The defendants (C.R. Bard) in the PowerPort Catheter class action lawsuit are asking the judge overseeing the case to strike certain allegations in the proposed master complaint. A master complaint is a document which outlines in great detail the allegations and injuries which are the basis of the lawsuit. Bard claims they improperly expand the scope of the multi-district litigation (MDL) lawsuit. The defendant contends that the plaintiffs are attempting to add new allegations that are not relevant to the focus of the lawsuit. The argument pertains to whether certain components of the product are defective. The defendants are asking a judge to rule on the issue prior to responding to the proposed master complaint. The judge responded and noted that he will take the request under consideration. Recently, Hon. David G. Campbell, the judge in charge of the Bard PowerPort Lawsuit held a conference to discuss motions and responses filed by both parties. Additionally, Court announced it will hold the first Case Management Conference on November 16, 2023, in federal court for the District of Arizona, where the case has been consolidated. The Court is also expected to make a decision on the selection of a Plaintiff’s leadership committee in the coming weeks. Lawyers selected for the leadership positions will be responsible for representing the best interests for all plaintiffs. Nearly 40 plaintiffs’ attorneys have just been proposed to hold leadership positions in the ongoing Bard PowerPort lawsuit. This proposal nominates the following professionals: 3 attorneys to serve as Co-Lead Counsel; 11 attorneys to serve on the Executive Counsel or Liaison Counsel; 13 attorneys to serve on the Steering Committee; and, 12 attorneys to serve as Sub-Committee Members. It will be the responsibility of leadership committee members to carry out specific functions throughout the litigation process. Some of these functions may include, but are not limited to, conducting investigations and interviews that have a shared interest in all claims, preparing and presenting arguments before the Court, and engaging in settlement discussions that lay the groundwork for resolving all claims related to Bard PowerPort lawsuit injuries. These attorneys are obligated to act in the best interest of every individual that has brought a claim for injury against Bard PowerPort, and that each individual claim will remain separate and distinct from one another. As a component of organizing these lawsuits, it is anticipated that the Judge will order the plaintiffs and the defendant to select several “test” cases for trial. These so-called “bellwether” trials will serve as a means of assessing how juries might react to specific injuries, evidence, and testimony. The results of these test trials will form the groundwork for later settlement negotiations. This litigation is still in the very beginning stages of a process that is expected to take several years. On August 8, 2023, the JPML granted that request and created MDL No. 3081 for people claiming injuries from Bard PowerPort devices. This centralized litigation is called In re: Bard Implanted Port Catheter Products Liability Litigation, Judge David G. Campbell in the District of Arizona oversees the cases. Multiple plaintiffs who had filed Bard PowerPort lawsuits filed a motion asking the Judicial Panel on Multidistrict Litigation (JPML) to create a multidistrict litigation (MDL). If this motion is approved, people alleging injuries from Bard’s PowerPort will have the option to join a federal, consolidated litigation.December 22, 2025: North Carolina Woman Sues Bard After PowerPort-Related Blood Clot
December 2, 2025: More Than 300 People Harmed by Bard PowerPorts Join Litigation
November 17, 2025: Woman in Minnesota Files Updated Bard PowerPort Complaint in Federal MDL
November 4, 2025: Attorneys in Bard PowerPort Lawsuits Ready Jury Questionnaires
October 23, 2025: Lawsuits Against Bard’s PowerPort Rise Over 2,000
October 17, 2025: Pretrial Discovery Process Continues in Bard PowerPort Lawsuit
October 2, 2025: Judge Sets Deadlines for Bard PowerPort Lawsuit Set for Bellwether Trial
September 17, 2025: Court Updates Medical Expert Disclosure Deadline in Bard PowerPort Lawsuit
September 4, 2025: Bellwether Trials Scheduled in Bard PowerPort Litigation
August 1, 2025: Judge Approves Final List of Bellwether Trials in Bard PowerPort MDL
July 3, 2025: Bard PowerPort Cases Approach 1,800 As More Victims and Surviving Loved Ones Take Action
June 3, 2025: Timeline Adjustments Made in Bard PowerPort Trials
May 1, 2025: Bard PowerPort Lawsuits Top 1,400 As Litigation Moves Forward
April 8, 2025: Attorneys in Bard PowerPort Lawsuits Try To Align on Pre-Trial Proceedings
March 25, 2025: West Virginia Woman Passes Away After Filing Bard PowerPort Lawsuit
March 3, 2025: Bard PowerPort Manufacturer Faces 108 More Lawsuits in Federal MDL Against It
February 4, 2025: More Patients File Lawsuits Against Bard PowerPort Manufacturer
January 9, 2025: Bard PowerPort Judge Enters Order Setting Key Discovery Deadlines
December 12, 2024: Bard PowerPort MDL Plaintiffs File Second Amended Master Complaint Against Becton Dickinson
December 2, 2024: Parties Submit Memorandum Ahead of Case Management Conference in the PowerPort MDL
November 7, 2024: Virginia Plaintiffs File Amended Complaint in Bard PowerPort Lawsuit
November 1, 2024: Plaintiff’s Estate Files Amended Complaint After Plaintiff’s Death
October 1, 2024: Alaska Man Files Bard PowerPort Lawsuit Alleging Fracture and Migration of Implant
September 24, 2024: Plaintiff Files Bard PowerPort Lawsuit Alleging Injuries from Defective Implantable Port
August 19, 2024: Louisiana Woman Files Bard PowerPort Lawsuit Alleging Serious Complications
August 1, 2024: Plaintiff Alleges Device Failure in Bard PowerPort Lawsuit
July 24, 2024: PowerPort Case Numbers Continue to Climb
July 14, 2024: Upcoming Case Management Conference Scheduled in Bard PowerPort Lawsuit
July 7, 2024: Upcoming Status Conference for Bard Implanted Port Catheter MDL
June 9, 2024: Parties File Joint Memorandum Highlighting Discovery Disputes
May 27, 2024: Intense Discovery Phase Underway in Bard PowerPort Lawsuit
May 15, 2024: Judge Addresses Plaintiff Profile Forms and Document Requests in Bard PowerPort Lawsuit
April 24, 2024: Judge Campbell Orders Remote Depositions in Bard PowerPort Lawsuit
April 9, 2024: Progress in Bard PowerPort Lawsuit – Key Agreements Reached
March 27, 2024: Key Study Highlights Risks in Bard PowerPort Lawsuit
March 21, 2024: Court Approves PowerPort Lawsuit Plaintiff Profile Forms
March 9, 2024: Slow Growth of PowerPort Lawsuit a Topic of Discussion
March 1, 2024: Bard PowerPort Lawsuit Surpasses 100 Cases
February 26, 2024: Common Benefit Expense Fund Accounting Structure
February 11, 2024: Litigation Continues for Bard PowerPort Lawsuit
January 20, 2024: Judges to Rule on Bard PowerPort Reservoir Defects
January 1, 2024: Status Conference Set for Bard PowerPort Port catheter lawsuit
December 3, 2023: Court Approves Short-Form Complaint in PowerPort Port Catheter “Class Action” Lawsuit
November 21, 2023: Bard Files Motion to Strike Allegations from PowerPort catheter Master Complaint
October 1, 2023: Court Holds Initial Conference in Bard PowerPort Lawsuit
September 14, 2023: Leadership Structure Proposed in Bard PowerPort Lawsuit
August 8, 2023: Federal MDL Created for Patients Alleging Harms from Bard PowerPort Catheters
May 24, 2023: Plaintiff Attorneys Seek Centralization of Bard PowerPort Lawsuits
Frequently Asked Questions (FAQs)

