Complete the form below to see if you qualify for a Depo Provera Lawsuit

Women across the United States are filing Depo-Provera brain tumor lawsuits after being diagnosed with intracranial meningiomas after using the injectable birth control. In their lawsuits, women allege that Depo-Provera’s active ingredient, medroxyprogesterone acetate (MPA), may increase a woman’s risk for developing hormone-sensitive brain tumors called meningiomas.
Although these tumors are not typically cancerous, they can have several negative impacts on a woman’s health. Further allegations in lawsuits include that the manufacturer of the drug, Pfizer, Inc., knew or should have known about the potential increased tumor risk and failed to properly warn consumers.
Cases have been consolidated into a multidistrict litigation (MDL) in Florida federal courts. This consolidation will help streamline the legal process for women. Women can file a lawsuit as part of MDL 3140 IN RE: Depo-Provera (Depot Medroxyprogesterone Acetate) Products Liability Litigation if they were diagnosed with brain tumors (meningiomas) after taking the birth control shot.
Due to the widespread use of the contraceptive, significantly more lawsuits are expected to be filed in the near future. If you have been diagnosed with a brain tumor after taking Depo-Provera, you may be eligible for compensation and can speak with an attorney about your legal options.
Depo-Provera Brain Tumor Lawsuit Overview
Depo-Provera is a contraceptive injection primarily used to prevent pregnancy. It is a widely used, long-term birth control method containing the hormone MPA (medroxyprogesterone acetate). Although it is highly effective at preventing pregnancy (it can be up to 99% effective), some research suggests it may increase the risk of a type of brain tumor called a meningioma. People who have been diagnosed with brain tumors after receiving Depo-Provera injections are filing lawsuits, alleging the drug contributed to their condition.
Legal documents filed against Pfizer and other manufacturers allege that the companies knew or should have known that patients were at an increased risk of developing meningiomas, particularly after prolonged Depo-Provera use. Symptoms of meningiomas include headaches, vision problems, seizures, and other neurological issues. Meningiomas have the potential to cause severe disability, permanent impairment, or life-threatening conditions.
What Is the Depo-Provera Lawsuit About?
Primary claims in Depo-Provera lawsuits include that Depo-Provera manufacturers allege they knew their drug increased the risk of brain tumors, yet they failed to properly warn doctors and patients about these risks. Additionally, Pfizer and other defendants are accused of overlooking or downplaying adverse event reports. Lawsuits say the companies failed to make timely updates to the Depo-Provera’s label and properly warn people about the risks of these conditions. (However, Pfizer argues the FDA prevented it from updating the warning label to include the risk of meningiomas.)
Lawsuits allege that manufacturers ignored growing evidence and prioritized profits over patient safety. Depo-Provera patients who develop brain tumors are encouraged to seek legal counsel for assistance with filing a claim.
Key Takeaways
Quick Facts About the Depo-Provera Litigation:
- MDL Number: 3140
- Federal Judge Overseeing the MDL: M. Casey Rodgers
- Court MDL Centralized in: Northern District of Florida
- Injury: Intracranial meningioma (brain tumor)
- Drug: Depo-Provera (medroxyprogesterone acetate)
- Main Defendant: Pfizer
- Cases filed: 5,508 (as of June 1, 2026)
- Scheduled Daubert Hearings: June 24 & 25, 2026
- First Scheduled Bellwether Trial: December 2026
Depo-Provera Lawsuit – 2026 Update
June 1, 2026: Depo-Provera Brain Tumor MDL Grows to Over 5,500 Claims
As of June 1, 2026, there are 5,508 lawsuits in the MDL that accuse Pfizer and several other companies of failing to warn women that Depo-Provera use may increase their chance of developing a brain tumor. This is a large uptick in claims month-over-month. There are 1,831 more lawsuits in the MDL as of June 1st than there were on May 1, 2026. This MDL continues to move at an efficient pace.
May 4, 2026: Woman Sues Pfizer, Claiming Depo-Provera Use Caused Tumor, Hearing Loss, and Vision Loss
A woman from Virginia has filed a lawsuit against Pfizer, the company that manufactures the injectable birth control Depo-Provera. According to her legal complaint, she received Depo-Provera injections from 2001 to about 2012. Several years after she started using Depo-Provera, her complaint alleges, she experienced symptoms such as vertigo, dizziness, and vision loss. Her lawsuit says her doctors discovered a brain tumor called an intracranial meningioma in 2003, but neither she nor her doctors were aware of a possible connection between the tumor and Depo-Provera until public reports announced a March 2024 study about it. She is suing Pfizer, alleging that the company should have warned her about the potential risks of developing an intracranial meningioma after using Depo-Provera.
May 1, 2026: Preemption Ruling Expected in Depo-Provera MDL as More Lawsuits Join Litigation
There are currently 3,769 active lawsuits in the centralized Depo-Provera litigation. Whether or not these lawsuits move forward in the MDL depends on the results of preemption hearing. Pfizer is arguing that the FDA prevented it from updating Depo-Provera’s label with a warning for meningioma risk, which was eventually added to the label. This argument is called preemption. If Judge Rodgers does not rule in Pfizer’s favor, the lawsuits in the MDL will be able to move forward. Currently, Daubert (causation) hearings are scheduled for June 24 and 25, 2026. At these hearings, the judge will hear arguments about whether or not Depo-Provera and meningiomas are scientifically linked.
April 7, 2026: Causation Hearings in Depo-Provera MDL Rescheduled to Begin June 24, 2026
The judge overseeing the Depo-Provera lawsuits has rescheduled the causation hearings to June 24 through June 26, 2026. The hearings had to be rescheduled because of a conflict with the judge’s schedule. On the new hearing dates, the parties will be discussing evidence to show that Depo-Provera use may lead to an increased risk of developing an intracranial meningioma. There are 3,490 lawsuits that have been consolidated in the Depo-Provera MDL, as of April 1, 2026.
March 20, 2026: Judge Reappoints Plaintiffs’ Attorneys to Leadership Positions in Depo-Provera MDL
Attorneys have been appointed to one-year leadership terms in 14 committees in the Depo-Provera multidistrict litigation. Pfizer and other brands have been accused of failing to warn patients that Depo-Provera use may be associated with an increased chance of developing an intracranial meningioma. Enough lawsuits were filed about this matter that these claims were consolidated into a multidistrict litigation in federal court, which is still accepting new cases. Creating committees and appointing attorneys to help manage the plaintiff side of the litigation may help to efficiently move the lawsuits through the legal process.
March 11, 2026: Depo-Provera Judge Issues Updates on Litigation
On March 11, Judge Rodgers issued Case Management Order (CMO) 11 for the Depo-Provera MDL. CMO 11 shared several important litigation updates. As of March 6, 2026, there are 3,790 unique plaintiffs in this litigation. She said she is reviewing Pfizer’s pre-emption motion, which says that the FDA pre-empted (prevented) Pfizer from adding a meningioma warning to the drug’s label. The order also stated that general causation expert depositions have been scheduled from February 28 through March 20, 2026. The next Depo-Provera MDL Case Management Conference will be held on Friday, April 17, 2026, at 9:00 a.m. (CDT). This litigation continues to move quickly. Women who developed a brain tumor (meningioma) after taking Depo-Provera can still speak with a lawyer about filing as part of this federal litigation.
March 2, 2026: More Than 1,000 New Depo-Provera Lawsuits Filed Between February and March
Between February and March 2026, the number of pending lawsuits in the Depo-Provera MDL increased by 1,001. As of March 2, there are 3,099 pending Depo-Provera lawsuits in the MDL. As of February 2, there were 2,098 pending lawsuits in the MDL. This is the largest month-over-month increase since the MDL was formed. Women alleging brain tumors from their Depo-Provera (medroxyprogesterone acetate) use can still talk to an attorney about pursuing a lawsuit as part of this litigation.
February 24, 2026: Woman Sues Pfizer, Claims Depo-Provera Use Led Her to Develop Brain Tumor, Lose Hearing
A woman is suing Pfizer and other companies that manufactured the Depo-Provera injections she received from about 1999 to 2017. She started to experience painful symptoms, including feeling fullness and hearing loss in her left ear. In 2017, imaging studies revealed that she had an intracranial meningioma, a type of brain tumor. Although these tumors are not usually cancerous, they can cause the symptoms that the woman experienced and greatly impair someone’s quality of life. In her lawsuit, the woman says that Depo-Provera’s manufacturers should have warned her about the potential risks of developing a brain tumor after taking the injectable birth control.
February 11, 2026: Women in U.K. Continue to Speak Out About Developing Brain Tumors After Using Depo-Provera
More women in the United Kingdom allege that receiving Depo-Provera injections has caused them to develop brain tumors called intracranial meningiomas. While intracranial meningiomas are rarely cancerous, treating them involves surgery, radiation, and other invasive methods. Moreover, these brain tumors can develop behind a woman’s eyes, which can lead to vision loss. In the United States, over 2,000 lawsuits have been filed against Pfizer, alleging that the company should have warned U.S.-based patients that using Depo-Provera may raise the risk of developing an intracranial meningioma. Such accounts from women mirror findings in clinical research of a higher rate of meningioma diagnosis among long-term users of medroxyprogesterone acetate.
February 24, 2026: Woman Sues Pfizer, Claims Depo-Provera Use Led Her to Develop Brain Tumor, Lose Hearing
A woman is suing Pfizer and other companies that manufactured the Depo-Provera injections she received from about 1999 to 2017. She started to experience painful symptoms, including feeling fullness and hearing loss in her left ear. In 2017, imaging studies revealed that she had an intracranial meningioma, a type of brain tumor. Although these tumors are not usually cancerous, they can cause the symptoms that the woman experienced and greatly impair someone’s quality of life. The woman alleges that Depo-Provera’s manufacturers should have warned her about the potential risks of developing a brain tumor after taking the injectable birth control.
February 2, 2026: More Than 2,000 Lawsuits Now Filed as Part of the Depo-Provera Litigation
As of February 2, 2026, there are now 2,098 lawsuits in the multidistrict litigation (MDL) for women who developed brain tumors after taking Depot-Provera, according to information released by the Joint Panel for Multidistrict Litigation. That is an increase of 346 lawsuits since January 5, 2026, when there were 1752 lawsuits in the MDL. This litigation is still moving forward as parties participate in the discovery process. The next case management conference will be held on Friday, February 20, 2026, at 9:00 a.m CT. Women who took Depo-Provera and developed brain tumors can still talk to an attorney about filing a lawsuit as part of this litigation.
January 20, 2026: Attorneys Submit Joint Proposed Agenda for Depo-Provera Litigation
A Case Management Conference in the Depo-Provera brain tumor MDL is scheduled for January 23, 2026. Attorneys for plaintiffs and attorneys for the defendants have submitted a proposed agenda for the upcoming conference. In that proposed agenda, they lay out trial milestones in the Depo-Provera litigation throughout 2026. For example, the first trial (pilot trial) scheduled in the Depo-Provera litigation is proposed for December 7, 2026. Judge Rodgers will have to approve the proposed agenda in order for it to move forward. Women who developed brain tumors after using Depo-Provera can still speak to an attorney about filing a case as part of this federal litigation.
January 16, 2026: Depo-Provera MDL Now Includes Over 1,700 Cases
As of January 5, 2026, there are 1,752 Depo-Provera complication claims that are part of the consolidated federal MDL. Women who are part of the Depo-Provera MDL were not told that this injectable birth control shot could raise their chances of developing a brain tumor, which, although commonly noncancerous, can result in vision loss and other adverse outcomes. These legal claims allege that Pfizer and other Depo-Provera manufacturers knew of the elevated risk but did not tell U.S. consumers about it.
January 2, 2026: Long Island Woman Develops Tumor After Taking Depo-Provera
A woman named Elizabeth Fleurisma took Depo-Provera birth control shots for about eight years, and then she started experiencing severe headaches and pain. A trip to the emergency room revealed that she had a tumor the size of a lime in her brain. The tumor was an intracranial meningioma, which is non-cancerous, but can cause significant problems if it grows and pushes onto nerves and other structures. Elizabeth had to have a lengthy surgery and undergo over 25 radiation treatments. She believes that her long-term use of Depo-Provera caused her to develop the intracranial meningioma, and she is suing Pfizer to seek compensation for her pain, suffering, and other losses. Her case reflects broader concerns raised in clinical research about prolonged exposure to medroxyprogesterone acetate and an increased tumor risk.
View Previous Depo-Provera Lawsuit Updates From 2024 & 2025
About the Depo-Provera Lawsuit:
New Research Links Depo-Provera to Brain Tumors in Women
Depo-Provera Manufacturer: Pfizer
Federal Lawsuit Filed Against Pfizer for Harms Caused by Depo-Provera
Eligibility Criteria in the Depo-Provera Lawsuit
Evidence Needed to File a Depo-Provera Lawsuit
Recoverable Damages in the Depo-Provera Lawsuit
How to File a Depo-Provera Lawsuit
Depo-Provera Lawsuit Settlement and Payout Amounts
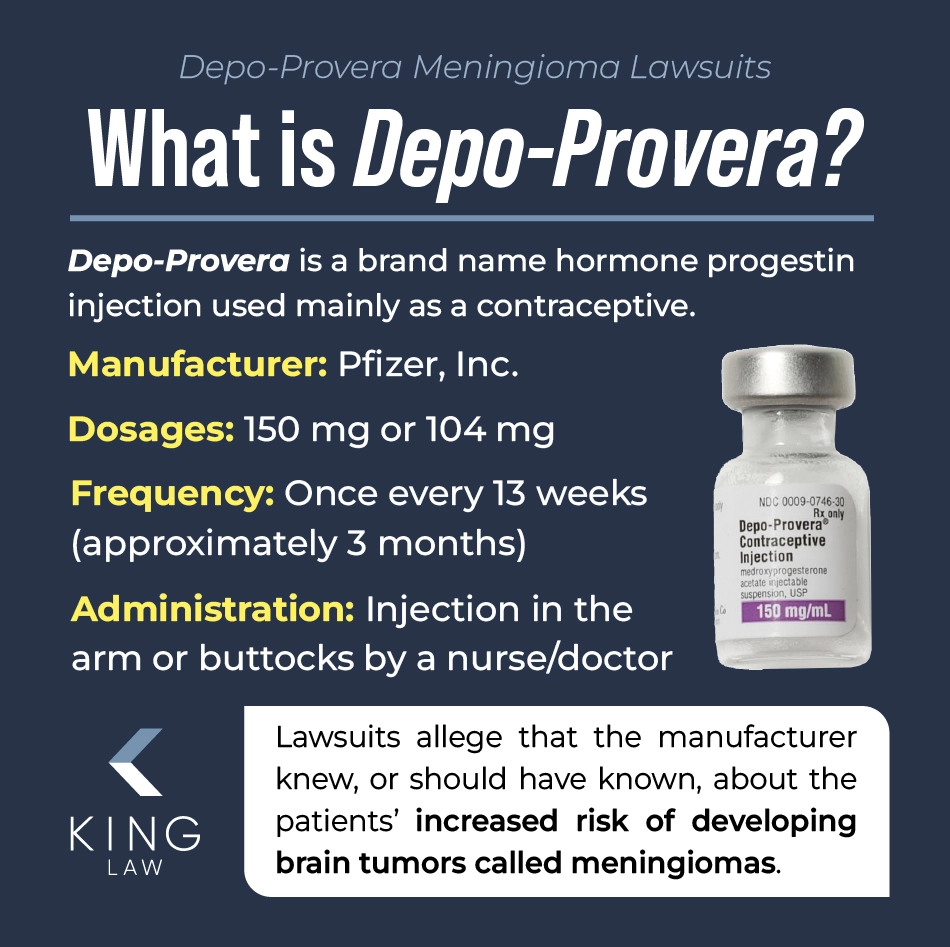
What Is Depo-Provera?
Depo-Provera is a brand name for medroxyprogesterone acetate, a synthetic form of the hormone progestin used primarily as a contraceptive injection that prevents pregnancy by inhibiting ovulation, thickening cervical mucus, and thinning the uterine lining. It is injected into the arm or buttocks and administered every 12 weeks.
The medication, also known as the birth control shot or Depo-Provera shot, has a high rate of effectiveness and may also be prescribed for managing conditions like endometriosis, heavy menstrual bleeding, and reducing the risk of endometrial cancer. It can prevent pregnancy for up to 14 weeks if administered correctly and on schedule.
How Does Depo-Provera Work?
Depo-Provera works through several mechanisms, including preventing ovulation, thickening cervical mucus, and thinning the uterine lining (i.e., the endometrium), ultimately creating an environment that is less than favorable for sperm survival and function.
How Depo-Provera Works:
- Inhibition of Ovulation: Depo-Provera prevents the ovaries from releasing an egg (ovulation), eliminating the possibility of fertilization.
- Thickening of Cervical Mucus: The injection thickens cervical mucus, making it difficult for sperm to pass through the cervix and reach an egg.
- Thinning of the Uterine Lining: Depo-Provera thins the uterine lining, reducing the likelihood that a fertilized egg could implant and develop.
- Effect on Sperm: The hormone in Depo-Provera creates an environment that is less favorable for sperm survival and function, further preventing pregnancy.
When administered correctly on schedule, Depo-Provera is a highly effective contraceptive method. It is injected into a muscle, generally the arm or buttocks, every 12 weeks (approximately every 3 months). Allegations against the manufacturer of Depo-Provera include that prolonged use of the medication may put patients at an increased risk for developing brain tumors and other severe conditions. Depo-Provera also has a range of side effects, which some women experience after taking the drug.

New Research Investigates Depo-Provera Use And Brain Tumors in Women
A 2024 study published in the British Medical Journal (Use of progestogens and the risk of intracranial meningioma: national case-control study) found that using drugs like Depo-Provera with the active ingredient medroxyprogesterone acetate for more than a year may increase the risk of intracranial meningioma by 5.6x. This finding was based on data from 18,061 women who underwent surgery for these typically benign brain tumors, which develop in the meninges. Meningiomas are generally benign (non-cancerous) tumors that develop in the meninges, the membranes covering the brain and spinal cord.
Researchers have suggested that the observed association may be related to the interaction between progestins and progesterone receptors found in some meningiomas.
Another study, published in 2025, also found an increased risk of brain tumors in women who took Depo-Provera injections. The study, which was published in JAMA Neurology, is called “Depot Medroxyprogesterone Acetate and Risk of Meningioma in the US.” Researchers looked at data for more than 10 million women in the U.S. They found that women who took injectable forms of DMPA (e.g., Depo-Provera) were almost 2.5 times more likely to develop brain tumors than women who took other forms of birth control or no birth control. The study also found that the risk of intracranial meningiomas was higher for women who took Depo-Provera for more than 4 years or who started the drug after age 31.
Key Studies on Depo-Provera and Meningioma Risk
| Study | Year | Journal | Key Finding |
|---|---|---|---|
| Roland et al. | 2024 | BMJ | Women with prolonged exposure to medroxyprogesterone acetate had approximately a 5.6-fold higher risk of intracranial meningioma compared with non-users. |
| Toh et al. | 2025 | JAMA Neurology | Use of injectable depot medroxyprogesterone acetate (DMPA) was associated with roughly a 2.5-times higher rate of meningioma diagnosis compared with women using other contraceptive methods. |
In some cases, discontinuing progestin-based therapy like Depo-Provera has been associated with stabilization or even regression of meningiomas, suggesting a potential reversibility of tumor growth. Although meningiomas are usually not life-threatening, surgery to remove them can be complicated and present risks.


The Relationship Between Meningiomas and Hormones
The active ingredient in Depo-Provera is medroxyprogesterone acetate (MPA). MPA is a synthetic progestin that interacts with progesterone receptors. Progestin is a hormone, and some tumors have hormone receptors. Hormones can affect how some tumors grow or shrink.
Many intracranial meningiomas contain progesterone receptors on their tumor cells. Studies have found that many meningiomas have these receptors. Because Depo-Provera contains a synthetic progestin, researchers have investigated whether exposure to progestins may influence the growth of hormone-sensitive meningiomas in some patients.
Is There a Relationship Between How Long Someone Takes Depo-Provera and Tumor Growth?
Some studies have examined how longer or repeated progestin exposure may be related to the frequency of meningioma diagnosis (for example, this 2024 study published in the BMJ). Researchers sometimes describe this as a “dose-response” pattern. This means that patients with greater cumulative exposure to certain progestins had higher observed rates of meningioma compared with those with shorter exposures. Said more plainly, some researchers have found that people who took medications containing progestins for longer periods of time had higher rates of meningiomas than those who took them for shorter periods of time.
Why Researchers Study Hormones and Meningiomas
Researchers have investigated how hormones may influence the development or growth of meningiomas. Epidemiological studies have reported that women develop meningiomas roughly two to three times more frequently than men. So, some scientists have investigated how hormones might account for that difference.
Scientists have also found that many meningiomas contain progesterone receptors. Tumor cells with these receptors can respond to hormonal signals. Because of these findings, researchers have investigated whether exposure to certain hormones or hormone-like medications may influence tumor growth in some patients.
Who Are Women Filing Depo-Provera Lawsuits Against?
Women who developed intracranial meningiomas after Depo-Provera use are filing lawsuits against the manufacturers of Depo-Provera and its authorized generics. Defendants named in Depo-Provera lawsuits include:
- Pfizer, Inc.
- Viatris Inc.
- Greenstone LLC
- Prasco, LLC d/b/a Prasco Labs
- Pharmacia & Upjohn Co, LLC
Although the primary defendant in Depo-Provera lawsuits is Pfizer, other defendants are listed. Several lawsuits argue that these other defendants:
- Were either owned or acquired by Pfizer at the times they were manufacturing Depo-Provera
- Were making a generic equivalent at a Pfizer-owned/operated facility
- Were producing a generic equivalent with the exact formulation of Pfizer’s brand-name Depo-Provera
Many of the existing lawsuits argue that Pfizer has been directly involved in manufacturing brand-name and generic equivalent Depo-Provera since 1993. Therefore, these lawsuits say Pfizer should be held liable for harming women.
Defendant and Depo-Provera Manufacturer: Pfizer
Pfizer, Inc., has been the manufacturer, marketer, and distributor of Depo-Provera since acquiring the rights through a merger with Pharmacia in 2003. The company is responsible for ensuring the drug’s global safety, efficacy, and quality standards. Under Pfizer’s ownership, Depo-Provera has become a widely used contraceptive with different formulations, including the standard Depo-Provera Contraceptive Injection and the lower-dose Depo-SubQ Provera 104.
However, the company has faced various lawsuits related to Depo-Provera, including claims concerning side effects like bone density loss and potential impacts to conditions such as meningiomas. The company must manage these legal issues while continuing to market and distribute Depo-Provera.
Federal Lawsuit Filed Against Pfizer for Harms Caused by Depo-Provera
On October 1, 2024, a California woman filed a lawsuit against Pfizer after taking Depo-Provera and developing a brain tumor. Kristina Schmidt used Depo-Provera for about 17 years and was diagnosed with a brain tumor at the age of 37.
Schmidt’s attorneys allege that her specific tumor, an intracranial meningioma, has been linked to progesterone-based contraceptives like Depo-Provera. Despite the alleged known link, Schmidt’s attorneys say Pfizer failed to adequately update Depo-Provera’s drug label to warn patients about the possible connection between the drug and intracranial meningiomas. The FDA approved an update to Depo-Provera’s warning label in December of 2025. Depo-Provera’s warning label now lists the risk of meningioma, especially with extended use of the drug.

Accusations Made Against Pfizer in the Schmidt Lawsuit
According to the complaint filed by Schmidt’s lawyers, Pfizer should have known that Depo-Provera increases the risk of brain tumors. However, they have not disclosed this information to patients or doctors. The complaint states, “The association between progesterone and meningioma has been known or knowable for decades, particularly for sophisticated pharmaceutical corporations like Defendants engaging in FDA-required post-market surveillance of their products for potential safety issues. That duty includes an obligation to keep current with emerging relevant literature and, where appropriate, perform their own long-term studies and follow-up research.”
Said more simply, Depo-Provera’s manufacturer (Pfizer) is required by the FDA to monitor the outcomes of people who use Depo-Provera. Schmidt’s lawyers accuse Pfizer of failing to perform the following actions:
- Monitoring for new side effects, especially ones that don’t happen immediately
- Conducting medical studies that examine the safety of Depo-Provera
- Closely following scientific literature that explored safety concerns about Depo-Provera
Because Pfizer did not take these actions or warn patients about the potential dangers of Depo-Provera, Schmidt’s lawyers are seeking damages to offset the pain, suffering, and losses she endured. Other plaintiffs will file lawsuits citing similar circumstances and seeking similar damages.
Eligibility Criteria in the Depo-Provera Lawsuit
In order to qualify for a Depo-Provera lawsuit, individuals must have used the brand-name (Pfizer) Depo-Provera, Depo-SubQ Provera, or an authorized generic version at least twice and must have been diagnosed with a meningioma or brain tumor after usage. Eligibility may be further influenced by the duration of use, time elapsed since last use, and specific diagnosis, potentially entitling them to file a lawsuit against the manufacturer for compensation.
Eligibility criteria for a Depo-Provera lawsuit:
- Usage: Must have used brand-name Depo-Provera (Pfizer), brand-name Depo-SubQ Provera (Pfizer), or an “authorized generic” version of Depo-Provera at least twice.
- Diagnosis: Diagnosed with meningioma or a brain tumor after using Depo-Provera.
- Other Considerations: The duration of Depo-Provera or its generic version usage, the time elapsed between the last use of Depo-Provera, the diagnosis, and the diagnosis of meningioma and brain tumors.
Due to the potential association between Depo-Provera and an increased risk of brain tumors in some women, individuals who have suffered a brain tumor after taking the medication may be able to file a lawsuit against the manufacturer.
Evidence Needed to File a Depo-Provera Lawsuit
It is vital to gather the necessary evidence in order to substantiate your claim. Evidence that may help support your case includes comprehensive medical records, prescription records, personal and witness testimony, financial records proving damages, and proof that the product used was the Pfizer-manufactured Depo-Provera or an authorized generic.
Evidence Necessary for a Depo-Provera Lawsuit:
- Documentation of Depo-Provera Use: Medical records, including prescription records and doctor’s notes, showing that the plaintiff received Depo-Provera, Depo-SubQ Provera, or an authorized generic at least twice.
- Diagnosis Records: Proof of diagnosis for a meningioma or brain tumor, with pathology reports, MRI or CT scan results, and neurologist or oncologist records showing the diagnosis occurred after starting Depo-Provera.
- Treatment Records: Documentation of treatments received for the tumor, such as surgery, radiation therapy, or medication.
- Pharmacy and Prescription Records: Detailed records showing the dispensing of Depo-Provera or its authorized generic, establishing the timeline and frequency of use.
- Plaintiff’s Testimony: A personal account of Depo-Provera use, the onset of symptoms, and the impact of the tumor on their life, including pain, suffering, and loss of income.
- Witness Testimony: Statements from family, friends, or coworkers corroborating the impact on the plaintiff’s life.
- Financial Records: Documentation of economic losses due to medical bills, lost wages, and other expenses related to the tumor.
- Impact on Quality of Life: Evidence showing how the diagnosis affected the plaintiff’s quality of life, including work incapacity, loss of enjoyment, or psychological trauma.
- Timeline of Events: A clear timeline of the start of Depo-Provera use, the tumor’s diagnosis, and the condition’s progression.
- Verification of Pfizer’s Involvement: Evidence that the Depo-Provera used was the brand-name product manufactured by Pfizer, or an authorized generic, is crucial for establishing the defendant in the lawsuit.
Specific Pieces of Evidence to Collect:
- Hospital bills related to treatment for the brain tumor or meningioma.
- Prescription records for Depo-Provera, Depo-SubQ Provera, or authorized generics.
- Doctor’s notes detailing the prescription and administration of Depo-Provera.
- Diagnosis of meningioma or brain tumor, including medical reports.
- Imaging results (MRIs, CT scans, etc.) confirming the presence of the brain tumor or meningioma.
- Pathology reports providing details on the type and characteristics of the tumor.
- Treatment records, including surgery, radiation therapy, or medication details.
- Pharmacy receipts for Depo-Provera or Depo-SubQ Provera purchases.
- Dispensing records showing the dates and dosages of Depo-Provera or its authorized generic.
- Prescription refill history from pharmacies or healthcare providers.
- Detailed medical bills and receipts for treatments, hospital stays, and medications.
- Pay stubs or employment records showing lost wages due to illness or treatment.
- Receipts for out-of-pocket expenses related to the brain tumor treatment or care.
- Written statements from the plaintiff detailing their experience with Depo-Provera and the onset of symptoms.
- Testimonies from family members, friends, or coworkers about the impact of the condition on the plaintiff’s life.
- Notes or records from therapy or counseling sessions related to the psychological effects of the diagnosis.
Recoverable Damages in the Depo-Provera Lawsuit
Depo-Provera patients diagnosed with a brain tumor may be entitled to recover a range of damages, including past and future medical expenses, compensation for lost wages and diminished earning capacity, damages for physical pain, emotional suffering, and loss of enjoyment of life.
Damages in a Depo-Provera Lawsuit:
- Past Medical Expenses: Costs for diagnosis, treatment, surgery, hospitalization, and medications up to the present.
- Future Medical Expenses: Projected costs for ongoing or future care, including surgeries, rehabilitation, long-term care, and medications.
- Past Lost Wages: Compensation for income lost due to an inability to work during treatment or recovery.
- Future Lost Earnings: Compensation for reduced earning potential due to the lasting effects of the condition.
- Loss of Earning Capacity: Damages for diminished future earning potential as a result of the condition.
- Physical Pain: Compensation for the physical pain endured due to the tumor and its treatment.
- Emotional and Mental Suffering: Compensation for emotional distress, anxiety, depression, and mental anguish caused by the condition.
- Diminished Quality of Life: Compensation for the loss of enjoyment in activities, hobbies, and daily living.
- Permanent Disability or Disfigurement: Damages for any permanent impairment resulting from the tumor or its treatment, as well as compensation for permanent physical disfigurement caused by surgery.
- Loss of Consortium: Compensation for the impact on the plaintiff’s relationship with their spouse, including loss of companionship and marital relations.
- Punitive Damages: Awarded in cases of particularly reckless or malicious conduct by the defendant, meant to punish and deter similar actions.
- Out-of-Pocket Costs: Reimbursement for additional costs directly related to the tumor, such as transportation, home care, or special equipment.
- Legal Fees and Court Costs: Potential recovery of legal representation costs (attorney’s fees) and reimbursement for filing fees, expert witness fees, and other lawsuit-related expenses.
Individuals who have lost a loved one as a result of Depo-Provera use may be entitled to a wrongful death claim. Recovery for a wrongful death lawsuit may include compensation for costs related to the plaintiff’s death, such as funeral and burial services, compensation for the financial support the deceased would have provided (loss of support), and damages for the loss of the deceased’s companionship and emotional support to family members (loss of companionship).
How to File a Depo-Provera Lawsuit
To file a Depo-Provera lawsuit, it is critical to follow specific steps and procedures, including confirming eligibility and gathering necessary evidence to prove your case. Consulting with an experienced attorney as early in the process as possible can help ensure the highest probability of success.
Steps to File a Depo-Provera Lawsuit:
- Confirm Eligibility: Ensure you have a confirmed diagnosis of meningioma.
- Consult a Highly-Qualified Lawyer: Seek out attorneys who are well-versed in pharmaceutical litigation or product liability, particularly those with experience in Depo-Provera or hormone-related cases. Many law firms offer free consultations.
- Gather and Prepare Necessary Documentation: Collect all relevant medical records that confirm your brain tumor diagnosis and evidence of your Depo-Provera usage.
- File the Lawsuit: Your attorney will draft and file a legal complaint against Pfizer, the manufacturer of Depo-Provera, in the appropriate court.
- Participate in Discovery Phase: During the Discovery Phase, both sides will exchange relevant information and evidence, including medical records, expert testimonies, and depositions.
- Settlement Negotiations: Before trial, there could be an attempt to settle the case out of court. Your lawyer will negotiate with Pfizer’s representatives to secure fair compensation.
- Trial: If a settlement cannot be reached, your case may proceed to trial, where your attorney will present your evidence to a judge or jury.
- Post-Trial: If the trial outcome is unfavorable, you may have the option to appeal the decision.
- Monitor Health and Follow-Up: Continue to monitor your health and receive ongoing medical care as needed for your condition.
Our page on how to file a Depo-Provera lawsuit contains additional information and nuance to help you navigate your potential claim.
Depo-Provera Lawsuit Settlement and Payout Amounts
Settlement amounts in Depo-Provera lawsuits can vary widely depending on factors such as the severity of the condition, medical expenses, lost wages, pain and suffering, and the potential for punitive damages. While payout amounts are speculative due to the early stages of litigation, typical settlements are expected to range from $100,000 to $5 million or more, depending on the specifics of each case.
Factors Influencing Depo-Provera Lawsuit Settlements:
- Severity of the Condition: The extent, size, location, and impact of the brain tumor or meningioma on the plaintiff’s health and daily life significantly influence settlement amounts. Cases involving severe, life-threatening tumors or those causing permanent disability tend to result in higher payouts.
- Medical Expenses: Costs for past and future medical treatments, including surgeries, hospital stays, medications, ongoing care, and rehabilitation, are critical factors. Higher medical expenses generally lead to larger settlements.
- Lost Wages and Earning Capacity: If the condition caused the plaintiff to miss work or reduced their ability to earn income, this financial loss will be considered in the settlement. Significant loss of income can result in larger compensation.
- Pain and Suffering: Non-economic damages such as physical pain, emotional distress, and loss of enjoyment of life are subjective but can substantially influence settlement amounts. Cases with significant pain and emotional trauma often see higher payouts.
- Legal and Expert Testimony: The strength of the evidence, including expert testimony linking Depo-Provera to the tumor, impacts the settlement. Strong, well-documented cases tend to achieve better outcomes.
- Punitive Damages: If the manufacturer (Pfizer) is found to have acted with gross negligence or willful misconduct, punitive damages may be awarded to punish and deter future misconduct, significantly increasing the payout.
- Settlement vs. Trial: Most cases are settled out of court, typically resulting in a guaranteed, though potentially lower, payout compared to what might be awarded by a jury at trial. A trial could result in a higher payout but also carries the risk of receiving nothing if the case is lost.
Settlements might range between $100,000 and $500,000 for smaller, treatable meningiomas with limited life impact. For large, debilitating tumors with extensive medical treatment and significant life impact, settlements or verdicts could range from $1 million to $5 million or more. Severe cases with solid evidence of negligence could also result in settlements or verdicts in the multi-million dollar range.
Settlement amounts vary widely and depend on each case’s specific details. Consulting with a leading attorney is essential to assessing the potential value of a Depo-Provera lawsuit and effectively navigating the legal process.
Depo-Provera Lawsuit Statute of Limitations and Deadlines
Statutes of limitations in personal injury lawsuits, including Depo-Provera lawsuits, vary by state and typically range from 1 to 6 years. The timeline potentially starts from the date of injury or the discovery that the injury may have been caused by Depo-Provera. Due to the significant variance in the statute of limitations for these kinds of cases, contacting an attorney is essential.
An attorney can help ensure timely filing and assist with collecting proper documentation. Affected individuals should start gathering necessary documentation early, as it can take time to collect medical records and proof of Depo-Provera use.
Depo-Provera lawsuits are generally based on personal injury or product liability claims. For personal injury claims, most states have a statute of limitations between 2 to 3 years from the date of injury or discovery. Product liability claims have similar time frames, with the clock starting when the injury or harm is discovered.
The statute of limitations may begin when the brain tumor or meningioma is diagnosed or when the possible link to Depo-Provera is discovered, extending the filing period if the injury wasn’t immediately apparent. It may be paused if the plaintiff is a minor or legally incapacitated, starting once they turn 18 or regain capacity.
Additionally, different time frames apply for wrongful death claims, usually starting from the date of death. An attorney with extensive experience in pharmaceutical litigation can determine the specific statute of limitations for your case and any applicable exceptions.
Contact a Depo-Provera Lawyer
Depo-Provera patients who are diagnosed with a brain tumor or meningioma are encouraged to contact King Law to schedule a free consultation. The attorneys at King Law are well-versed in handling pharmaceutical litigation and will work strategically to secure the compensation you deserve. Our legal team is committed to advocating for those affected by Depo-Provera, ensuring that each case is handled with expertise and care.
Frequently Asked Questions (FAQs)
Previous Depo-Provera Lawsuit Updates
December 17, 2025: Pfizer Updates Depo-Provera’s Label to Include Risk of Brain Tumors
On December 12, 2025, the FDA notified Pfizer that it had approved updates to Depo-Provera’s warning label to include an increased risk of meningiomas. The risk of brain tumors is now present on the labels for two forms of the Depo-Provera: Depo-Provera CI and Depo-Subq Provera 104. Depo-Provera’s label now says, “Cases of meningiomas have been reported following repeated administration of medroxyprogesterone acetate, primarily with long term use,” as well as other warning language about brain tumors. More than 1,000 women who used Depo-Provera and developed meningiomas have filed lawsuits against Pfizer. These lawsuits allege Pfizer has known that long-term exposure to the active drug in Depo-Provera could cause brain tumors for decades. However, they waited until 2024 to seek updates to the U.S. prescribing label.
December 15, 2025: Almost 250 More Women Who Took Depo-Provera Have Joined Brain Tumor Lawsuits
Between November and December, 245 more women have joined the MDL against Pfizer for its defective Depo-Provera birth control shot. There are now 1,470 active lawsuits filed by women and their families after women received Depo-Provera injections and developed a brain tumor. While the type of tumor that Dep-Provera can increase the risk of is not usually cancerous, it can lead to vision loss, severe migraines, seizures, and other complications. Pfizer issued warnings about this risk to physicians and consumers in Europe and Canada. However, women and doctors in the United States were not warned about this same risk.
December 2, 2025: More Advocates, Doctors Warn About Brain Tumor Risk from Depo-Provera Shot
Advocates from the University of California San Francisco continue to warn women about the brain tumor risks when taking Depo-Provera. Depo-Provera puts medroxyprogesterone acetate (a type of artificial progesterone) in the woman’s system. Women in Canada and overseas were told that Depo-Provera could increase the risk of a brain tumor. However, women in the United States were not, and they are filing lawsuits against drug companies.
November 24, 2025: Plaintiffs In Depo-Provera Lawsuits Await Hearing Results
During November of 2025, the judge overseeing the group Depo-Provera lawsuit heard pre-emption arguments from Pfizer. Most pharmaceutical companies try to have cases against them dismissed, citing preemption. This is a legal concept that says the federal regulations (usually set by the FDA) kept the company from making changes to its drug’s warning label. Pfizer, which makes Depo-Provera, is arguing that the FDA kept them from adding a warning for brain tumors to Depo-Provera’s drug information label. Now that the November hearing is complete, women who have filed brain tumor lawsuits are waiting to see if their cases will move forward in federal court.
November 14, 2025: Number of Depo-Provera Brain Tumor Claims Surpasses 1,200
Hundreds of women who received Depo-Provera injections developed brain tumors (intracranial meningiomas) that affected their quality of life. They say they did not know this was a potential side effect, and they are filing lawsuits in state and federal courts. Currently, there are 1,225 Depo-Provera lawsuits pending in a federal group claim, and more are being added each month. Women are seeking to hold Pfizer and other pharmaceutical companies accountable for not creating a safer product or warning women about the risks.
November 3, 2025: Federal and State Courts Handling Depo-Provera Lawsuits Meet
At an October 27, 2025, case management conference, the judge overseeing the federal Depo-Provera MDL met with judges from the New York and Delaware state courts. More than a thousand women have filed lawsuits at the federal and state levels after they developed intracranial meningiomas from the Depo-Provera shot. The judges and attorneys held the meeting to discuss ways to coordinate and clarify case matters to help the Depo-Provera lawsuits move forward efficiently. Meanwhile, women who developed brain tumors after Depo-Provera use can still join this litigation.
October 29, 2025: Woman Files Depo-Provera Lawsuit After Brain Tumor Changes Her Life
A 45-year-old woman in Louisiana has joined the group lawsuit for women who developed brain tumors after Depo-Provera use. Robin Phillip used the birth control shot for years and eventually developed a meningioma, a type of brain tumor possibly associated with prolonged Depo-Provera use. Phillip had surgery, lost her left eye, and had to relearn to walk because of her brain tumor. Phillip’s lawsuit alleges that Pfizer has known about the meningioma risk for decades and had a duty to warn women. Phillip says that she never would have taken Depo-Provera if she had been told it raised her risk of developing a brain tumor.
October 22, 2025: Depo-Provera Lawsuits Remain Active in State and Federal Courts
According to a recent update from attorneys in the group Depo-Provera litigation, cases are moving forward at the state and federal levels. There are currently 1,661 women who have filed lawsuits against Pfizer. Each of these women says they developed brain tumors (meningiomas) from the birth control shot. As of October 20, 2025, there are 1,222 cases in the federal multidistrict litigation (MDL). There are also hundreds of lawsuits filed at the state level, including 332 plaintiffs in Delaware, 78 in New York, 20 in California, 6 in Illinois, 1 in Pennsylvania, 1 in Connecticut, and 1 in New Mexico. General causation expert disclosures and depositions will occur in late 2025 and early 2026. During these hearings, lawyers on both sides will present and discredit scientific evidence and testimony linking Depo-Provera use to brain tumors.
October 15, 2025: Lawsuits in Depo-Provera Multidistrict Litigation Make Progress
The Depo-Provera brain tumor MDL continues to move forward as the parties (lawyers) submit a status report on the litigation to Judge Rodgers. According to this document, plaintiffs are working with counsel to turn in the required paperwork for their claims. Additionally, the parties indicated there are about 105 individual Depo-Provera claims pending in state courts. Some of these may be transferred into the federal MDL in the future, while others have already been moved from the federal MDL into state courts. More plaintiffs can join the Depo-Provera group claim if they developed an intracranial meningioma or another severe complication after taking this injectable birth control shot.
October 1, 2025: Louisiana Woman Sues Pfizer After Developing Tumor Because of Depo-Provera Use
Robin Phillip took Depo-Provera birth control shots for many years, just like 24.5% of sexually active American women. She received the shots for decades until she developed a brain tumor, an intracranial meningioma, that caused her to have surgery, lose vision in her left eye, and experience neurological challenges. She discovered that the Depo-Provera injections were the reason for her condition, and she is suing Pfizer for not warning her about the risks. Many women have had similar experiences and are speaking out against Pfizer for its negligence.
September 27, 2025: Requirements to Join Depo-Provera Lawsuit Make It Easier for More Women to File
The updated filing requirements for the Depo-Provera MDL make it easier to file lawsuits against Pfizer and other companies. Many women develop non-cancerous/benign brain tumors years or decades after taking Depo-Provera. Because of this, some women are having trouble tracking down medical records, which are required to file a lawsuit. Now, according to the court’s new order, eligible women who do not have these records can still maintain their lawsuits (if they meet specific requirements). This is a big win for women who were harmed by Depo-Provera but whose previous providers did not maintain historical records or are no longer in business.
September 25, 2025: Attorneys Prepare For Important Hearing in Depo-Provera Lawsuits
More than 800 lawsuits have been filed by women alleging Depo-Provera caused them to develop brain tumors. Leading attorneys representing women in these lawsuits are preparing for an important milestone in the case–the preemption hearing. This hearing will be held next week, and it will decide whether or not the federal group litigation moves forward. Pfizer is making a preemption claim, saying that the FDA prevented them from updating Depo-Provera’s drug warning label to include the risk of brain tumors. However, Pfizer only brought this information to the FDA in 2024, and many argue that Pfizer withheld information and downplayed the risk for years. Pre-emption is a common tactic taken by defense attorneys, though it is not typically successful. If Pfizer does convince the courts that the FDA is at fault, the lawsuits filed by women will be sent back to state courts. Women and their attorneys will be fighting for a positive outcome from this hearing.
September 15, 2025: Depo-Provera Legal Cases Nearly Double Between July 1st and September 1st
As of September 1st, there are 806 lawsuits in the federal MDL involving Depo-Provera, a marked increase from the 435 claims on the MDL’s roster in July. Depo-Provera is an injectable birth control shot used to help women prevent unwanted pregnancies, manage problematic periods, and address other health conditions. However, patients believe Pfizer and the other companies that manufactured Depo-Provera were untruthful about the risks of this injection. Patients who took Depo-Provera shots developed a type of brain tumor, an intracranial meningioma, and are filing lawsuits to seek compensation.
September 10, 2025: As Pfizer Fights Liability, Another Study Examines Depo-Provera and Tumors
A September 2025 study published in JAMA Neurology provides more evidence that Depo-Provera and brain tumors are connected. The study found that women who used injectable birth controls with depot medroxyprogesterone acetate had a “…statistically significant increased relative risk of developing a meningioma diagnosis,” than women who took other forms of birth control or no birth control. In fact, women taking Depo-Provera were almost 2.5 times more likely to develop a type of brain tumor called an intracranial meningioma. The study looked at data from more than 10 million women. It is published at a time when Depo-Provera’s manufacturer, Pfizer, is trying to sidestep liability by claiming the FDA would not let the company add this risk to the drug’s warning label. However, how transparent Pfizer was about the true risk remains to be seen.
September 1, 2025: Pfizer Tries to Hide Behind FDA Labeling Laws in Depo-Provera Lawsuit
Pfizer’s attorneys are attempting to avoid liability by blaming the FDA’s laws about medication labels. The document Pfizer submitted to the court alleges that Pfizer asked to update the label to include more risks of taking Depo-Provera, but the FDA denied this request. Drug companies often resort to this type of argument, but it is rarely successful. The plaintiffs’ attorneys will file a response in the coming weeks. In the meantime, more women who developed brain tumors after taking Depo-Provera are encouraged to join the lawsuit.
August 15, 2025: Pfizer Preserves Resources As It Handles Pressure from Depo-Provera Lawsuits
Pfizer is being sued by hundreds of Depo-Provera patients because the pharmaceutical giant failed to warn its US-based customers of the heightened risks of brain tumors. Victims’ legal teams continue to fight back against Pfizer’s efforts to get the claims thrown out. Meanwhile, Pfizer takes steps to conserve its resources by cutting its partnership with clinical drug company Acepodia. The move is thought to be temporary while Pfizer battles legal challenges to Depo-Provera.
August 1, 2025: New Study Says Depo-Provera Substantially Increases Brain Tumor Risk
As women continue to file lawsuits against Depo-Provera manufacturers, new research is emerging that highlights the risk of taking the birth control shot. A study called “Use of medroxyprogesterone acetate and risk of meningiomas: a comparative safety study,” found that women who took Depo-Provera for more than 1 year had a 3.5 times greater risk of developing a meningioma (brain tumor) than women who took combination birth control pills. These findings will support women who are seeking compensation after not being warned about this dangerous side effect.
July 17, 2025: Almost 100 New Depo-Provera Lawsuits Filed
Between June and July, almost 100 new plaintiffs joined the federal group lawsuit for women who developed brain tumors after using Depo-Provera. There are now 435 active lawsuits against Pfizer and other manufacturers, up from 348 filings in June. This surge in filings comes at a time when Pfizer hopes that its pre-emption argument stops this litigation. Pfizer argues the FDA rejected their request to add the risk of brain tumors to Depo-Provera’s warning label, which is a legal theory called preemption. Women can still file lawsuits as part of this group.
July 2, 2025: New Deadlines Set in Group Depo-Provera Lawsuit
The multidistrict litigation for Depo-Provera represents hundreds of women who have developed brain tumors after taking the injectable birth control. The judge overseeing this group litigation, Judge M. Casey Rodgers, has set a series of deadlines to ensure the deadline for preemption is met. As in the case with most litigations against drug companies, Pfizer is claiming protection under preemption. They are essentially arguing that the FDA prevented them from updating their label to include a warning about brain tumors. Although this is a frequent argument, it is rarely successful. Judge Rodgers has issued schedule updates to make sure pre-emption arguments are closed by the October deadline.
June 17, 2025: Parties Submit Status Update in Depo-Provera MDL Ahead of Conference
The plaintiffs and defendant corporations in the Depo-Provera lawsuit filed a status update to track important case matters before the upcoming meeting. Most of these updates concern the discovery process, where the injured consumers and pharmaceutical companies are seeking information and responding to record requests. Pfizer says it has shared over 10 million pages, with more to come. The plaintiffs will continue gathering proof to show they were injured by Depo-Provera. Plaintiffs’ lawyers will review the documents submitted by Pfizer and the other defendants. They will be looking for clues that these companies knew that Depo-Provera could cause brain tumors but failed to disclose this information to women.
June 2, 2025: Judge In Depo-Provera Lawsuit Creates Process to Clean Up Plaintiff’s Complaints
a process to find errors or omissions in lawsuits filed by women harmed by Depo-Provera. Clarifications included in this process include a list of specific types of brand-name and generic Depo-Provera that a woman must have received and the specific types and subtypes of brain tumors she must have been diagnosed with. The goal of creating a process to correct plaintiffs’ lawsuits is to keep this litigation moving as efficiently as possible, which has been a goal of Judge Rodgers since the beginning of the Depo-Provera litigation.
May 20, 2025: Judge Clarifies Claim Requirements for Filing a Depo-Provera Lawsuit
Judge M. Casey Rodgers has provided important clarifications for women who are filing lawsuits related to Depo-Provera-induced brain tumors. In Pre-Trial Order 22, Judge Rodgers has clarified that each woman who is part of the group lawsuit is required to complete a Plaintiff Proof of Use/Injury Questionnaire. Additionally, each woman needs to provide “documentary proof” that meets the court’s injury threshold. Generally, if a woman used Depo-Provera and developed an intracranial meningioma, she is eligible for the group lawsuit. However, proper and timely submission of documentation is crucial to filing a viable lawsuit. Women who have suffered such injuries should reach out to a qualified attorney to give their case the best chance of securing a settlement.
May 13, 2025: Pfizer Hands Over 1 Million Documents in Depo-Provera Litigation
The three defendants listed in Depo-Provera lawsuits (Pfizer Inc., Pharmacia & Upjohn Co. LLC, and Pharmacia LLC) said they have turned over 1 million documents to the courts and plaintiffs’ lawyers. This means that lawyers representing women who suffered brain tumors from Depo-Provera now have about 8 million pages of documentation from the defendants. Pfizer said these documents should be more than sufficient to litigate preemption and general causation issues, two of the most important issues in the case. Many defendants try to flood plaintiff lawyers with paperwork and try to omit crucial information from the delivery. Women’s lawyers will be hard at work reviewing this documentation and looking for key omissions. However, the science is strong in the Depo-Provera cases, and the defendant’s efforts are unlikely to help them escape liability.
May 1, 2025: Generic Depo-Provera Manufacturers Must Participate in Pre-trial Discovery
In the Depo-Provera lawsuit, Judge Rodgers issued an order clarifying expectations for generic drug manufacturers in the group lawsuit. Due to a stipulation between plaintiffs’ attorneys and Pfizer, it appears Prasco may not be part of the lawsuit. However, Greenstone LLC, and Viatris Inc., are expected to remain as plaintiffs and participate fully in pre-trial information sharing and other actions. This is good news for the many women who took generic forms of Depo-Provera made by Greenstone and Viatris. Plaintiffs’ lawyers will be working hard to determine what these companies knew and when, regarding how Depo-Provera could increase the risk of brain tumors.
April 16, 2025: Co-Defendants in Depo-Provera Lawsuit Miss Key Deadline
Judge Rodgers, who is overseeing the group lawsuit for women harmed by Depo-Provera, ordered that co-defendants named in these lawsuits submit information on time. After missing the initial deadline, co-defendants Greenstone and Viatris submitted the court-ordered affidavits. Judge Rodgers reiterated the importance of strict compliance with deadlines. By missing this deadline, Greenstone and Viatris, who manufactured generic versions of Depo-Provera, may have eroded some of their credibility in the case.
April 1, 2025: Judge Facilitates Record Collection For Women Who File Depo-Provera Lawsuits
Women who have joined the group lawsuit against the makers of Depo-Provera will have to submit documentation from medical providers, insurance companies, and pharmacies to make their case. Getting this information just became easier, due to an order from Judge Rodgers, who is overseeing this litigation. The order removes barriers to record retrieval for many women. Some women took Depo-Provera decades ago, so tracking down records has been difficult. Judge Rodgers looked to simplify the record-obtaining process for women and their attorneys. She also requested information from Pfizer about distribution, which should bolster some women’s cases.
March 18, 2025: Jude in Depo-Provera Cases Says Women Can Direct File in Florida
In a move that could be intended to speed up the lawsuit and settlement processes, women who developed brain tumors after their Depo-Provera use can file their lawsuits directly in Federal courts. In many multidistrict litigations (MDLs), people alleging harm file in the state court system first, and then their cases are transferred to federal courts. But, in the Depo-Provera MDL, Judge M. Casey Rodgers has ordered that women can file directly in Florida Federal courts without losing any of the benefits of filing in state courts. This move is another example of Judge Rodgers trying to be as efficient as possible throughout this litigation.
March 3, 2025: Judge in Depo-Provera Lawsuits Appoints Special Master and Data Administrator
Following a March 3, 2025, case management conference, the judge overseeing the group Depo-Provera lawsuits has issued two important orders. Judge M. Casey Rodgers appointed the Honorable David Herndon as a Special Master to assist with managing the Depo-Provera multidistrict litigation. Judge Rodgers shared she will oversee five Pilot cases, efficiently and effectively. Judge Rodgers also appointed a data administrator to oversee the large volume of documents that will be included in this case. These two orders likely indicate that Judge Rodgers is interested in moving this litigation forward as quickly as possible. This is welcome news for the many women who developed brain tumors after taking Depo-Provera.
February 26, 2025: Attorneys Discuss Next Steps In Depo-Provera Lawsuits
Now that a group lawsuit has been formed for women harmed by Depo-Provera, legal professionals are discussing what happens next in these lawsuits. There will be important milestones throughout 2025 for this litigation. Attorneys and judges will work to establish a timeline, attorneys on both sides will exchange important documents, lead attorneys will be assigned to the case, and arguments will be heard about whether or not women who took generic versions of Depo-Provera will have viable lawsuits. To learn more about how Depo-Provera lawsuits are progressing, watch this video.
February 21, 2025: Depo-Provera Lawsuits in the MDL Are Moving Forward
The multidistrict litigation (MDL) against Depo-Provera and Pfizer is moving forward. In the two weeks since the group lawsuit was created, there have already been orders and meetings relevant to women harmed by Depo-Provera. For example, on February 19, 2025, the Honorable James V. Ingold issued a conditional order moving 27 civil lawsuits filed at the state level to Florida federal courts under U.S. District Judge M. Casey Rodgers. On February 21, 2025, Judge Rodgers held a case management conference to discuss multiple issues relevant to this group lawsuit. Judge Rodgers is an experienced mass tort judge. She oversaw the 3M earplug product liability litigation. Many legal professionals believe this group lawsuit will move forward at a fast pace, meaning women harmed by Depo-Provera will be able to negotiate for settlements and awards sooner, possibly within the next 1 to 2 years.
February 7, 2025: Depo-Provera MDL Approved; Women With Brain Tumors Will Be Able to Join Mass Lawsuit
On February 7, 2025, a multidistrict litigation (MDL) was formed for women who took Depo-Provera and developed brain tumors (meningiomas). The consolidation is called: MDL 3140 IN RE: Depo-Provera (Depot Medroxyprogesterone Acetate) Products Liability Litigation. This MDL will simplify the lawsuit-filing process for the thousands of women harmed by the birth control shot. The Judicial Panel on Multidistrict Litigation (JPML) ruled that lawsuit consolidation will occur in the Northern District of Florida. The judge presiding over the case will likely be the Honorable M. Casey Rodgers. This consolidation gives women harmed by Depo-Provera a way to file a lawsuit as part of a group, while maintaining individual compensation for their injuries.
February 3, 2025: Depo-Provera Hearing Confirms an MDL, But No Ruling on Where Lawsuits Will be Combined
The Judicial Panel on Multidistrict Litigation (JPML) held a hearing on January 30 for the pending Depo-Provera multidistrict litigation (MDL). Plaintiffs for women who developed brain tumors after taking Depo-Provera battled with Pfizer’s lawyers about which federal court these lawsuits will be consolidated in—California or New York. Plaintiffs’ lawyers will keep pushing for California so more women who developed brain tumors can pursue lawsuits against Pfizer. Many in the legal community expect an order from the JPML judges by February 7, 2025. That order should reveal more details about this MDL.
January 28, 2025: Women Who Filed Depo-Provera Lawsuits Waiting for MDL Consolidation Decision
Many women who experienced brain tumors after taking Depo-Provera are waiting to hear how their lawsuits will play out. A hearing will be held in the first week of February to confirm that a multidistrict litigation (MDL) will be formed. Many women and their attorneys hope the lawsuits will be consolidated in California federal courts, as opposed to the defendant’s suggestion of New York. If the lawsuits are consolidated in California, women who took generic versions of Depo-Provera will have better lawsuit filing options. Women harmed by Depo-Provera will know more about their lawsuit filing options in early February.
January 16, 2025: Depo-Provera Lawsuits Will Likely Proceed as MDL and Not a Class Action Lawsuit
Many Lawsuits filed by women who experienced brain tumors after using Depo-Provera will likely become part of a multidistrict litigation (MDL), rather than a class action lawsuit. An MDL means many lawsuits will be consolidated under one federal judge, but each woman will get to keep her individual claim. In a class action lawsuit, women would not be able to keep individual claims. Assuming the lawsuits are consolidated into an MDL, women could complete short-form complaints and work toward receiving settlements for their specific injuries and hardships.
January 15, 2025: Pfizer Argues That FDA Would Not Update Depo-Provera’s Warning Label To Include Risk of Brain Tumors
In Pfizer’s response to the petition for a Depo-Provera multidistrict litigation, the company claims it wanted to update Depo-Provera’s drug label to include the risk of meningiomas, which are a type of brain tumor. However, Pfizer says the FDA said there was not enough evidence to make the update, so the label was not updated. Pfizer says the FDA overruled Pfizer (a legal concept called a preemption defense). Preemption defenses are common tactics in pharmaceutical product liability cases. However, these tactics are rarely successful.
January 10, 2025: Next Step In Depo-Provera Lawsuits–An MDL Hearing at the End of January
Women who filed Depo-Provera lawsuits after being diagnosed with brain tumors are waiting to see how the lawsuits will be handled by the courts. On January 30, 2025, the U.S. Judicial Panel on Multidistrict Litigation (JPML) will hold a hearing that will determine if a multidistrict lawsuit (MDL) will be created and which state the federal lawsuits will be consolidated in. Plaintiffs’ attorneys want the lawsuits to be overseen by California federal courts. Defendants want these cases to play out in New York federal courts. Because Pfizer has agreed an MDL should be created, it is likely these group lawsuits will move forward. This MDL will simplify the legal process for thousands of women whose brain tumors may be associated with the Depo-Provera birth control shot and who deserve compensation for their injuries.
January 3, 2025: Pfizer Agrees Depo-Provera Lawsuits Should Be Consolidated Into Multidistrict Litigation
On December 30, 2024, Pfizer responded to a petition that asked Depo-Provera lawsuits to be consolidated in federal court. The original request was filed by attorneys who represented 22 women who developed brain tumors after using Depo-Provera. Pfizer and the other defendants have responded saying they agree a multidistrict litigation (MDL) should be formed. MDLs are created when many people are harmed by the same product and sustain similar injuries. However, in their response, Pfizer asked for the lawsuits to be consolidated in New York, as opposed to California. Plaintiffs’ attorneys will continue to fight for consolidation in California. The Judicial Panel on Multidistrict Litigation will decide where these cases will be heard. Many legal professionals believe the MDL could be created in February 2025. This would mean women who developed brain tumors after using Depo-Provera would be one step closer to justice and possible settlements.
January 2, 2025: Depo-Provera Lawsuits May Consolidate and Move Toward Settlements in 2025
At the end of 2024, attorneys representing 22 women who developed brain tumors after Depo-Provera use took steps to consolidate their lawsuits in a federal court. Attorneys for these women asked the Judicial Panel on Multidistrict Litigation (JPML) to create a multidistrict litigation (MDL) for women who developed brain tumors after taking the birth control shot. If the JPML approves this motion, women who say they were harmed by Depo-Provera could file cases as part of a group, similar to a class action lawsuit. Women would have the option to join the Depo-Provera MDL and work on receiving a settlement.
December 23, 2024: Black and Hispanic Women May Be Disproportionately Affected by Meningiomas
According to data from the Centers for Disease Control and Prevention (CDC), 42% of Black women and 27% of Hispanic women have used Depo-Provera at some point in their lives. This means Black and Hispanic women may experience more cases of brain tumors alleged to be associated with Depo-Provera use than their peers. In contrast, 20% of white women and 7% of Asian women have used the three-month injectable. This data highlights disparities in the types of birth control used by different populations of women and how some groups may be adversely impacted by Depo-Provera.
December 18, 2024: California Woman Files Depo-Provera Lawsuit Years After Tumor Diagnosis
A woman in Bakersfield, California, has filed a Depo-Provera lawsuit more than 10 years after being diagnosed with her first brain tumor. Faith Lowery used Depo-Provera from 2001 to 2014, receiving approximately 50 birth control injections. In 2014, Lowery started experiencing severe headaches, nausea and vomiting, muscle weakness, vision changes, and other ailments. She was diagnosed with a meningioma and had to have brain surgery. She was diagnosed with another meningioma in 2022. Although Lowery’s injuries occurred years ago, she was able to file a lawsuit because she recently discovered that her tumors might have been caused by Depo-Provera. With new information available on how Depo-Provera may cause brain tumors, women who had tumors years ago can still file lawsuits.
December 11, 2024: Women Who Took Generic Depo-Provera Would Benefit From Lawsuit Consolidation in California
Many of the Depo-Provera lawsuits filed in state courts could become part of a multidistrict litigation (MDL). At the end of November, several attorneys filed a motion to transfer 22 Depo-Provera lawsuits into an MDL. With good reason, they requested the lawsuits be consolidated in California federal courts. California has strong innovator liability laws that would allow women injured by Depo-Provera to sue Pfizer, even if those women took generic depot medroxyprogesterone acetate. This is important because Depo-Provera has been on the U.S. market for more than 30 years, meaning many women took generic versions of the drug. Consolidation in California would mean more women could receive compensation for the Depo-Provera-related brain tumors.
December 3, 2024: Request To Consolidate Depo-Provera Lawsuits Would Give Women Future Options for Filing Lawsuits
Attorneys representing women who developed brain tumors after Depo-Provera use filed for lawsuit consolidation in federal court. If this happens and a Depo-Provera multidistrict litigation is created, it would give many women another way to file lawsuits. In their Motion to Transfer, the plaintiffs’ attorneys also stated the motion was, “also intended to encompass any future cases filed involving usage of Depo-Provera and the development of cerebral meningioma.” Many women who used Depo-Provera have not developed brain tumors yet, but they could develop tumors in the future. If this motion to consolidate is approved, the MDL would give future claimants (women harmed by Depo-Provera) an option to file as part of this litigation.
December 2, 2024: Attorneys Name Six Defendants In Request to Create a Depo-Provera MDL
Attorneys representing 22 women have filed federal lawsuits against 6 manufacturers of Depo-Provera and its generic equivalents (medroxyprogesterone acetate). In their motion to transfer existing lawsuits into a multidistrict litigation (MDL), attorneys named six defendants—Pfizer; Pharmacia & Upjohn Co. LLC; Greenstone LLC; Viatris Inc.; Pharmacia LLC; and Prasco LLC. If the 22 existing lawsuits are consolidated into an MDL, each of these defendants could be held liable for manufacturing and promoting a contraceptive drug possibly associated with an increased risk of tumors in women.
November 27, 2024: Depo-Provera Cases Move Toward Potential MDL Formation
Plaintiffs’ lawyers have requested that the Judicial Panel on Multidistrict Litigation consolidate 22 filed Depo-Provera lawsuits. Consolidation would create a Depo-Provera MDL. The plaintiffs’ lawyers have proposed that the cases be consolidated in the Northern District of California.
The defense is expected to request that the cases be transferred to New York, which has laws more favorable to pharmaceutical companies. New York is also the corporate headquarters of Pfizer, the largest defendant and the current manufacturer of Depo-Provera.
The panel, chaired by Karen Caldwell, will hear the application in early 2025. We anticipate several filings between now and the date of the hearing.
November 26, 2024: What Are the Legal Grounds in the Depo-Provera Lawsuit?
The legal basis of the Depo-Provera lawsuits claims that Pfizer, the manufacturer, failed to disclose serious side effects on the drug’s warning label and knew that the injectable contraceptive was linked to brain tumors in women. If you have been diagnosed with a brain tumor or meningioma, you may qualify for compensation.
Robert King Law Firm has reviewed over 7,000 Depo-Provera brain tumor claims and offers free case reviews to individuals who have experienced severe side effects.
November 22, 2024: New Depo-Provera Lawsuits Names Healthcare Agency, Insurance Company, and Pharmacy As Defendants
A lawsuit filed on November 15, 2024, has named medical groups and pharmacies among its defendants. Madison Le filed a lawsuit in the Superior Court of California in Alameda County. In her lawsuit, she names Depo-Provera manufacturers, but she also names Kaiser Permanente International, Kaiser Foundation Health Plan Inc., and the Permanente Medical Group Inc., which are entities such as insurance companies, medical groups, and pharmacies. In her complaint, Le’s attorneys say these businesses also knew about the dangers of Depo-Provera by conducting their own independent verifications and research. Yet they continued to offer and profit from its administration.
November 21, 2024: Some Brain Tumors Associated with Depo-Provera Use Are Inoperable
Many women who develop intracranial meningiomas after Depo-Provera use have operations to remove the tumors. However, some women are not candidates for resection (removal). There are several reasons a meningioma may be classified as inoperable, including the tumor being located in a sensitive area of the brain or the tumor infiltrating brain tissue. Additionally, some tumors are near or touching critical structures such as the venous sinuses, arteries, or cranial nerves. Women who cannot have their meningiomas removed may need to consider other therapies, such as radiation therapy, hormone therapy, or treatments available through clinical trials. This can prolong their symptoms and suffering and greatly impact their lives.
November 20, 2024: Las Vegas Woman Diagnosed With 3 Brain Tumors After 20 Years of Depo-Provera Use
A woman in Nevada has filed a lawsuit against Pfizer alleging that her Depo-Provera use led to her developing three intracranial meningiomas. Tina Stephens-Smith took Depo-Provera’s birth-control injections for 20 years and recently developed symptoms such as headaches, blurred vision, and speech problems. After seeking medical care, she learned she had three, densely calcified meningiomas—two behind her ear and one at the base of her skull. Stephens-Smith and her attorneys say that Pfizer knew about this dangerous side effect for years and yet did not warn U.S. women.
November 19, 2024: New Study Recommends Comprehensive Preoperative Imaging for Meningioma Patients
A review article published in the Journal of Neuroimaging highlights the importance of pre-surgical imaging for people suffering from intracranial meningiomas. The article, titled “Intracranial meningioma: A review of recent and emerging data on the utility of preoperative imaging for management,” examined current and emerging imaging technologies and how they can be used to help patients diagnosed with meningiomas. The authors stated, “A strong foundation of knowledge in the latest meningioma imaging techniques will allow the neuroradiologist to help optimize preoperative planning and improve patient outcomes.” This underscores the importance of targeted, preoperative imaging for women who used Depo-Provera and developed a brain tumor that can be treated surgically.
November 18, 2024: Study Finds 53% Increased Risk of Developing Meningiomas After Depo-Provera Use
A broad-scale study published in the Swedish medical journal, Cancers, 53% increased odds of being diagnosed with a meningioma. The incidence of meningiomas in U.S. women is approximately11 cases per 100,000 women. That means the incidence rate of meningiomas in women who used Depo-Provera would be close to 16 women per 100,000.
November 15, 2024: Progesterone Study Strengthens Causation Claims in Depo-Provera Lawsuits
David Bailey’s study from January 2024 is critical to the Depo-Provera lawsuit. The study indicates that stopping progesterone drugs like Depo-Provera and Depo-SubQ Provera can lead to regression of meningioma size.
In order to be successful in court, lawyers will need to show that Depo-Provera can cause an increased risk of meningioma (general causation) and that Depo-Provera caused the meningioma in the plaintiff bringing the case (specific causation). Bailey’s study is critical because the shrinking effect observed after stopping progesterone supports both general and specific causation for women who have used Depo-Provera and developed a meningioma.
November 14, 2024: Concerns Continue to Grow Over Meningioma Risk Linked to Depo-Provera
Our law firm continues to receive numerous inquiries from individuals worried about the potential risk of developing brain tumors, particularly meningiomas, after using Depo-Provera. Meningiomas are a type of tumor that forms in the meninges, the protective membranes around the brain and spinal cord. These tumors grow slowly but can cause severe health issues due to increased pressure on brain tissue, nerves, and blood vessels. Meningioma symptoms often include headaches, vision problems, and neurological impairments, and can sometimes even lead to permanent disabilities.
November 12, 2024: Pfizer Includes Meningioma Warning for Patients Using Depo-Provera in Canada But Not for U.S. Women
Women in Canada who used Depo-Provera and other DMPA drugs have been warned about the association between DMPA and meningiomas for almost a decade. Since at least 2016, Pfizer has warned women that long-term Depo-Provera use may increase the risk of developing intracranial meningiomas. In theproduct monograph for Depo-Provera, which includes information for patients, Pfizer includes the following warning for patients in Canada, “Meningiomas have been reported following long-term administration of progestins, including medroxyprogesterone acetate (MPA).” The Canadian drug information pamphlet also advises physicians to use caution when prescribing Depo-Provera to patients with a history of meningiomas. The pamphlet also says physicians should immediately stop prescribing Depo-Provera if a patient develops a meningioma. Currently, Pfizer does not include any warnings or guidance about Depo-Provera and meningiomas in its prescribing information for patients in the United States. Many U.S. women are just learning about the association between Depo-Provera and brain tumors and are now filing lawsuits against Pfizer for not warning them about this danger.
November 9, 2024: New Depo-Provera Lawsuit Filed in California Alleging Link to Meningioma
Mayra Valencia is the plaintiff in a recent Depo-Provera lawsuit filed on November 4, 2024, in the Eastern District of California. The complaint alleges that the defendants knew or should have known that progesterone, analogue progestin, and Depo-Provera increase the risk of a meningioma. It further states that the Canadian version of the drug has included a meningioma warning since at least 2015. Ms. Valencia took Depo-Provera for twenty-three years and experienced blurred vision and headaches for years until an MRI revealed a brain tumor. In August 2024, at age 39, she underwent an orbitozygomatic craniotomy, during which her skull was drilled, and the meningioma was removed. The complaint also highlights studies dating back to 1983 showing the correlation between progesterone use and meningioma.
November 7, 2024: Depo-Provera Settlements and Awards Could Have High Dollar Values
Depo-Provera settlements could be larger than similar mass torts. The quality of the proof appears to be strong at this point. First, the study that we are looking at shows a 5.5x increase in the risk of the injury (brain tumors). 5.5x is one of the highest increases in risk we have seen for a sizeable lawsuit. Second, the type of injury is dramatic—a brain tumor. Next, the manufacturer is well funded. They will be able to pay a settlement of virtually any amount. Finally, the quantity of cases should not limit the settlement amounts. In mass torts where there are copious plaintiffs, the amount each person gets through a settlement is usually lower. This is because most defendants have some limit to what they can pay without going bankrupt. In this scenario, the use of Depo-Provera could lead to about 55 cases of meningioma per 100,000 users, per year. There is some question about the number of people taking Depo-Provera. Recent data from the CDC showed 2% of women aged 15 to 49 took Depo-Provera from 2017 to 2019. However, Depo-Provera use was likely higher in the 1990s and 2000s. According to a National Health Statistics Report from the CDC, by 1995, 4.5% of U.S. women aged 15 to 44 had used Depo-Provera, and 23% had used it from 2006 to 2010. Some estimates are several million users per year in the U.S. That means there would be about 1,300 meningioma cases per year. The drug has been used for about 32 years. That means that 5,000 to 30,000 women could potentially file lawsuits. Not everyone who qualifies for a Depo-Provera lawsuit will choose to file. The relatively low number of lawsuits could lead to higher settlement values. We anticipate average settlement values of more than $100,000, with some cases substantially higher than that.
November 1, 2024: Depo-Provera Lawsuits Focus on Meningiomas, Potential Breast Cancer Link
Depo-Provera has been closely scrutinized throughout its history. The drug has been approved and removed from use several times. This 1988 study highlights some of the criticisms in place shortly before the drug was approved as birth control in the United States. Notably, two different animal studies suggested an increased cancer risk for users of Depo-Provera. The lawsuits have largely focused on meningiomas, but there may also be an increased risk of breast cancer for long-term users of Depo.
November 1, 2024: Another Woman Battling Brain Tumors Files a Depo-Provera Lawsuit Against Pfizer and Other Defendants
A woman in California has filed a Depo-Provera lawsuit after being diagnosed with an intracranial meningioma. Anjanna Lawson began receiving Depo-Provera injections at the age of 16 and continued to receive them through September 2024. In July of 2023, Lawson was diagnosed with an intracranial meningioma and underwent a right pterional craniotomy to remove the tumor. However, in early 2024, a residual tumor was discovered, and Lawson had five sessions of stereotactic radiosurgery. Her tumor has still not been eradicated, and she endures vision loss, facial spasms, and a burning sensation in her face. In the complaint filed as part of her Depo-Provera lawsuit, she is seeking damages from Pfizer, Inc., Viatris, Inc., Greenstone, LLC, Prasco, LLC, Pharmacia & Upjohn LLC, and Pharmacia, LLC.
October 29, 2024: New Depo-Provera Lawsuit Filed in California with Potential for Multidistrict Consolidation
Another Depo-Provera lawsuit has been filed. Kathleen Fazio v. Pfizer, Viatris, Inc., Greenstone, LLC, Prasco Labs, Pharmacia, and Upjohn was filed on October 28, 2024, in the Central District of California. The five initial court filings in federal court suggest that plaintiff lawyers will soon request the Judicial Panel on Multidistrict Litigation to consolidate the Depo-Provera lawsuits. Consolidation means that the discovery phase for each Depo-Provera case would be handled by a group of lawyers appointed by a single federal court judge.
It is noteworthy that no lawsuits have been filed in a New York federal court. Pfizer is based in New York and would be a logical place for Depo-Provera lawsuits to be filed. However, plaintiff lawyers have avoided filing in New York due to prior unfavorable rulings in federal cases. The plaintiff lawyers are carefully selecting jurisdictions where lawsuits will be filed. We anticipate early cases will be filed in California, Massachusetts, and potentially Vermont and Illinois. These four states implicitly or explicitly accept a legal theory known as innovator liability, which is expected to be significant in claims brought against Pfizer by users of generic Depo-Provera.
October 28, 2024: Californian Woman Files Depo-Provera Lawsuit As Cases Against Pfizer Increase
A Central California woman has filed another Depo-Provera lawsuit in the Central District of California. The lawsuit will be referred to as Monique Jones v. Pfizer, et. al. We are beginning to see a trend of where cases are being filed, with two cases filed in the Northern District of California and one in the Southern District of Indiana. We suspect this trend to continue because Indiana and California have strong state laws on innovator liability. Innovator liability is a legal concept where the innovator (the original manufacturer) can be held liable for alleged injuries inflicted by generic drugs. Generic drugs must mirror the exact language of the original warning label. So, if a person takes a generic drug, the originator (innovator) of the drug can be liable because they intentionally or negligently withheld information about the safety of the drug. This concept is included in the lawsuit filed by Jones and her legal team.
October 25, 2024: Second Depo-Provera Lawsuit Filed Against Pfizer by Indiana Woman
On October 15, a woman in Indiana filed a lawsuit against Pfizer, claiming her brain tumor and subsequent injuries were caused by Depo-Provera use. Plaintiff Lesley Noble received Depo-Provera injections over the course of 20 years. Noble’s intracranial meningioma was discovered in 2017. She underwent invasive surgery to remove her brain tumor, but after 6 months, her tumor aggressively regrew. She underwent 36 rounds of radiation during her second treatment, all while she was still taking Depo-Provera, not knowing it likely caused her tumors. Lesley Noble and her husband Justin Noble are demanding a jury trial for the many injuries Lesley has endured.
October 22, 2024: Pfizer and Co-Defendants Face 9 Accusations In First Depo-Provera Lawsuit
Pfizer, Inc. and its co-defendants, Viatris, Inc., Greenstone LLC, Prasco Labs, and Pharmacia & Upjohn, face nine allegations of wrongdoing in a federal Depo-Provera lawsuit. Kristina Schmidt filed a lawsuit in California Federal Courts that claims her brain tumor was caused by years of Depo-Provera use. In that lawsuit, Schmidt’s lawyers list nine allegations against the defendants, including Failure to Warn, Design Defect, Negligence, Negligent Failure to Warn, Negligent Design Defect, Negligent Design Defect, Negligent Misrepresentation, Fraudulent Misrepresentation, Breach of Express Warranty, and Breach of Implied Warranty. The charges accuse Pfizer and the other defendants of knowingly designing, marketing, and manufacturing a drug that could cause intracranial meningiomas without properly warning physicians or patients.
October 17, 2024: Evidence Suggests Exposure to Hormones in Depo-Provera May Increase the Risk of Spinal Tumors
We are investigating Depo-Provera cases that involve meningiomas of the spinal cord. Recent studies have examined Depo-Provera use and brain tumors, specifically intracranial meningiomas. New research is focusing on meningiomas that form on the spinal cord. An article published in the journal of Neuro-Oncology Advances adds that while less than 13% of meningiomas are on the spinal cord, meningiomas make up about a quarter of the tumors on the spinal cord. According to the article, external hormone sources, such as injections like Depo-Provera, may be associated with a higher risk of developing meningiomas in the brain or spinal cord.
October 11, 2024: Pfizer’s Legal Battle Over Depo-Provera and Generic Drug Liability
One of the central issues in the Depo-Provera lawsuit is whether Pfizer is legally responsible for meningiomas and brain tumors filed by women who used “generic” versions of Depo-Provera. In Mutual Pharmaceutical Co. v. Bartlett, the United States Supreme Court ruled that manufacturers of generic drugs are not legally responsible if the drug they make has the same active ingredient as an approved drug. However, in the Depo-Provera case, the “generic” manufacturers are mostly wholly owned subsidiaries of Pfizer.
Additionally, the “generic” drugs mentioned in the Depo-Provera lawsuits are exact copies of Depo-Provera and were manufactured at the same sites as Depo-Provera, in locations owned by Pfizer. For example, the complaint in the recently filed case of Schmidt v. Pfizer also names Greenstone LLC as a defendant. Greenstone LLC was owned by Pfizer but now operates under the Viatris umbrella.
We expect questions about whether Pfizer is legally responsible for meningiomas caused by “authorized generics” to be aggressively litigated for several years as part of this lawsuit. Pfizer has owned the rights to Depo-Provera since 2002, when they purchased Upjohn.
October 4, 2024: Woman Files Lawsuit Against Pfizer Alleging Depo-Provera Caused Her Brain Tumor
A woman has filed a lawsuit after taking Depo-Provera and developing an intracranial meningioma, a type of brain tumor. Kristina Schmidt filed her case against Pfizer in the United States District Court of the Northern District of California. Her attorneys allege Depo-Provera substantially contributed to the development of her brain tumor 17 years after taking the contraceptive. Since her diagnosis, she has endured significant, invasive treatments and experienced serious injuries. In the complaint filed in California Federal Court, her lawyers say that Pfizer, “knew or should have known for decades that Depo-Provera, when administered and prescribed as intended, can cause or substantially contribute to the development of meningiomas.” This case is part of a growing number of lawsuits against Pfizer for its injectable contraceptive.
October 4, 2024: Depo-Provera Tied to Highest Cancer Rate Increase Among Commercial Drugs
Lawyers are highly interested in pursuing the Depo-Provera lawsuit because early studies seem to indicate the drug makes the diagnosed injury over five times more likely to occur. This is one of the highest increases in cancer rates among commercially available drugs. In comparison to other cancer-related lawsuits, lawyers have argued that Roundup causes a 69% increase in the likelihood of cancer. There have been several verdicts over $1 billion in the Roundup lawsuit, and we believe the proof is even stronger in the Depo-Provera case.
September 30, 2024: Depo-Provera Lawsuit Expands as More Plaintiffs Learn of Potential Risks
The Depo-Provera lawsuit is gaining traction as more possible plaintiffs become aware of the allegations against Pfizer, the birth control manufacturer. A French study showed a potential link between the Depo-Provera shot and meningiomas, a tumor that arises from the protective membranes covering the brain and spinal cord. Symptoms can include headaches, vision changes, seizures, memory loss, and dizziness. Further research must be conducted to understand why Depo-Provera may increase the risk of tumor growth. With 74 million women worldwide having used the drug, the potential scale of this litigation is significant.
September 25, 2024: Depo-Provera May Increase Risk of Brain Tumors, Lawsuits Expected to Rise
Depo-Provera has been around for a long time—a really long time. Originally developed in 1959, the drug was approved by the FDA in 1974, only to have its approval withdrawn in 1978 due to concerns it could cause cancer. It was reapproved by the FDA as a contraceptive in 1992. With indications it could cause cancer more than 45 years ago, it was no surprise when the British Medical Journal discovered a connection between Depo-Provera and the development of meningiomas and other brain tumors. More studies are forthcoming, and with the sheer volume of women who have taken this drug over the past five decades, a large number of lawsuits are expected in the future.
September 17, 2024: Pfizer Faces Renewed Legal Challenges Over Depo-Provera and Brain Tumor Risk
Pfizer is again facing legal pressure over its drug Depo-Provera. Recent news on Depo-Provera followed a study showing that a particular type of brain tumor, called meningiomas, is more than five times more likely in women who took the drug as few as two times. There were previous lawsuits alleging that Depo-Provera led to bone density loss. One notable case was Cassandra Colville v. Pharmacia & Upjohn, filed on July 10, 2008. Since then, the drug and the rights to manufacture and sell Depo-Provera have been sold to pharmaceutical giant Pfizer.
September 13, 2024: Study Highlights the High Value of Meningioma Lawsuits Related to Depo-Provera
A 2020 study analyzed 47 cases of meningioma lawsuits. The study has gained attention recently as lawyers investigate meningiomas in women who took Depo-Provera. The study found that failure to diagnose was the basis of the lawsuit in 68.1% of the cases. The average verdict in lawsuits involving meningiomas was $3,409,650.22, and settlements averaged $867,555.56. The takeaway is that meningioma cases hold high value. If it is shown that Depo-Provera caused an increased likelihood of meningiomas, we expect settlement values of at least several hundred thousand dollars. Pfizer, the manufacturer of Depo-Provera, has the financial resources to pay large settlements.
September 10, 2024: Anticipated Federal Court Consolidation for Depo-Provera Claims Involving Brain Tumors
According to the CDC, approximately 24.5% of sexually active women have used the injectable contraceptive Depo-Provera at some point in their lifetime. Due to the drug’s widespread use, several women diagnosed with brain tumors are now filing claims against its manufacturers. While there are no significant developments in the Depo-Provera lawsuit at this time, our firm anticipates that these cases will ultimately be consolidated in federal court, which will initiate legal action against the drugmakers.
September 5, 2024: Depo-Provera’s History of FDA Rejections, Bone Density Concerns, and Possible Increased Risk of Brain Tumors
Depo-Provera was repeatedly denied approval by the FDA in the 1970s and 1980s, until it was finally approved in 1992. Since then, reports have suggested that the drug leads to reduced bone density and can cause significant side effects when women attempt to stop using it. Originally manufactured by Upjohn, Depo-Provera is now produced by Pfizer. Recent studies examined the drug and brain tumors.
August 2024: Depo-Provera cases continue to be filed nationwide. Affected individuals are encouraged to contact King Law to schedule a free case review.
March 2024: A study published in the British Medical Journal (BMJ) finds that people using an injectable form of medroxyprogesterone acetate for a year or more may have a higher risk of meningioma.
May 2008: The Superior Court of Quebec certified an action against Pfizer regarding Depo-Provera on behalf of a national class.
November 2004: The U.S. Food and Drug Administration (FDA) required a “black box” warning on the Depo-Provera label. The warning notes that prolonged use of the drug may cause irreversible and significant loss of bone density.
List of Articles and Sources Used in This Article
BMJ. Use of Progestogens and the Risk of Intracranial Meningioma: National Case-Control Study. 2023.
Cleveland Clinic, Depo-Provera Birth Control Shot.
Daniels, K. Ph.D., and Abma, J Ph.D. Use of Contraception in the United States. National Health Statistics Reports. Centers for Disease Control. December 14, 2023
Leigh, S. Could Birth Control Shot Increase Your Risk of Brain Tumor?. University of California San Francisco. October 2025.
NBC News. FDA Approves Label Change for Depo-Provera Adding Brain Tumor Warning. December 2025.
NBC News. Depo-Provera Birth Control Blamed for Woman’s Brain Tumor, Lawsuit Alleges. October 2025.
NBC Nightly News. Lawsuit Alleges Pfizer Should Have Warned Women About Tumor Risk from Birth Control.
Mayo Clinic. Meningioma – Symptoms and Causes. September 2025.
Maiuri, F. et al. Progesterone Receptor Expression in Meningiomas: Pathological and Prognostic Implications. Frontiers in Oncology. July 2021.
Pfizer. Pfizer Inc..
Pfizer. Depo-Provera CI (medroxyprogesterone acetate injectable suspension).
Pfizer Labeling. Depo-Provera Patient Information.
U.S. Judicial Panel on Multidistrict Litigation. Products Liability Litigation – MDL No. 3140 Transfer Order.
U.S. Judicial Panel on Multidistrict Litigation,
Monthly Report of Pending MDL Dockets By Actions Pending – December 2, 2025.
U.S. Judicial Panel on Multidistrict Litigation. Monthly Report of Pending MDL Dockets By Actions Pending – February 2, 2026.
U.S. Judicial Panel on Multidistrict Litigation. Monthly Report of Pending MDL Dockets By Actions Pending – January 5, 2026.
U.S. Judicial Panel on Multidistrict Litigation. Monthly Report of Pending MDL Dockets By Actions Pending – November 3, 2025.
U.S. Judicial Panel on Multidistrict Litigation. Monthly Report of Pending MDL Dockets By Actions Pending – September 2, 2025.
U.S. Food and Drug Administration, 2004. Depo-Provera Contraceptive Injection Label.
U.S. Food and Drug Administration, 2003. Depo-Provera Label.
Xiao, T. et al. Depot Medroxyprogesterone Acetate and Risk of Meningioma in the US. JAMA Neurology 2025.

 Depo-Provera Brain Tumor Lawsuit: A Comprehensive Guide
Depo-Provera Brain Tumor Lawsuit: A Comprehensive Guide