Complete the form below to see if you qualify for a Olympus Scope Lawsuit

Olympus makes endoscopes used in a variety of medical procedures, including ERCPs, colonoscopies and endoscopies. Some of these devices have been associated with post-operative infections and device malfunctions, which have seriously harmed patients.
Olympus now faces numerous lawsuits that accuse the company of selling defective products that led to serious infections and injuries. Plaintiffs say Olympus’s reusable devices (which include models like the TJF-Q180V, TJF-Q190V, and TJF-Q290V duodenoscopes, MAJ-891-forceps/irrigation plug, and the BF-1TH190 bronchoscope) left them vulnerable to life-threatening “superbug” infections. Several of these models, along with single-use Olympus devices like the Vizishot 2 FLEX needle, have been subject to FDA recalls.
Below, this page will cover the Olympus scopes infection lawsuits, how such infections occur, recalls and warnings associated with the devices, and how injured patients can seek compensation.
About the Olympus Scopes Lawsuit:
Olympus Scope Alerts, Recalls, and Lawsuit Updates
What Is the Olympus Scopes Lawsuit About?
What Procedures Are Olympus’ Scopes Used For?
Which Olympus Devices Are Involved in Lawsuits, Safety Alerts, and Recalls?
How Can Olympus Scopes Transmit Infection?
What Infections Have Been Linked to Olympus Scopes?
Can Olympus Scopes Cause Mechanical Injury or Organ Perforation?
What Did the 2018 Department of Justice Case Against Olympus Establish?
What Is the 2025 FDA Import Alert on Olympus Devices?
Who Can File an Olympus Scopes Lawsuit?
What Evidence Is Needed to File an Olympus Scopes Lawsuit?
How Can I File an Olympus Scopes Lawsuit?
How Long Does an Olympus Scopes Lawsuit Take?
Is There a Deadline to File an Olympus Scopes Lawsuit?
How Much Is an Olympus Scopes Lawsuit Worth?
Bottom Line – What Patients and Hospitals Should Know
King Law Is Investigating Olympus Scopes Infection Claims
Olympus Scope Alerts, Recalls, and Lawsuit Updates
Below is a summary of actions taken by Olympus and the FDA regarding device malfunctions and corrections for Olympus scopes and accessories, as well as associated litigations. As of June 2026, cases remain individual product liability actions filed in state and federal courts.
April 24, 2026: King Law Releases Video Explaining Olympus Scopes Lawsuits and Infection Risks
King Law releases new video featuring Attorney Robert King, Brianna Benedict, and litigation partner Brett Vaughn discussing the Olympus scope lawsuits. In the video, Brett Vaughn explains how reusable Olympus endoscopes used in procedures like colonoscopies, endoscopies, and ERCPs may expose patients to dangerous bacteria, potentially leading to severe infections and sepsis within days of the procedure.
The discussion highlights key issues driving the litigation, including contamination risks, challenges with proper sterilization, and ongoing FDA scrutiny of Olympus devices. You can watch the full video on YouTube.
April 15, 2026: People Continue to File Olympus Scope Lawsuits
Some people who had endoscopic procedures with Olympus Scopes developed severe infections after their procedures. As people realize their infection may have been caused by bacteria on the reusable scopes, some people are consulting attorneys to understand if they have a viable lawsuit. Our team can help people understand if a surgical infection is possibly related to a reusable scope and if they have grounds to file a lawsuit for their losses. Some patients pursuing personal injury claims against Olympus, related to infections and sepsis after a scoping procedure.
October 14, 2025: Olympus Issues Urgent Field Safety Notice Regarding Duodenoscope
Olympus releases an urgent field notice regarding ongoing infection risks with TJF-Q190V, which the company says have been linked to two infection-related deaths and five serious injuries. The notice updates cleaning and preprocessing procedures. Included is a visual inspection guide that recommends using 10x magnification tools to examine the duodenoscope for contamination.
September 30, 2025: FDA Issues Endoscopic Aspiration Needle Recall
The FDA
August 6, 2025: Olympus Sends Warning Letter About Aspiration Needle
Citing documented injuries, Olympus sends a warning letter to customers (physicians) who have purchased products with an endoscopic aspiration needle made by Olympus. The letter tells customers, “Do not use any ViziShot 2 Flex (19G) devices with a lot number listed in Attachment 1.”
June 24, 2025: The FDA Issues Import Alerts on Multiple Olympus Devices
Citing ongoing quality system regulation violations, the FDA issued an import alert on certain Olympus medical devices manufactured at specific facilities in Japan. The import alerts prevent future shipments of some Olympus devices into the U.S., including certain models of:
- Ureteroscopes
- Bronchoscopes
- Laparoscopes
- Automated endoscope reprocessors
The FDA also urges providers who are currently using the products to follow labeling and reprocessing procedures to properly clean Olympus devices and associated accessories.
May 29, 2025: Olympus Announces Release of Specialized Drying Cabinet to Aid Scope Disinfection
Olympus’ scopes and accessories have been the subject of multiple recalls and alleged infection outbreaks. Now, the company has released the ScopeLocker Air endoscope drying cabinet, as a way to more thoroughly dry its reusable scopes. The scopes contain channels and, according to Olympus, completely drying out those channels is critical to scope disinfection. Olympus says the cabinet will help better suit medical facilities’ “reprocessing needs” for scope cleaning. Olympus has been the subject of lawsuits filed by patients who say they contracted infections from the company’s reusable scopes and accessories.
December 18, 2024: Another Reusable Olympus Device Faces Recall Over Infection Risks
The FDA posts a Class 1 recall, its most serious classification, of the Olympus MAJ-891 Forceps/Irrigation Plug, a reusable accessory for the company’s endoscopes, due to the risk of transferring biomaterial between uses, especially when not properly disassembled. The device is linked to 120 infection-related injuries as well as one death. The FDA’s recall alert notes the improper reprocessing of the MAJ-891 can “result in infection, urinary tract infection, or sepsis and, in some cases, could result in death.”
December 8, 2023: Olympus Issues Recall of Olympus Single-Use Distal Cover on Endoscopes
Olympus issued a Class 2 Device Recall of its Single Use Distal Cover MAJ-2315, which is used with the EVIS EXERA III Duodenovideoscope (model #: TJF-Q190V). The company recalls the cover because it can “…unexpectedly detach from the duodenoscope during use.” This increases the risk of mucosal injury, tissue damage, bleeding, perforation, or obstruction. The FDA issued its official recall on January 16, 2025.
June 8, 2023: FDA Issues a Class 1 Recall for Some Olympus Scopes
Olympus issued a recall for certain bronchofiberscopes and bronchovideoscopes, with an FDA Class 1 recall following on July 17, 2023. The recall is for “laser-compatible bronchoscopes after reports of burns occurring inside the airways and lungs due to endobronchial combustion (airway fire).” These burns can result in hospitalization, ICU care, and death.
January 18, 2021: Olympus Issues Class 2 Device Recall of EVIS EXERA Duodenovideoscope Model
Olympus issued a Class 2 Device Recall for the EVIS EXERA Duodenovideoscope (TJF Type 190V). After initiating a recall, Olympus issued notices to customers who purchased the TJF-Q190V. Olympus says the recall is issued because of reports that the scope collects tissues and breaks down during procedures. According to Olympus, it had received complaints from customers (physicians) that device malfunction resulted in mucosal injuries to the esophagus, stomach, and duodenum.
January 31, 2020: Olympus’s Next-Gen Duodenoscope Also Carries Risks
Olympus released a new duodenoscope model, the TJF-Q190V, in January 2020, which introduces disposable endcaps meant to allow easier cleaning access to the problematic elevator channel.
December 10, 2018: Olympus, Regulatory Official Plead Guilty to Distributing Misbranded Medical Devices
According to a
January 8, 2016: Class 2 Device Recall Issued for Olympus’ EVIS EXERA II Duodenovideoscope
Olympus issued a
March 4, 2015: FDA Provides “Updated Information for Healthcare Providers Regarding Duodenoscopes”
The FDA provided information to physicians after Olympus duodenoscopes were involved in reports of infection. The FDA says it is not recalling the scope because “…removal of the device from the market could lead to an insufficient number of available duodenoscopes to meet the clinical demand in the United States…” However, the FDA does reinforce cleaning protocols for the device in hopes of decreasing the risk of infection.
2012 to 2015: Reports of Duodenoscope “Superbug” Outbreak Make News
Hospitals around the world report outbreaks of drug-resistant bacterial infections following the introduction of the Olympus TJF-Q180V duodenoscope. Erasmus Medical Center in the Netherlands reports one of the first outbreaks related to TJF-Q180V, involving 22 patients who had recently undergone procedures using the device. Olympus subsequently issued a safety warning in Europe, but not in the U.S. American hospitals soon reported outbreaks, including Virginia Mason Medical Center in Washington, Advocate Lutheran General Hospital in Illinois, and Ronald Reagan UCLA in California. Four of the patients infected at Virginia Mason died of their illness. The device’s difficult-to-sterilize elevator channel, which can trap patient fluids, is identified as a likely cause.
What Is the Olympus Scopes Lawsuit About?
People who have filed Olympus scopes lawsuits allege that device defects and inadequate warnings led them to experience serious infections and internal injuries. Here is a summary of the litigation:- Lawsuit summary: Olympus scopes lawsuits are product liability claims that allege some endoscope devices made by Olympus Medical Systems Corporation contain design and labeling defects that have resulted in patient injuries. These defects have led to potentially deadly infections or internal injuries in some patients.
- History of device recalls and warnings: Olympus endoscopes and accessories have been the subject of numerous FDA recalls, culminating in an import alert. This alert prevents future shipments of certain Olympus devices from entering the U.S.
- Injuries cited in lawsuits: Numerous patients have been diagnosed with antibiotic-resistant bacterial infections, internal bleeding, or burns.
- Procedures where Olympus scopes are used: The endoscopes are used in colonoscopies, bronchoscopies, enteroscopies, and gastroscopies utilizing Olympus scopes or accessories.
- How these injuries occur: The claims, which cite FDA recalls and warning notices, describe ineffective cleaning protocols, designs that trap fluids and are difficult to clean, and defects that can lead to mechanical injuries.
- Allegations: The lawsuits claim Olympus failed to promptly warn its markets of known design flaws.
What Procedures Are Olympus’ Scopes Used For?
Olympus’s scopes are used in common medical procedures involving the diagnosis, examination, or treatment of internal organs and systems. They are inserted into the patient, allowing physicians to see and operate internally. They are a core tool in many “minimally invasive” procedures. Common procedures that might use an Olympus scope include:
- Endoscopic Retrograde Cholangiopancreatography (ERCP): A procedure used to diagnose issues involving the liver, gallbladder, bile ducts, or pancreas. It may utilize the Olympus TJF-Q190V.
- Colonoscopy: A procedure primarily used to screen patients for rectal and colon polyps and cancers, it involves using a flexible scope to examine the rectum and colon. It may utilize products from Olympus’s EVIS EXERA III line.
- Bronchoscopy: A procedure that allows medical providers to examine and perform procedures on the lungs and air passageways using an endoscope or bronchoscope.
- Enteroscopy: A less common procedure that uses specialized endoscopes to get to the hard-to-reach small intestines, typically in cases of obscure bleeding or Crohn’s disease.
- Gastroscopy: A procedure that allows for operations in the upper GI tract, such as the stomach, duodenum, and esophagus. Olympus makes numerous products used in gastroscopy.
These procedures allow physicians to see deep inside the body without making large incisions.
What is an Endoscopic Procedure?
An endoscopic procedure uses a flexible tube with attached cameras to view internal structures in the body. Endoscopic procedures are often used as a less traumatic alternative to open surgeries, allowing physicians to diagnose or even perform some surgeries through orifices like the mouth or rectum, or through a small incision. So-called “minimally invasive” medical procedures often involve scopes.
Endoscopic Procedures & Olympus Scopes
Endoscopes are used in a large number of procedures. In addition to viewing internal structures, Olympus scopes may be used for:
- Performing surgeries
- Placing stents
- Removing blockages
- Widening passageway
- Sealing internal wounds
- Draining fluids
- Delivering medicines
There are different types of scopes made by Olympus, including endoscopes and duodenoscopes. The term “endoscope” refers to the family of instruments, while a duodenoscope is an endoscope with specialized side-viewing cameras used in a subset of endoscopic procedures, such as ERCP (i.e., endoscopic retrograde cholangiopancreatography).

What are endoscopes and why are Olympus endoscopes part of a lawsuit?
Which Olympus Devices Are Involved in Lawsuits, Safety Alerts, and Recalls?
The Olympus lawsuits involve several different models of endoscopes and accessories. Some of these models have been the subject of FDA recalls and field safety notices, with adverse events reported between 2012 and 2025 (see above timeline for some of these actions). These devices and their accessories have been associated with multiple reported deaths and hundreds of adverse event reports. Below are the Olympus medical devices that have been named in lawsuits, recalls, or warning letters.
TJF-Q180V Duodenoscope
The Olympus TJF-Q180V duodenoscope, trade name EVIS EXERA II, was designed to be a reusable scope for ERCPs. The scope had a critical design flaw in its closed elevator channel mechanism. The feature, which was billed as being easier to clean, ended up providing a trap for fluids. This fluid buildup allowed drug-resistant bacteria to collect in the device and infect some patients.
The TJF-Q180V, introduced in 2010, would soon be linked to superbug infection outbreaks, eventually leading to a recall and redesign. Olympus and one of its senior executives would end up pleading guilty to failing to report adverse events, resulting in an $80 million fine for the company. The redesign, which utilized disposable endcaps, was prone to dislodging during use, leading to lacerations and burns.
TJF-Q190V, TJF-Q290V, and TJF-Q170V Duodenoscopes
The 2020 successor to the TJF-Q180V, the Olympus TJF-Q190V (EVIS EXERA III), along with two additional Olympus duodenoscopes, the TJF-Q290V (EVIS LUCERA Elite), and TJF-Q170V (170-series duodenoscope), are also named in the lawsuits. These devices had two major issues:
- The MAJ-2315 disposable distal cover used by the scope devices was prone to detaching during procedures, a situation that could cause internal lacerations or block airways.
- The duodenoscope had an increased infection risk, especially if the Olympus duodenoscopes were not cleaned shortly after the procedure.
Olympus released updated cleaning protocols that recommended examining the devices for biological material under 10x magnification in 2025, after its duodenoscopes were linked to 2 more deaths and 5 injuries.
MAJ-891 Forceps/Irrigation Plug (Class I Recall)
Some accessories would continue to be a safety concern with Olympus endoscopes, including the MAJ-891 Forceps/Irrigation Plug, which was subject to an FDA Class 1 recall in 2025 after being linked to 120 injuries and one death. The reusable Olympus endoscope accessory is being pulled from the market as it can provide an excellent breeding ground for deadly bacteria when not properly disassembled.
ViziShot 2 FLEX (Class I Recall)
Another Olympus endoscope accessory named in lawsuits is the ViziShot 2 FLEX. The ViziShot 2 is a biopsy needle accessory used in bronchoscopies. A batch of the needles shipped before May 2025 have a defect that can potentially cause components to break off or detach mid-procedure, leading to tissue damage or blockages and a recall. One patient death has been reported from such a malfunction.
Bronchoscopes and Endobronchial Combustion Risk
The FDA issued a Class 1 recall on 19 models of Olympus bronchoscopes in June 2023 due to combustion risks. The recall includes 15,586 devices distributed between January 20, 1999, and May 3, 2023, models:
- BF-PE2
- BF-TE2
- BF-P60
- BF-1T60
- BF-MP60
- BF-XT40
- BF-1T150
- BF-P150
- BF-Q170
- BF-XT160
- BF-Q180-AC
- BF-P180
- BF-1T180
- BF-1TQ180
- BF-Q180
- BF-1TH190
- BF-H190
- BF-Q190
- BF-XT190
At the time the recall was issued, Olympus Corporation of the Americas had reported three patient injuries (including one patient death). Voluntary label updates made by Olympus in October 2025 advise surgical teams to monitor oxygen concentrations before firing a laser and taking other precautions.

How Can Olympus Scopes Transmit Infection?
Reusable Olympus endoscopes have been linked to infections after patients were exposed to drug-resistant bacteria. These infections can occur when biological materials (e.g., fluids) become trapped in the devices’ channels, even after the scope has been disinfected. Reusable endoscopes, including those made by Olympus, undergo reprocessing and sterilization between uses. If the devices cannot be sterilized, they may transmit infection.
Reports of contamination persisted through several different models, design changes, and cleaning protocol updates issued by Olympus. Cleaning protocols are designed and prescribed by the endoscopes’ manufacturer, in this case, Olympus. Delaying manual cleaning was found to increase the risk of infection.
According to precleaning and manual cleaning instructions issued by Olympus, steps to disinfect Olympus scopes include:
- Precleaning immediately after use at the bedside
- Leakage testing
- Manual cleaning with brushing and flushing in the reprocessing room
- Visual inspection under 10x magnification
- High-level disinfection or sterilization
- Drying and storage
The FDA, the Center for Disease Control and Prevention (CDC), and the American Society for Microbiology also release surveillance sampling and culturing recommendations to monitor the state of medical instruments. Postmarket surveillance studies suggest rates of “high-concern” contamination rates of 4% to 6% with older fixed-cap endoscopes and around 0.5% with models using disposable endcaps.
Olympus Scopes and Antibiotic-Resistant Superbug Infections
Some of the Olympus scope infections, particularly those associated with the older TJF-Q180V duodenoscope, involved an antibiotic-resistant type of bacteria called Carbapenem-Resistant Enterobacteriaceae (CRE). Known more colloquially as “nightmare bacteria” or a form of “superbug,” CRE is resistant to even the last resort antibiotics (e.g., carbapenems) used to treat the most stubborn infections. CRE can be life-threatening, especially if it enters the bloodstream.
What Infections Have Been Linked to Olympus Scopes?
Olympus scopes have been a vector for infectious organisms, primarily bacteria. A 2022 clinical study of 16 clinical sites in the U.S. sampled TJF-Q180V and TJF-160F/VF duodenoscopes. The TJF-Q180Vs had a 4.1% contamination rate with High-Concern organisms. These were:
- Klebsiella (gastrointestinal origin)
- Enterococcus (gastrointestinal)
- Escherichia (gastrointestinal)
- Pantoea (gastrointestinal)
- Pluralibicter pyrinus (gastrointestinal)
- Staphylococcus lugdunensis (human origin, non-gastrointestinal)
- Staphylococcus aureus (human)
- Acinetobacter (environmental origin)
- Candida (environmental and human)
- Erwinia bilingae (environmental)
- Pseudoxanthomonas (environmental)
- Ralstonia (environmental)
- Roseomonas (environmental)
- Sphingomonas mucosissima (environmental)
- Brevundimonas (waterborne)
- Massilia (waterborne)
- Pseudomonas (waterborne)
Klebsiella, Enterococcus, and Escherichia are all highly resistant to antibiotics, with Klebsiella in particular accounting for most CRE infections. Although these bacteria often exist harmlessly in the gastrointestinal tract, a contaminated scope can “deliver” them to parts of the body that lack natural defenses against gut bacteria, such as the bile duct or the bloodstream. This can result in sepsis, bloodstream infections, or organ failure, particularly in immunocompromised individuals.
What Are the Signs of Infection After an Endoscopy or Colonoscopy?
Serious infections tend to show up quickly after a contaminated endoscopy or colonoscopy. The onset for endoscope-related sepsis is typically between 24 and 48 hours, though some infections can take as long as a month to present. Symptoms can vary depending on which organs or systems are infected. Common symptoms of infection after a scoping procedure include:
- Fever and chills
- Low blood pressure
- Elevated heart rate
- Cough (lung infections)
- Abdominal pain (abdominal infections)
- Rash (sepsis)
- Joint pain (sepsis)
These symptoms can come on suddenly and intensely. It is advised that people experiencing any of these symptoms after a scoping procedure seek medical attention immediately.
Studies and Statistics About Infections After Endoscopic Procedures
Several high-impact studies have been published on endoscopic infections. Notable studies include:
- A 2018 study published in the journal Gut: A study called “Rates of infection after colonoscopy and osophagogastroduodenoscopy in ambulatory surgery centres in the USA” found that post-endoscopic infections were more common than previously thought, with 3 in 1,000 patients developing an infection within 7 days of an osophagogastroduodenoscopy. Ethics approval was conducted by Johns Hopkins Medicine Institutional Review Board.
- A 2013 Georgetown study on ERCP complications: The study (called “Complications of Endoscopic Retrograde Cholangiopancreatography: How to Avoid and Manage Them”) found an acute cholangitis (a serious infection of the bile ducts) infection rate of 1.4% of ERCP patients, with a mortality rate of 7.85%.
- A
2025 review of digestive endoscopy-associated infections : A scientific review titled “A Comprehensive Review of Digestive Endoscopy-Associated Infections: Bacterial Pathogens, Host Susceptibility, and the Impact of Viral Hepatitis” notes the need for longer-term monitoring of ERCP patients, citing a study (Hutfless et al.) that analyzed over 800,000 ERCPs. It reported a post-procedural infection rate of 3.5% at 7 days and 7.7% at 30 days. Risk factors for infection included emergency ERCP, pre-existing infections, chronic comorbidities, age over 65 years, male sex, and non-white ethnicities.
Additional studies have associated ERCPs with a higher risk of infections than most other endoscopic procedures.
Can Olympus Scopes Cause Mechanical Injury or Organ Perforation?
Olympus scopes have also been linked to mechanical injuries and burns. Design flaws in specific scope or accessory models can perforate organs, cause internal bleeding, or cause other traumas. Reported injuries from Olympus scopes include:
- Airway and internal fires from bronchoscopes
- Lacerations from disposable endcaps
- Organs becoming snagged by disposable endcaps
- Needle and tip detachment with biopsy accessories
These device malfunctions are distinct from physician malpractice and can occur under proper use and may support a product defect claim.
What Did the 2018 Department of Justice Case Against Olympus Establish?
The 2018 Department of Justice case against Olympus ended in a guilty plea, with the company admitting it failed to file adverse event reports with the FDA following documented infections in Europe. The Pseudomonas aeruginosa and Escherichia coli outbreaks occurred after patients were treated with Olympus’s TJF-Q180V duodenoscope.
Olympus was fined $80 million. The senior official overseeing the division, Hisao Yabe, also pleaded guilty and was sentenced to one year’s probation and a $5,000 fee. The case underscored the importance of timely regulatory reporting and post-market surveillance obligations.
What Is the 2025 FDA Import Alert on Olympus Devices?
On June 24, 2025, the FDA issued an import alert on 58 Olympus devices, giving the agency the authority to prevent future shipments of those devices from entering the United States. Olympus Medical Systems Corporation is headquartered in Japan. The alert cited ongoing quality system violations at Aizu, its manufacturing site. Devices identified include models of:
- Bronchoscopes
- Laparoscopes
- Automated endoscope reprocessors
- Ureterorenoscopes
The import alert also contained a warning to providers currently using those Olympus devices to strictly follow reprocessing procedures for reusable scopes.
Who Can File an Olympus Scopes Lawsuit?
King Law’s eligibility criteria for filing an Olympus scopes lawsuit are as follows:
- You had a medical procedure that utilized an Olympus scope.
- You developed sepsis or an infection requiring intravenous antibiotics after an endoscopic procedure (including a colonoscopy or an ERCP procedure).
- You had no history of such an infection within a year prior to the procedure.
These criteria represent our current eligibility requirements. However, these requirements can change. Contact our firm if you have any questions. King Law is investigating multiple claim types for Olympus scopes, including lawsuits related infections after a colonoscopy.
Patients can seek compensation for out-of-pocket expenses, lost wages, or lost earning potential resulting from the injury. They may also qualify for non-economic damages such as pain and suffering or loss of quality of life related to their injuries.
Can Family Members File a Claim for Wrongful Death?
In cases where an individual died of an Olympus scope infection, family members may be able to file a wrongful death lawsuit. The criteria are similar, but may have different statute of limitations depending on your jurisdiction. For example, the statute of limitations would begin at the time of death rather than the discovery of the injury.
What Evidence Is Needed to File an Olympus Scopes Lawsuit?
While each case is unique, many Olympus scopes lawsuits will require similar evidence to support their claims. Here are some types of evidence that are commonly used to support Olympus scopes claims:
- Medical records confirming infection or injury
- Procedure records identifying the specific Olympus device used
- Hospital notification letters (if applicable)
- Culture reports or infectious disease diagnoses
- Studies and regulatory actions establishing a link between the device model and infections
- Medical staff depositions
Note that you do not have to collect all of this evidence yourself. Your attorney can assist you in obtaining medical device records and regulatory documentation to support your legal claim.
How Can I File an Olympus Scopes Lawsuit?
If you file an Olympus scopes lawsuit for yourself or a loved one, you will need to follow a legal process. Here are the typical steps to filing a lawsuit related to infections and injuries caused by an Olympus scope:
- Get a free consultation with an experienced attorney.
- Hire an attorney when you find one who suits your needs.
- Identify the Olympus device used in your procedure.
- Complete investigation and collection of evidence, including review of recall statuses, infection timing, and witness depositions.
- Identify defendants and file your claim.
- Pursue compensation through a settlement with defendants or litigation.
Your attorney can help you gather evidence and testimonies, as well as select the best legal theory under which to pursue your case.
How Long Does an Olympus Scopes Lawsuit Take?
Timelines for resolving medical device cases, like those involving Olympus Scopes, can vary substantially. How long a civil lawsuit takes will depend on factors such as your circumstances and the circumstances of other similar cases against Olympus.
The cases, though numerous, haven’t yet been consolidated into a multidistrict litigation (MDL), which would eventually expedite such cases. Other factors that could affect resolution times are the number of defendants, the severity of the injury, and the damages sought.
Is There a Deadline to File an Olympus Scopes Lawsuit?
Olympus scopes lawsuits must be filed within a deadline determined by the statute of limitations. Statutes of limitations differ by state and legal claim. Medical device cases typically fall under personal injury or wrongful death deadlines. The statute of limitations typically will begin on the date the individual discovered their injury or, in wrongful death cases, the date of the victim’s death. However, exceptions may be made if you only recently discovered your injury was due to negligence on the part of Olympus. Consult with an attorney to verify deadlines that apply to your case.
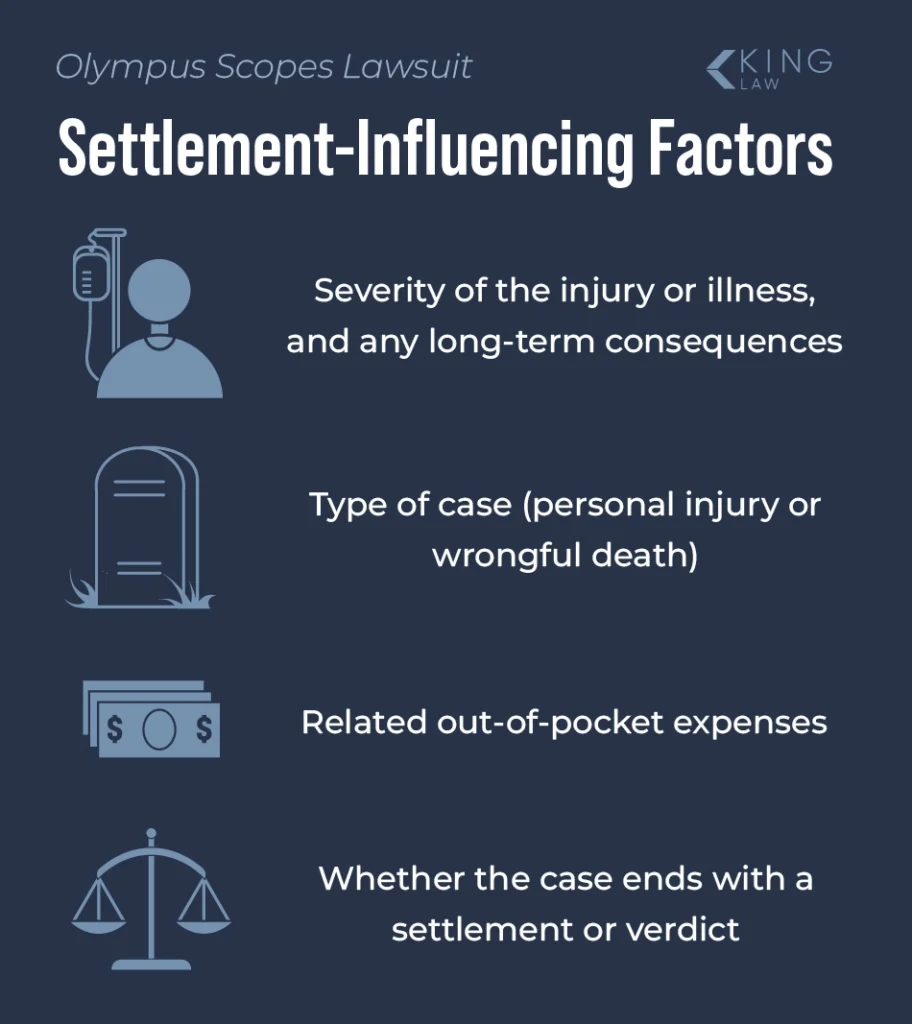
How Much Is an Olympus Scopes Lawsuit Worth?
Compensation awarded in an Olympus scopes lawsuit depends on a number of factors. These include:
- The severity of the illness or injury
- Whether the case is personal injury or wrongful death
- Out-of-pocket expenses related to the injury
- Long-term damages associated with the injury
- Whether the case ends in a settlement or jury verdict
Long-term damages may include both economic and non-economic considerations, such as lost earning potential, pain and suffering, and loss of companionship (in the case of wrongful death).
Bottom Line – What Patients and Hospitals Should Know
The Olympus scopes lawsuits involve a complicated timeline of product defects, alleged corporate negligence, and regulatory actions. Here are the big takeaways:
- Olympus scopes have been subject to multiple recalls.
- Contamination risks are the result of reprocessing complexity and design flaws.
- Regulatory scrutiny of Olympus has increased since 2015.
- The infection risks are quite serious and can be life-threatening.
- Olympus has had quality control issues that have also resulted in mechanical injuries during endoscopic procedures.
Most of these cases are still in progress.
King Law Is Investigating Olympus Scopes Infection Claims
King Law is currently investigating Olympus scope infection cases nationwide, including lawsuits involving bronchoscopes, duodenoscopes, endoscopes, and similar devices. Our attorneys have decades of experience securing compensation in complex medical device cases. Our lawyers work on a contingency basis, so there are no upfront costs to pursuing your case. We are committed to holding manufacturers accountable for device defects and negligence. Read more about our early involvement in the Olympus scopes litigation.
Contact an Olympus Scopes Lawsuit Attorney Today
If you or a family member has been diagnosed with an antibiotic-resistant infection after undergoing an endoscopic procedure with an Olympus device, you may be due compensation for your injuries. Contact King Law for a free, no-obligation case evaluation at (585) 496-2648. There’s no obligation to move forward unless you want to.
Frequently Asked Questions (FAQs)
The Olympus scopes lawsuits allege that the medical device company failed to disclose the risks of antibiotic-resistant infections related to endoscopic procedures that used their devices, despite receiving multiple reports of injuries.
More than 50 Olympus device models have been subject to recalls or regulatory actions, including several models of bronchoscopes, duodenoscopes, and endoscope accessories.
The FDA issues a Class 1 recall in instances where a product has a reasonable chance of causing serious injury, serious illness, or death. Examples include a contaminated or mislabeled product or a device with a high chance of catastrophic failure.
Both ERCP and colonoscopy procedures can cause a superbug infection, but research shows that an ERCP has a higher chance of doing so.
Infections involving numerous types of bacteria and yeast have been linked to Olympus endoscopes. The most dangerous infections involve sepsis, bloodstream infections, and organ failure.
CRE are bacteria that are resistant to carbapenems, the antibiotics of last resort. These infections are very hard to treat.
The small channels in reusable endoscopes can provide ideal breeding grounds for bacteria, which can resist chemical disinfectants. Contaminated endoscopes can then transmit the bacteria to patients during subsequent procedures.
Olympus duodenoscopes provide small channel surfaces and chambers that are ideal breeding grounds for bacteria that produce chemical-resistant biofilm. One particular model featured a “sealed” elevator chamber that couldn’t be reached by mechanical brushes for cleaning.
Olympus was fined $80 million for failing to disclose adverse events involving its TJF-Q180V duodenoscope to the FDA. A senior official also pled guilty, facing probation and a $5,000 fine.
The 2025 FDA Import Alert identified 58 Olympus devices manufactured in Japan with persistent quality control issues. It gives the FDA the authority to prevent those models from entering the U.S. market.
Symptoms that may indicate an infection or injury after an endoscopy include fever, chills, rashes, swelling, coughing, or bleeding.
An endoscope-related infection may present in as little as 24 hours or as long as 30 days or more.
Yes, Olympus scopes have been linked to both sepsis and organ failure due to infection.
Some Olympus scope accessories have had design flaws that can mechanically injure patients, causing perforations, lacerations, snagging, choking, or burns.
Individuals who presented with an antibiotic-resistant infection or serious injury after an endoscopic procedure.
Infection reports, medical records, witness testimonies, regulatory actions, and studies can all serve as evidence in an Olympus scopes lawsuit.
The deadline to file an Olympus scopes lawsuit varies from state to state. Most statutes of limitations fall between 1 and 3 years from the discovery of the injury or time of death. However, some factors affect deadlines. An attorney can advise on filing deadlines.
No, the Olympus scopes case is currently a collection of individual lawsuits, though it could be consolidated into an MDL in the future.
The amount someone alleging injuries from Olympus scopes could receive will vary based on personal and external factors in the lawsuit. People claiming more severe injuries may be able to pursue higher amounts in damages.

