Complete the form below to see if you qualify for a Rybelsus Lawsuit
Our firm is currently not evaluating cases of gallbladder complications.

Rybelsus Lawsuit Overview
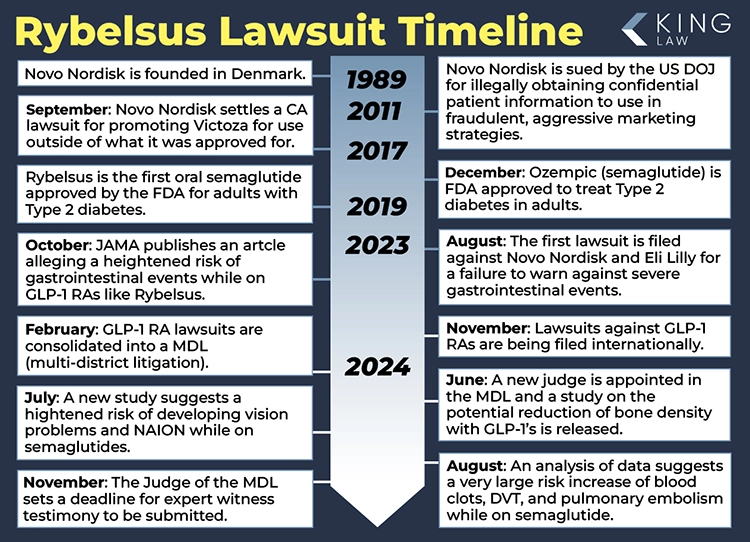
People are filing Rybelsus lawsuits after experiencing numerous severe side effects from taking the drug. Rybelsus, the brand name for an oral semaglutide manufactured by Novo Nordisk, has come under scrutiny after patients have reported serious side effects. Rybelsus originally received FDA approval in 2019 and is used to treat type 2 diabetes.
It works by increasing insulin and decreasing blood sugar. The drug also causes delayed gastric emptying. Lawsuits allege that severe delays in gastric emptying have led people to develop conditions like gastroparesis (stomach paralysis) and bowel obstructions.
Lawsuits allege that the manufacturer failed to properly warn consumers about the potential for adverse health conditions associated with using the drug. Some health issues patients have reported are severe and can lead to long-term complications. Individuals who have suffered severe side effects after taking Rybelsus may be eligible to take legal action and should consult with an attorney as soon as possible.
Our firm is currently investigating Rybelsus lawsuits involving:
- Gastroparesis/stomach paralysis
- Gastrointestinal obstruction/ileus
- Daily vomiting lasting at least 3 weeks
- Vision loss/blindness/NAION
- Blood clots/deep vein thrombosis (DVT) or pulmonary embolism (PE)
If you have been diagnosed with any of the above conditions, or have experienced severe and prolonged vomiting, please contact us right away for a free Rybelsus lawsuit evaluation.
Rybelsus Lawsuit Update – 2026 Update
March 5, 2026: Rybelsus Manufacturer, Novo Nordisk, Cited for Not Reporting Adverse Drug Events to FDA
The FDA has sent a warning letter to Novo Nordisk because it says the company’s written policies for tracking, reporting, and evaluating postmarketing adverse drug experiences to the FDA were insufficient. According to the FDA’s rules, the manufacturer of Rybelsus is supposed to send the FDA a report within 15 days after it finds out about an adverse drug event experienced by someone taking the company’s products. The company is also required to have an appropriate process for monitoring and evaluating these reports. However, the FDA found that the company did not have or follow the proper procedures for notifying the FDA about these adverse event reports. Moreover, the FDA report references many instances where Novo Nordisk canceled, rejected, or failed to report multiple adverse drug experiences about patients taking semaglutide (the active ingredient in Rybelsus).
March 2, 2026: Over 3,200 Claims Filed by Patients Who Took Rybelsus, Other GLP-1s
As of March 2, 2026, there are 3,363 pending lawsuits in MDL -3094 IN RE: Glucagon-like Peptide-1 Receptor Agonists (GLP-1 RAs) Products Liability Litigation. These claims have been filed by patients who took Rybelsus or another type of GLP-1 and experienced gastrointestinal and other serious injuries. Some of the patients who have filed lawsuits say they were not told that Rybelsus could cause ileus, stomach paralysis (gastroparesis), acute pancreatitis, or other severe complications. Legal filings allege that some patients who took Rybelsus had to be hospitalized to treat these severe complications, and some have died. Investigations are still underway to determine if Rybelsus or another GLP-1 caused or contributed to these conditions and deaths.
February 26, 2026: Competitor’s New GLP-1 Tablet May Have More Side Effects in Head-to-Head Clinical Trial Against Rybelsus
Novo Nordisk’s competitor (Eli Lilly) is developing an oral GLP-1 pill (i.e., orforglipron) that is similar to Rybelsus. In a recent clinical trial of this new drug, 58% of participants taking the competitor’s drug experienced “mild-to-moderate side effects,” and about 10% stopped the medication because of side effects. In contrast, 48% of participants taking Rybelsus reported side effects, and 5% left the clinical trial due to side effects. Even so, Rybelsus has been called out in lawsuits filed by patients who say they suffered severe, unexpected complications. These lawsuits claim that Novo Nordisk should have told patients that Rybelsus may increase the risk of possible side effects like gastroparesis, bowel blockages, blood clots, and pancreatitis.
February 4, 2026: New Form of Oral GLP-1 Like Rybelsus May Be Available Later this Year
Rybelsus is Novo Nordisk’s oral semaglutide tablet that has been on the market since about 2019. Like the injectable form known by a different name, the drug is prescribed to help people manage their type 2 diabetes and may also be given for off-label weight loss purposes. Novo Nordisk says it may launch a new version of Rybelsus in the second quarter of 2026, available in different doses than Rybelsus’ current offerings. Novo Nordisk and other GLP-1 manufacturers are facing dozens of lawsuits filed by people who say they took Rybelsus or another type of GLP-1 and developed unexpected and severe complications.
February 2, 2026: Brazilian Company May Offer Generic Semaglutide Drugs, Like Rybelsus
Hypera, a drug company in Brazil, has announced plans to make a cheaper, generic version of semaglutide in March 2026, when Novo Nordisk’s patent protections expire in Brazil. Semaglutide is the active ingredient in Rybelsus, as well as Ozempic and Wegovy. In November 2025, Brazilian courts denied a request from Novo Nordisk to extend the medical patent for 5 years. Novo Nordisk has renewed lobbying efforts to extend the patent. If Hypera is allowed to make a generic version of semaglutide, that could mean they gain market share for Rybelsus and Novo Nordisk’s other semaglutide GLP-1 drugs.
January 30, 2026: FDA Approves Labeling Update for Rybelsus (Semaglutide) Tablets
The FDA has updated the label for Rybelsus, a semaglutide pill prescribed for diabetes management and other approved uses. The update adds the term “semaglutide tablets” and a branded reference to Novo Nordisk’s new oral semaglutide that will be released soon. The new label may clear up misunderstandings some patients may have about what Rybelsus is and the other semaglutide pill available. Rybelsus patients have brought legal actions against Novo Nordisk, alleging that the medication caused them to experience severe complications that were not included on Rybelsus’ previous warning labels.
January 16, 2026: Court in Brazil Denies Patent Extension Request for Rybelsus Tablets, U.S. Rybelsus Lawsuits Continue
A Brazilian court has rejected Novo Nordisk’s request to extend its patent protection for its Rybelsus tablets in that country. Rybelsus is used in the U.S. and elsewhere to help patients with diabetes. Without the patent protections, generic pharmaceutical companies may be able to make lower-cost versions of Rybelsus in Brazil. U.S. patients who took Rybelsus have experienced severe side effects, like gastroparesis, pancreatitis, and blood clots, which they say they were not warned about. They claim Novo Nordisk should have included warnings about these complications on Rybelsus’ label.
January 2, 2026: Rybelsus Patients in U.K. Advised to Be Careful When Transitioning to New Formulation
The formula for Rybelsus medication in the UK may be changing in 2026. However, the old and new versions may be available at the same time during the transition. As a result, the UK government is advising patients and their doctors to pay close attention to the differences in formulas and how this may affect dosing decisions. U.S. patients are suffering from complications after taking Rybelsus for diabetes. They are filing lawsuits because they allege they were not properly warned that side effects like gastroparesis or pancreatitis could occur.
December 15, 2025: Nearly 3,000 Lawsuits Pending Against Rybelsus’ Novo Nordisk and Other GLP-1 Manufacturers
The number of lawsuits involving Rybelsus and other GLP-1 drug complications has reached 2,947 in the federal MDL (MDL 3094) against Novo Nordisk and its competitors. These lawsuits have been filed by Rybelsus patients and their surviving loved ones, and they accuse Novo Nordisk of misleading consumers about the likelihood of some side effects. For example, Rybelsus patients have lost their vision from NAION (reduced blood flow to the eye), which occurs suddenly and is usually irreversible. These lawsuits have been consolidated into a federal MDL, and more patients can join if they have experienced complications after taking Rybelsus. Read more about the Rybelsus vision loss MDL here.
December 1, 2025: Novo Nordisk Stock Dips After Rybelsus Has Poor Showing in Clinical Trials
Rybelsus’ maker, Novo Nordisk, experienced a 5% loss in its share price. This drop came after Rybelsus showed disappointing results in a clinical trial. Novo Nordisk was looking to broaden its semaglutide drugs’ clinical applications. So, researchers were investigating whether Rybelsus might decrease Alzheimer’s symptoms. However, in phase III evoke and evoke+ trials, Rybelsus failed to control Alzheimer’s symptoms better than a placebo. Meanwhile, Novo Nordisk faces new lawsuits filed by people who experienced severe side effects and disease after using Rybelsus.
November 14, 2025: Rybelsus Use Rises in India, Sparking Concerns from Doctors
Indian doctors have reported an unprecedented surge in patients seeking Rybelsus prescriptions to help lose weight. However, the doctors caution against relying on unnatural ways to shed pounds. Diabetologists say that most GLP-1 patients regain lost weight within a year after they stop taking the drug, which encourages dependence. Weight loss also tends to stop once patients lose “about 15% of their body weight.” Patients in the United States are filing lawsuits against Novo Nordisk and other drug companies, alleging insufficient warning labels on Rybelsus and similar products.
October 17, 2025: FDA Approves Rybelsus To Reduce the Risk of Major Adverse Cardiovascular Events in Adults with Diabetes
Despite the numerous negative side effects experienced by people taking Rybelsus, Novo Nordisk announced the FDA has approved the drug as a cardiovascular event preventative. Rybelsus is now indicated as a preventative medication for major adverse cardiovascular events (MACE) for people with type 2 diabetes. Although the drug may prevent some severe cardiovascular events and deaths, it carries other severe risks. Thousands of people have filed lawsuits against Novo Nordisk, alleging that Rybelsus and other GLP-1 drugs made by the company caused severe illnesses and injuries.
October 16, 2025: Rybelsus Patients Have Higher Risk of Kidney Problems Than Tirzepatide
A new study has found that semaglutide drugs, like Rybelsus, are associated with a higher rate of acute kidney injury than other types of GLP-1 drugs. The authors also tracked an increase in hospitalizations and deaths for those taking Rybelsus (semaglutide) versus those taking tirzepatide drugs. Rybelsus is widely popular for helping patients manage type 2 diabetes. However, Rybelsus comes with side effects like delayed gastric emptying, vision problems, kidney injuries, and other complications. Patients are filing Rybelsus lawsuits in state and federal courts.
October 1, 2025: Rybelsus Lawsuits Gain Momentum as Novo Nordisk Expands Reach in the European Union
Thousands of people have taken Rybelsus to help manage their diabetes. However, this oral medication has allegedly caused some people to experience debilitating side effects, like gastroparesis, vision loss, and pancreatitis. They are filing lawsuits against Novo Nordisk for making a dangerous drug and not warning them about the safety risks. Even so, Novo Nordisk is requesting a label change in the European Union to encourage people to take Rybelsus for cardiovascular issues, too.
September 15, 2025: Study Finds Over Half of New Rybelsus Users Stop the Drug Within a Year
A new Danish study found that more than half of new Rybelsus patients stopped using the semaglutide injections within a year after starting them. The study included 77,310 new patients who were taking semaglutide drugs like Rybelsus. Of those patients, 40,262 went off the drug within a year. Researchers believe the high costs and potential complications may have led participants to stop this medication early. Rybelsus patients in the United States are filing lawsuits against Novo Nordisk for failing to warn them of unwanted and sometimes life-threatening side effects, including blindness and a stopped or slowed digestive system.
September 4, 2025: Hundreds of New Lawsuits Filed For Rybelsus and GLP-1 Drugs
Rybelsus has helped many people treat their type 2 diabetes, but the drug has also caused severe side effects in some patients. The drug’s maker, Novo Nordisk, is being sued by people who have experienced side effects like severe gastrointestinal conditions, blood clots, vision loss, and pancreatitis. Between August and September 2024, 486 new people joined the group lawsuit for people harmed by Rybelsus and other GLP-1 drugs. There are now 2,676 active lawsuits in the litigation. Those harmed by Rybelsus should contact a lawyer about eligibility for a lawsuit.
August 21, 2025: Rybelsus Side Effect Lawsuits Carry a $2 Billion or Higher Price Tag, Experts Say
Lawsuits against Rybelsus’ manufacturer, Novo Nordisk, may have a price tag of $2 billion or more, according to legal experts. Rybelsus is an oral medication prescribed off-label to suppress hunger and increase weight loss. However, patients claim they were not told about the possible complications. The number of cases involving glucagon-like peptide-1 (GLP-1) drugs, such as Rybelsus, now totals over 2,100 lawsuits. More patients are continuing to file individual claims after they suffer complications from Rybelsus.
August 19, 2025: Judge Decides Which Rybelsus Claims Can Continue and Which Need to Be Refiled
Novo Nordisk had filed a motion to dismiss, attempting to throw out many of the claims made by patients who took Rybelsus and developed complications like gastroparesis. After hearing arguments in support and against this motion, Judge Marston issued a decision. She has allowed some of the Rybelsus claims to continue, but others need to be amended first. The claims that are still standing include those that said Novo Nordisk was untruthful in how it described Rybelsus and its side effects on the label. Some of the other allegations being brought against Novo Nordisk were tossed because they were not specific enough. But the judge gave instructions about what information the plaintiffs would need to refile those later on.
August 18, 2025: Plaintiffs Must Have Gastric Emptying Test to Support Gastroparesis Claim Against Rybelsus
Rybelsus is a popular GLP-1 that many people take to improve their diabetes symptoms or heart health. However, some Rybelsus patients allege the drug caused gastroparesis, which is when the gut slows down and does not empty its contents. People have sued Rybelsus and Novo Nordisk, claiming the company did not adequately warn them of this risk. To have a chance of winning their claims, they need to prove they received a valid gastroparesis diagnosis. The judge overseeing the group Rybelsus claims has decided that gastric emptying studies are the approved method to diagnose gastroparesis. While some of the current Rybelsus claimants had a gastric emptying study (like a breath test) conducted before filing their claim, others did not. The plaintiffs who have not had this test already may ask their doctor to perform it now and submit the results to the court.
August 15, 2025: New Oral GLP-1 May Outperform Rybelsus and Lower Side Effect Risks
Rybelsus is an oral GLP-1 that helps people manage their diabetes. According to hundreds of lawsuits filed across the country, Rybelsus may cause severe complications in some people. Drug company Lexaria has developed a dissolvable form of Rybelsus capsules that may outperform the existing Rybelsus drug in terms of weight loss benefits. Additionally, the small clinical trials showed that patients taking the dissolvable form of Rybelsus did not experience as many side effects.
August 1, 2025: People Who Lost Their Vision on Rybelsus May Be Able to Join Pending Group Lawsuit
A request filed in the state of New Jersey may lead to the creation of a group lawsuit for people who took Rybelsus and developed NAION (non-arteritic anterior ischemic optic neuropathy). NAION is a type of sudden vision loss that is often permanent. A growing body of research suggests that Rybelsus and other GLP-1 drugs may be associated with this serious condition. People who took Rybelsus and lost their vision should consult with an experienced attorney to understand their options for possible compensation.
July 17, 2025: World Health Organization Issues Warning About Rybelsus Causing Serious Eye Condition
The World Health Organization has warned prescribers that Rybelsus users have a higher chance of developing a severe eye condition. The disorder, called non-arteritic anterior ischemic optic neuropathy (NAION), can come on suddenly and cause permanent vision loss. While NAION is rare, the rates among Rybelsus patients were high enough to catch the attention of the medical community and prompt recent changes to the Rybelsus label in Europe.
July 2, 2025: Rybelsus Lawsuits Climb as More Patients Are Injured by GLP-1 Drug
The total number of cases against Novo Nordisk and other GLP-1 makers has now reached 1,882, including lawsuits filed over Rybelsus side effects. This is an uptick of 83 from May. Rybelsus patients who experienced severe complications are suing the pharmaceutical company that makes this oral diabetes drug. They believe Rybelsus caused them to suffer from eyesight loss, pancreas issues, a paralyzed gut system, and other side effects.
June 10, 2025: EMA Finds Rybelsus Increases the Risk of Eye Strokes and Vision Loss
After reviewing non-clinical studies, clinical trials, post-marketing surveillance, and medical literature, the European Medicines Agency (EMA) has determined that Rybelsus increases a person’s risk of developing NAION. When someone has NAION (non-arteritic anterior ischemic optic neuropathy), they experience sudden and usually permanent vision loss. The EMA notes that the condition is still rare, but due to the high number of people taking semaglutide drugs like Rybelsus, patients should be aware of this potential side effect. NAION is one of the conditions that has led people to sue the GLP-1 drug’s manufacturer, Novo Nordisk.
June 3, 2025: Courts May Have to Decide on Diagnosis Criteria in Rybelsus Lawsuits
Many patients harmed by Rybelsus have joined a group lawsuit for people harmed by GLP-1 drugs. That group lawsuit is called the Glucagon-like Peptide-1 Receptor Agonists (GLP-1 RAs) Products Liability Litigation. Attorneys on both sides are trying to reach an agreement about diagnostic criteria for one of the chief injuries in the case, gastroparesis. Plaintiffs want to use a common medical practice called a differential diagnosis. Defendants argue that to receive compensation, patients must meet specific testing requirements. The decision of the courts could have a major impact on the outcome of Rybelsus lawsuits.
May 5, 2025: More Lawsuits Filed Against Novo Nordisk for Rybelsus and Other GLP-1s
There are now 1,809 lawsuits pending against GLP-1 manufacturers as part of the federal, group litigation. This litigation represents people harmed by Rybelsus and other GLP-1 drugs. It is expected that thousands more plaintiffs will join this lawsuit as they experience the negative and severe side effects of Rybelsus.
April 16, 2025: Experts Called To Testify at May 14th Hearing in Rybelsus Lawsuit
Gastroparesis, or paralysis of the stomach, intestines, and gut, is one of the adverse reactions people are alleging after taking Rybelsus and other GLP-1s. This has led people to file lawsuits against Novo Nordisk and other pharmaceutical companies. On May 14, 2025, experts will be called in to testify at a hearing about the diagnostic process for gastroparesis. Understanding this process will give plaintiffs evidence to build their cases and create a strong legal strategy against the at-fault drug companies.
April 2, 2025: As Rybelsus Boasts Cardiovascular Benefits, Other Experience Severe Negative Side Effects
Data from a recent medical trial have demonstrated that Rybelsus cuts the risk of cardiovascular events by 14%. This news may have mixed reception from patients who are weighing the pros and cons of taking Rybelsus. Some people experience severe gastrointestinal injuries, vision loss, and blood clots while taking Rybelsus. Some of those people have pursued lawsuits to be compensated for their injuries. The total number of lawsuits against Novo Nordisk and other GLP-1 manufacturers topped 1,600 in March, and more people are expected to join this litigation as they sustain injuries from Rybelsus.
March 3, 2025: Lawsuits Involving Rybelsus Gain Momentum in 2025
Many people who experienced severe digestive injuries and other severe side effects after taking Rybelsus have joined a group lawsuit against the drug’s manufacturer, Novo Nordisk. There are now 1,521 active lawsuits in the group lawsuit against GLP-1 drug makers (the Glucagon-like Peptide-1 Receptor Agonists (GLP-1 RAs) Products Liability Litigation). As more people harmed by Rybelsus file lawsuits, Judge Karen Spencer Marston is pressing the litigation forward. At a conference last week, she set a date for the Rule 702 hearing, which is a hearing where she will decide what information from which expert witnesses will be included in the trial. This hearing will take place on May 14, 2025. People harmed by Rybelsus still have time to file lawsuits and join this group litigation.
February 6, 2025: Lawsuits Against Rybelsus Manufacturer Novo Nordisk Increase
143 new lawsuits were filed related to injuries experienced after taking Rybelsus and other GLP-1 drugs. Between December 2024 and February 2025, 143 lawsuits were filed as part of the consolidated federal litigation against GLP-1 drug manufacturers. People continue to experience severe gastrointestinal injuries after taking Rybelsus. As people are injured by Rybelsus, they continue to join the consolidated lawsuit.
December 13, 2024: Warning Label for Rybelsus Updated to Include Gastrointestinal, Pancreatic, and Gallbladder Language
On December 9, 2024, the FDA posted an updated drug warning label for Rybelsus. The new label warns of acute pancreatitis, including fatal and non-fatal hemorrhagic or necrotizing pancreatitis. The FDA also added that patients who took Rybelsus were 2 to 7 times more likely to develop severe gastrointestinal issues than patients who took the placebo. The new label also warns of the risk of acute gallbladder inflammation, called cholecystitis. These label updates reflect the severe side effects some people experience when taking GLP-1 drugs like Rybelsus. These injuries have led some patients to file lawsuits against Novo Nordisk.
December 2, 2024: 1,300 More People File Rybelsus and Other GLP-1 Lawsuits in November
The multidistrict litigation lawsuit (MDL) against Rybelsus manufacturer Novo Nordisk and other GLP-1 manufacturers has 1,300 pending lawsuits as of December 2, 2024. Many people who have filed lawsuits claiming that GLP-1 drugs caused them severe injuries have joined MDL 3094, Glucagon-like Peptide-1 Receptor Agonists (GLP-1 RAs) Products Liability Litigation. As more people take GLP-1 drugs like Rybelsus to treat type-2 diabetes, it is likely more people will join the MDL with the goal of receiving compensation and settlements.
November 22, 2024: Scientists Say More Research Is Needed on Rybelsus and Other GLP-1 Drugs and Possible Cancer Risks
A recent study examined data from people who took GLP-1 drugs like Rybelsus for 1 to 3 years. Researchers found those people had a 58% increased risk of developing cancers of the thyroid. The study, which was titled “Glucagon-like peptide 1 receptor agonists and thyroid cancer: is it the time to be concerned?” and published in Endocrine Connections, encouraged the scientific community to further examine GLP-1 drugs and whether they increase the risks of certain cancers.
November 7, 2024: Deadline for Submitting Expert Reports Set in Rybelsus Litigation
The judge in the GLP-1 multidistrict litigation (MDL 3094) has issued an order when attorneys for the plaintiffs and defendants have to submit expert reports. Judge Karen Spencer Marston gave plaintiff attorneys until November 18, 2024, to submit their reports. Defendants, including Rybelsus manufacturer Novo Nordisk, have until December 23, 2024, to submit their reports. Expert reports are written documents from expert witnesses that are used throughout different phases of the trial. These reports contain expert opinions and scientific evidence supporting those opinions.
October 4, 2024: More Plaintiffs Join the Federal Rybelsus Lawsuit In September
Comparing numbers from August to September, there was a 20% increase in lawsuits filed against GLP-1 drug manufacturers as part of the consolidated federal lawsuit. As of October 1, 2024, there were 1,090 cases pending in MDL 3094, which includes Novo Nordisk, the maker of Rybelsus. This rapid increase in case filings is the result of more Americans taking drugs like Rybelsus and experiencing severe side effects. Tens of thousands of cases are expected to be filed against GLP-1 agonist drug manufacturers, including Novo Nordisk, in the coming months.
August 2, 2024: Comprehensive Data Analysis Suggests Rybelsus May Be Associated with an Increased Risk of Blood Clots, DVT, and Pulmonary Embolisms
Trial data from an efficacy study point to an increased risk of blood clots for people taking semaglutide drugs, like Rybelsus, for type 2 diabetes. The findings were published in the Endocrine Journal. The meta-analysis signaled a 266% increase in the risk of deep vein thrombosis (DVT). This type of blood clot can cause pulmonary embolisms, which can lead to death. As more people take semaglutide drugs to treat their diabetes or achieve weight-loss goals, more people may be at risk for this potentially serious condition.
July 10, 2024: Rybelsus Lawsuit to Include Vision Loss Claims Following JAMA Study
A recent study in The Journal of the American Medical Association (JAMA) links the use of Rybelsus to a rare form of vision loss and blindness. The 2024 JAMA Ophthalmology study reported higher NAION risk among semaglutide users. The study’s authors reviewed data from 16,827 people. Novo Nordisk has not included vision loss caused by NAION as a side effect of Rybelsus or other drugs using semaglutide. We are accepting clients who have used Rybelsus and experienced vision loss or blindness.
June 17, 2024: New Judicial Orders and Developments in MDL Proceedings
The first status conference in the Rybelsus lawsuit with the new judge, Karen Marston, was held on June 10, 2024. The focus was on the process of moving the MDL forward. Since the conference, the judge has issued two orders: first, lawyers who wish to file cases into the GLP-1 MDL (MDL 3094) will not have to pay a fee to be admitted pro hac vice; second, the parties have filed a motion, seemingly with permission from the judge, in regards to privilege logs. Parties in lawsuits are required to turn over relevant information to the other side. However, sometimes there are privileges that can be asserted. If a document is not turned over due to privilege, then the document is logged and the opposing side is informed that it exists.
June 8, 2024: New Judge Appointed to Rybelsus Lawsuit, In-Person Conference Scheduled
Case Management Order number 7 has informed the parties of a new judge in the Rybelsus lawsuit. Judge Karen Spencer Marston will now preside over this litigation. We expect the Ozempic lawsuit to be very large and include many complex legal motions. Judge Marston was a top student at Wake Forest Law School, and her legal acumen will certainly be tested in this case. The judge wasted no time in moving forward with the proceedings; an in-person conference is scheduled for June 10, 2024.
June 6, 2024: Rybelsus Lawsuit Paused Until New Judge Is Appointed Following Judge Pratter’s Death
The Rybelsus lawsuit in federal court is currently paused until a new judge is appointed due to the sudden death of Judge Pratter. We anticipate that additional cases will continue to be filed in New Jersey state courts. Federal court is the suitable venue when the involved parties are from different states. Cases against Novo Nordisk by New Jersey residents will likely need to be filed in New Jersey court. It is also possible that other plaintiffs will choose to file their claims in New Jersey state court.
May 2, 2024: Rybelsus Lawsuit Progresses with Scheduled ‘Science Day’ in June 2024
April 1, 2024: Key Developments in Rybelsus Lawsuit – Leadership Motion and New Jersey Filings
There are two important developments in the Rybelsus lawsuit. First, a group of lawyers has made an application to work together as the lead attorneys. The motion filed by plaintiffs’ lawyers seems like a compromise between several law firms. There is one objection to the leadership slate. We expect the group of nearly two dozen lawyers to be approved soon. We are pleased that our litigation partner, Dan Nigh, is a proposed member of the executive committee of the federal Rybelsus lawsuit. The second development is the filing of Rybelsus cases in New Jersey state court. Some lawyers feel it is an advantage to bring their cases away from the large group. The largest group of Rybelsus state court cases will likely be in New Jersey because it is the North American headquarters of Rybelsus manufacturer Novo Nordisk.
March 23, 2024: Leadership Committee Proposed in Rybelsus Lawsuit
Plaintiff lawyers have filed an important motion in the Rybelsus Lawsuit. The motion filed March 22, 2024, asks Judge Pratter to appoint several lawyers to leadership positions in the lawsuit against Rybelsus manufacturer Novo Nordisk. It is no surprise that our litigation partner, Daniel Nigh, Esq., appears on the motion as an executive team member. Daniel is an experienced lawyer and has brought several cases against drug companies in the past, including a role as co-lead counsel for the valsartan lawsuit . Judicial appointment of lawyers to leadership roles is an important part of the case because it gives the lawyers the authority to start setting schedules for discovery and requesting documents from the manufacturer. We expect a large portion of the case to center around the marketing tactics used by Rybelsus’ manufacturer Novo Nordisk.
March 20, 2024: Rybelsus Lawsuit Status Conference Held
The first status conference in the Rybelsus lawsuit was held this week. The Judge discussed how the plaintiff’s lawyers would work together to move the cases towards a bellwether trial. We expect an order on plaintiff leadership as soon as the end of the month. The judge also requested the parties to participate in a Science Day. A science day will allow the lawyers to describe the scientific issues they expect to see in a case involving Rybelsus. Rybelsus is the same active ingredient as Ozempic and Wegovy, but different from Mounjaro. It is the only drug that is taken in pill form, which will likely be addressed at science day and throughout the lawsuit. Finally, the lawyer for Eli Lilly requested an early motion for summary judgment date. The judge stated that the motion to dismiss was a long way away. There have been a lot of questions about whether Mounjaro and Eli Lilly should be part of the case. It appears the Judge thinks they are an appropriate party.
March 13, 2024: Novo Nordisk Touts New Weight-Loss Pill
Novo Nordisk, the manufacturer of Rybelsus, has received a lot of press for a potential weight loss pill. Rybelsus was the first pill form of Semaglutide, a diabetes medicine that promotes weight loss. The drug was approved in 2019 and is typically taken in 7- or 14-milligram doses. Novo Nordisk confirmed they are testing a similar pill that would be 50 milligrams of Semaglutide and be approved for weight loss. Some indications are that higher doses lead to more severe side effects. This week, Novo Nordisk also confirmed they have completed a Phase 1 trial of Amycretin, another pill designed specifically for weight loss. The Rybelsus lawsuit alleges that Novo Nordisk has not properly warned of the dangers associated with the use of the drug. The lawsuit also alleges that extreme marketing tactics have led to an unreasonable risk of off-label use.
March 10, 2024: Key Date in Rybelsus lawsuit
This is an important week in the Rybelsus lawsuit. On Thursday, there will be the first status conference before the Honorable Gene E.K. Pratter. We expect that several plaintiff’s lawyers will ask the judge to appoint them to a leadership role in the case. The typical MDL lasts three to five years, so it is a big commitment for the lawyers. We also expect there to be millions of documents produced related to the design, testing, and adverse event reports around the drugs included in the lawsuit. The burden of proof is on the plaintiffs, and it is a large undertaking. One of the first important issues to address will be what drugs will be part of the lawsuit. We expect that, at a minimum, Rybelsus, Wegovy, and Ozempic will be included because they have the same active ingredient (semaglutide), which is patented by Novo Nordisk.
March 2024: There are currently 58 lawsuits filed against Novo Nordisk, the manufacturer of Ozempic, Rybelsus, and Wegovy.
February 2024: The U.S. Judicial Panel on Multidistrict Litigation ordered all federal Ozempic lawsuits to be centralized in the Eastern District of Pennsylvania. Ozempic is an injectable semaglutide manufactured by the same company as Rybelsus.
September 2023: The U.S. Food and Drug Administration requires Novo Nordisk to add warnings to Ozempic that the semaglutide may cause serious gastrointestinal issues such as intestinal blockage and ileus.
January 2023: The U.S. Food and Drug Administration approves a label update for Rybelsus tablets. The update allows Rybelsus to be used as a first-line treatment for type 2 diabetes, removing a previous requirement that it not be the initial therapy for patients with the condition.
An important note: Rybelsus remains FDA-approved for the treatment of type 2 diabetes. The lawsuits do not mean the drug has been recalled or deemed unsafe by regulators. Instead, plaintiffs allege that certain risks were not adequately disclosed.
On this page:
Rybelsus Side Effects and Symptoms
Rybelsus FDA-Approval and Its Black Box Warning
Manufacturer of Rybelsus – Novo Nordisk
Rybelsus and Its Ingredient Breakdown
Eligibility for Filing a Rybelsus Lawsuit
Statute of Limitations for Filing a Rybelsus Lawsuit
How to File a Rybelsus Lawsuit
What Is Rybelsus?
Rybelsus was the first oral glucagon-like peptide (GLP-1) receptor agonist treatment approved in the United States. It is a semaglutide like Ozempic and Wegovy, but it does not need to be injected. The drug was approved by the U.S. Food and Drug Administration (FDA) on September 20, 2019, to help treat type 2 diabetes in adults. Rybelsus is typically started at 3 mg daily for 30 days and is then increased to 7 mg. It may be increased to 14 mg if needed.
The drug, manufactured by Novo Nordisk, helps to improve blood sugar levels and has been known to promote weight loss by regulating appetite and slowing digestion. It mimics the GLP-1 hormone causing a delay in gastric emptying, resulting in patients feeling fuller longer.
Some scientific reports have emerged showing a possible association between the drug and serious side effects such as gastroparesis and bowel obstruction. Lawsuits allege that patients were not properly warned about potential serious adverse reactions associated with Rybelsus. Accusations against the manufacturer include that they knew of the increased risk for adverse health issues, especially with long-term use, and failed to adequately warn consumers.

How Does Rybelsus Work?
Rybelsus works as a glucagon-like peptide (GLP-1) receptor agonist. It mimics a hormone that regulates a person’s blood sugar and their appetite. The drug enhances insulin production after a meal and reduces glucagon secretion, which lessens glucose production by the liver. Patients report a longer feeling of fullness since the oral tablet helps to slow digestion.
While the primary use of Rybelsus is to treat type 2 diabetes by preventing the liver from making too much sugar and helping the pancreas to produce more insulin, many people use it to help promote weight loss. The drug signals the brain to regulate appetite, which can lead to a reduction in calorie intake.
It can have a significant impact on the digestive system since it delays gastric emptying, which may affect the absorption of other oral medications.
Rybelsus Side Effects and Symptoms
Reports have emerged, alleging severe side effects after the use of Rybelsus and other semaglutide drugs. Lawsuits allege that the manufacturer of Rybelsus, Novo Nordisk, knew of these side effects and failed to inform consumers. Side effects associated with use of the drug can cause long-term health complications that can have a personal and financial impact on patients.
Stated precautions and adverse reactions associated with the use of Rybelsus:
- Nausea
- Abdominal pain
- Diarrhea
- Decreased appetite
- Vomiting
- Constipation
- Acute Pancreatitis
- Diabetic retinopathy complications
- Hypoglycemia (with concomitant use of insulin secretagogues or Insulin)
- Acute kidney injury due to volume depletion
- Severe gastrointestinal adverse reaction
- Hypersensitivity reactions
- Acute gallbladder disease
- Pulmonary aspiration during general anesthesia or deep sedation:
Individuals who suffered these or other side effects after using Rybelsus may be able to take legal action against the manufacturer for a failure to adequately warn consumers of the adverse effects of using the drug. It is believed that there is an increased risk of severe medical conditions associated with the long-term use of Rybelsus.
Most Common Side Effects of Rybelsus
While there are wide-ranging reports of dangerous side effects linked to the use of Rybelsus, some health risks are more common than others.
Common side effects associated with Rybelsus:
- Constipation and diarrhea
- Stomach or abdominal pain
- Nausea and vomiting
- Decreased appetite
- Heartburn
- Gas and bloating
- Flu-like symptoms
- Gallbladder issues
When side effects are undisclosed or patients are not made aware of the potential for adverse health complications, doctors and consumers are unable to make informed decisions.
Blood Clots/DVT and Rybelsus
Some research has suggested that Rybelsus may put people at an increased risk of developing a specific type of blood clot, called deep vein thrombosis (DVT). However, this possible association is still being researched. This possible side effect was highlighted by an analysis of data from the PIONEER and SUSTAIN efficacy trials. The findings were published in the Endocrine Journal.
The comprehensive analysis found a 266% increase in DVT for people taking semaglutide drugs to treat their type 2 diabetes. DVT is a condition where a blood clot forms in a deep vein within the body, often in a leg. These clots can lead to serious complications.
Pulmonary Embolisms and Rybelsus
Rybelsus may allegedly be associated with a higher risk of developing a DVT, and these clots can lead to a serious event called a pulmonary embolism (PE). A pulmonary embolism occurs when a clot breaks free and travels to the lungs. Once the clot lodges in the blood vessels of the lungs, it can cause lung damage or block circulation, which can lead to death. PE contributes to more than 100,000 deaths each year.
As more people take semaglutide drugs, like Rybelsus, to manage their diabetes, increasing numbers of people may be at risk for this serious complication.
Vision Problems and Rybelsus
A recent medical study suggested semaglutide drugs, like Rybelsus, may cause a specific type of vision loss. These vision problems are caused by non-arteritic ischemic optic neuropathy (NAION). NAION occurs when the optic nerve does not get enough blood flow. This can cause an eye stroke and vision problems. These vision issues are likely permanent and untreatable.
The July 2024 study was published in JAMA Ophthalmology and examined people taking semaglutide drugs for diabetes treatment or weight loss. The study found people taking semaglutide drugs for diabetes were 4.28 times more likely to develop NAION.
People who take Rybelsus may be at an increased risk of vision problems due to the drug. If you or someone you love took Rybelsus and experienced vision loss, please contact our team to discuss your case and possible compensation.
Rybelsus FDA-Approval and Its Black Box Warning
On September 20, 2019, the FDA approved Rybelsus as the first oral GLP-1 treatment for type 2 diabetes. Prior to that date, only injectable GLP-1 treatments (such as Ozempic) had been approved. While it is currently used off-label for weight loss, it is not approved for this use through the FDA.
In January 2023, Novo Nordisk announced that the FDA had approved a label update for the drug. The updated label allows Rybelsus to be used as a first-line treatment option for adults with type 2 diabetes. Prior to this date, it was not approved for use as the initial therapy for patients with the condition.
Rybelsus Black Box Warning
Due to the potential of serious adverse reactions when using the drug, Rybelsus is required to carry a black box warning. Black box warnings are required by the FDA when there is a serious risk of harm to patients.
As indicated by the label, studies on lab animals showed an increased risk in the development of thyroid cancer. Patients with a family history of medullary thyroid carcinoma (MTC) or MEN 2 are at higher risk of thyroid C-cell tumors and should not use Rybelsus.
Manufacturer of Rybelsus – Novo Nordisk
Novo Nordisk is the manufacturer of Rybelsus, Ozempic, and Wegovy. Semaglutide, the generic name for the drug, was tested in clinical trials from 2016 to 2017, prior to approval by the FDA to treat type 2 diabetes in adults. Ozempic, an injectable semaglutide, was approved in 2017, while Rybelsus, an oral version of the drug was approved in 2019.
Wegovy, an injectable semaglutide, is the only drug of the three approved by the FDA for weight loss. Rybelsus is in clinical trials to determine if it is a safe and effective weight loss treatment. All three drugs have faced scrutiny after numerous patients reported serious side effects with their use. Nationwide lawsuits allege that Novo Nordisk knew of the gastrointestinal risks associated with the use of semaglutide and failed to warn consumers.
Rybelsus and Its Ingredient Breakdown
The active ingredient in Rybelsus is semaglutide (either 3 mg, 7 mg, or 14 mg), produced using DNA technology. However, there are a number of inactive ingredients in the oral tablet that are kept in pill form and stored in a bottle.
Inactive ingredients in Rybelsus:
- Magnesium stearate
- Microcrystalline cellulose
- Povidone
- Salcaprozate sodium
The active ingredient in Rybelsus has been linked to a number of severe health risks. Individuals who have experienced adverse health conditions after using Rybelsus or another semaglutide are strongly encouraged to consult with a legal representative.
Semaglutide Usage Linked to Severe Health Risks
There have been several serious health risks linked to the use of semaglutide, the generic name for Rybelsus. These health risks include stomach paralysis and bowel obstruction. Semaglutide is used to regulate blood sugar levels and increase insulin secretion; however, it may also provide a longer feeling of fullness, aiding in weight loss. Unfortunately, the drug is usually prescribed in a higher dose when used for weight loss, possibly increasing the risk that a patient will develop severe side effects such as gastroparesis, malnutrition-related disorders, deep vein thrombosis, and bowel obstruction.
Eligibility for Filing a Rybelsus Lawsuit
In order to file a Rybelsus lawsuit, you must first meet certain criteria, including proof of use of the drug. Working with an attorney can help you determine whether you may qualify for damages.
Eligibility criteria for a Rybelsus lawsuit with King Law:
- You must be able to show that you were actively using Rybelsus
- You suffered adverse health conditions, including ileus, gastroparesis, pancreatitis, DVT, PE, or another serious condition.
- You have evidence from a doctor or gastroenterologist about injuries you experienced after taking Rybelsus.
- You required medical intervention as a result of using the drug, such as hospitalization, emergency room visits, or a consultation with a gastroenterologist, ophthalmologist, or other medical professional.
The best way to determine if you meet the criteria for filing a Rybelsus lawsuit is to contact an attorney. It is important to note that you only have a limited amount of time to take legal action.

Evidence to Collect Before Filing Your Lawsuit:
If you have experienced significant health problems after taking Rybelsus, you may qualify for a personal injury lawsuit. Eligibility may depend on a number of factors, including your ability to prove how long you were taking Rybelsus and the injuries you experienced.
Evidence that may strengthen your Rybelsus lawsuit:
- Medical records
- Proof of prescription
- Emergency room visits and hospitalizations
- Doctor’s notes
- Documentation of missed work
The more evidence you can collect, the stronger your case will be. It is important to consult an attorney as early in the process as possible to determine your legal options.
Statute of Limitations for Filing a Rybelsus Lawsuit
All personal injury lawsuits are restricted by a statute of limitations, or legal time limit. Statutes of limitations are state-specific. An attorney can help determine if there are any exclusions or exceptions to the statute of limitations that may apply to your case. In many instances, the statute of limitations ranges between one and three years.
How to File a Rybelsus Lawsuit
You will need to follow several steps before filing a Rybelsus lawsuit. A lawyer can help you navigate the process and ensure you receive the best possible outcome in your case.
Steps for filing a Rybelsus lawsuit:
- Determine eligibility: In order to determine eligibility, you should contact a lawyer for a free case review. During the initial consultation, an attorney will help determine if you have a valid claim for damages.
- Gather evidence: The second step in a Rybelsus lawsuit is evidence gathering. In order to strengthen your case, you will need to collect evidence such as medical records, proof of out-of-pocket expenses, prescription documents, and expert testimonies.
- Filing the lawsuit: An attorney can aid you in filing the lawsuit in the correct jurisdiction and within the statute of limitations.
- Negotiations: Your attorney may enter into negotiations with the defendant to secure a favorable settlement in your case. If a settlement cannot be reached, your attorney may set the case for trial to obtain the outcome you deserve.
Throughout the proceedings, it is strongly recommended that you work with a qualified Rybelsus lawsuit attorney.
Rybelsus Settlement Amounts
There is not a guaranteed settlement amount in a Rybelsus lawsuit. The amount will vary greatly depending on the case, and some patients may receive large settlements while others do not receive a settlement. Since there is no standardized settlement amount, the value of your case will be determined by your individual damages.
Damages generally reflect the extent of your losses and the severity of your injuries. An attorney can help ensure that you receive the maximum compensation allowed by state or federal law.

Contact a Rybelsus Lawyer to File a Lawsuit
If you have suffered injuries related to your use of Rybelsus, it is strongly recommended that you contact a qualified attorney. At King Law, our legal team has decades of combined experience handling prescription drug claims. We are well-versed in handling complex lawsuits and will help you secure the compensation you deserve. Contact our office today to schedule a free, no-obligation consultation.
Frequently Asked Questions (FAQs)
Find answers to any major questions you have regarding eligibility in the Rybelsus lawsuits below.

