Complete the form below to see if you qualify for a Cartiva Toe Implant Lawsuit

Cartiva’s synthetic cartilage implant (SCI) was a big-toe implant that has been linked to serious complications and was recalled in October of 2024. Now, patients are filing lawsuits against Cartiva, alleging device defects caused their injuries.
Some Cartiva patients say they experienced issues, including implant failure, nerve damage, pain, and loss of motion. Some patients have had to have surgery to correct or remove the implant or a surgery to fuse their big toe joint (arthrodesis). Cartiva implant lawsuits are being filed in state courts and as part of a federal centralized litigation (MDL No. 3172).
This page provides information about the October 2024 recall of the Cartiva implant, including signs of implant failure and complications. This article also explores failure rates of Cartiva synthetic cartilage implants and who may qualify for compensation from a Cartiva revision surgery claim.
Key Information in the Cartiva Toe Implant Litigation
- Summary of Cartiva implant lawsuits: Lawsuits allege Cartiva SCIs were defective and caused persistent pain, limited range of motion, and implant failure, sometimes requiring removal or fusion surgery.
- Device used to treat: Osteoarthritis of the big toe and other joint disorders
- Product recall date: October 31, 2024
- FDA recall posting date: December 4, 2024
- Name of litigation: In Re: Cartiva Synthetic Cartilage Implant Products Liability Litigation
- MDL #: 3172
- Many claims involve: Implant removal/revision or fusion surgery
- Name of defendant: Cartiva, Inc.
- Core allegations: Product Liability; Negligent Design, Manufacture and/or Distribution; Misbranded and Adulterated Device; Common Law Product Liability and Negligence; Breach of Express Warranty; and Breach of Implied Warranty
- Name of venue: Eastern District of Arkansas (Federal Court)
- When the Cartiva MDL was created: February 5, 2026
- Judge assigned to MDL: The Honorable Kristine G. Baker
About the Cartiva Toe Implant Lawsuit:
Cartiva Implant Lawsuit Updates and Case Developments (2026)
What Is the Cartiva Synthetic Cartilage Implant (SCI)?
Why Are People Filing Cartiva Toe Implant Lawsuits?
Has the Cartiva Implant Litigation Been Consolidated Into an MDL?
Is the Cartiva Implant Lawsuit a Class Action?
Was There a Cartiva Implant Recall or Market Withdrawal?
What Are the Most Common Signs of Cartiva Implant Failure?
What Does Real-World Evidence Say About Cartiva Failure Rates?
Who Qualifies to File a Cartiva Implant Lawsuit?
What Proof Is Needed to File a Cartiva Implant Claim?
How Can I File a Cartiva Toe Implant Lawsuit?
How Long Does a Cartiva Implant Lawsuit Take?
Is There a Deadline to File a Cartiva Implant Lawsuit?
Estimated Cartiva Implant Lawsuit Settlement Amounts
King Law Is Investigating Cartiva Toe Implant Claims
Contact a Cartiva Implant Lawyer Today
Cartiva Implant Lawsuit Updates and Case Developments (2026)
Some patients with Cartiva synthetic cartilage implants have experienced complications and implant failures. According to legal complaints, some Cartiva patients claim they had toes amputated, the implant removed, or their big toe joint fused because of implant-related complications. On February 5, 2026, the Joint Panel on Multidistrict Litigation (JPML) created a centralized litigation to consolidate and streamline legal proceedings in Cartiva implant lawsuits.
March 2, 2026: Plaintiffs Join Newly Created Litigation for Cartiva Toe Implant Injuries
As of March 2, 2026, there are 14 pending lawsuits in the Cartiva Toe Implant MDL (MDL -3172 IN RE: Cartiva Synthetic Cartilage Implant Products Liability Litigation). This newly created MDL is a centralized federal litigation for people alleging injuries from malfunctioning Cartiva toe implants. People who had injuries or revision surgeries to address Cartiva toe implants can talk to a lawyer to see if they qualify to join this litigation.
February 5, 2026: Cartiva Lawsuits Consolidated into Federal MDL in Arkansas
The Judicial Panel on Multidistrict Litigation has approved a request filed by Cartiva patients to centralize the lawsuits against Cartiva’s manufacturer. The MDL format may allow the participating Cartiva patients to pool resources and streamline the legal process of their claims. The new MDL is called IN RE: CARTIVA SYNTHETIC CARTILAGE IMPLANT PRODUCTS LIABILITY LITIGATION, and it is MDL No. 3172. Cases are consolidated in the Eastern District of Arkansas and will be overseen by Judge Kristine G. Baker. Cartiva patients who experienced complications may qualify to file a legal claim and join the active MDL.
January 23, 2026: Maryland Woman Files Lawsuit After Cartiva Implant Fails
A woman in Maryland is suing Cartiva after her toe implant failed shortly after being implanted. She says she had her Cartiva implant removed because it failed “almost immediately” after it was implanted, causing her to experience severe pain, swelling, and mobility issues. She believes the device had a higher-than-expected failure rate and claims Cartiva should have warned her.
January 22, 2026: PA Woman Sues Cartiva Over Toe Implant Complications
A patient in Pennsylvania has filed a lawsuit against Cartiva, alleging that her Cartiva big toe implant failed and caused her to experience significant pain. She says the Cartiva implant failure caused her to have to undergo surgery to remove the implant. In her complaint, she questions the study that Cartiva presented to the FDA when it requested approval of the big toe implant. The results of the Motion Study suggested that Cartiva performed better than big toe fusions. However, according to the legal complaint, other researchers who have studied this have not gotten the same results as Cartiva.
November 5, 2025: Man in Pennsylvania Sues Cartiva After Implant Fails
A Pennsylvania man has sued Cartiva after he had a big toe implant installed, which he had hoped would relieve his symptoms. According to his complaint, the device caused him to experience complications and was removed about a year after implantation. After that surgery, the man suffered from a severe bone infection that resulted in his second and third toes being amputated. He believes Cartiva was aware of the failure rate of its device and should have informed patients.
October 31, 2024: Cartiva Implants Recalled Over Concerns about High Failure Rate for Device
The Cartiva synthetic cartilage implant is recalled from the market after reports of higher-than-expected rates of toe implant failure. The complications experienced by patients included removal, nerve damage, pain, and the implant sinking (called subsidence), moving out of place, or breaking. The Cartiva recall is global, meaning the recalled Cartiva devices have been sold in multiple countries. The Cartiva devices being recalled are CAR-06-US (6mm), CAR-08-US (8mm), CAR-10-US (10mm), and CAR-12-US (12mm). Before the FDA announced the recall, Stryker issued the recall on behalf of Cartiva, Inc., which was owned by Wright Medical Group NV. Stryker acquired Wright Medical Group NV in 2020.
June 2021: Study Published in Foot and Ankle Surgery Finds that Cartiva Implants Accounted for 23% of FDA Adverse Events for MTPJ Implants
Using the FDA’s adverse event reports (MAUDE) from 2010 through 2018, researchers found that 23.4% of the FDA’s adverse event reports for big toe joint implants involved Cartiva. The study found that component loosening and infection were the most common adverse events reported for hallux MTPJ implants.
What Is the Cartiva Synthetic Cartilage Implant (SCI)?
The Cartiva synthetic cartilage implant is a device implanted in someone’s foot to
Why Are People Filing Cartiva Toe Implant Lawsuits?
People are filing lawsuits against Cartiva because they experienced severe complications after having the big toe implant installed. The allegations in lawsuits against the Cartiva synthetic cartilage implant (SCI) include the following:
- Failing to disclose failure rates and risks of the Cartiva big toe implant
- Designing a defective big toe implant
- Misleading patients and doctors about the safety of the Cartiva big toe implant
Patients also allege that Cartiva’s defective design caused them to experience painful, costly, and disabling outcomes. The device was recalled in 2024 due to higher-than-expected rates of adverse events. The outcomes patients have mentioned in legal complaints include nerve damage, new or worsening pain, their toes sinking into the implant, and having to have the implant removed.
Cartiva Implant Recall Overview
Cartiva SCI implants were
Can a Cartiva Implant Failure Lead to a Lawsuit?
Not everyone who has issues with their Cartiva SCI implants will qualify for or choose to file a lawsuit. Currently, King Law is reviewing claims from people who had revision surgeries to correct issues with their Cartiva toe implants. A revision surgery is done to repair or remove a faulty device.
Does Implant Subsidence or Fusion Surgery Qualify Me for a Cartiva Lawsuit?
If you experience Cartiva subsidence (sinking of the implant) or a surgery to remove the Cartiva implant and fuse your bones, you may qualify for a lawsuit against Cartiva. Contact our firm to learn more about eligibility and possible compensation.
Has the Cartiva Implant Litigation Been Consolidated Into an MDL?
The Cartiva implant litigation has been consolidated into MDL 3172: IN RE: CARTIVA SYNTHETIC CARTILAGE IMPLANT PRODUCTS LIABILITY LITIGATION. The Cartiva lawsuits were consolidated into an MDL to help streamline the legal process for the participating cases. In an MDL, the parties collectively participate in the pretrial, discovery, trial preparation, and negotiation stages of their cases. This allows participating Cartiva patients to share resources with other Cartiva patients in the MDL.
In an MDL, each Cartiva patient’s case is still resolved individually. So, Cartiva patients who file a lawsuit in the MDL can have their own trial and negotiate an individual settlement with Cartiva. More Cartiva patients can join the MDL if they experienced complications and believe they qualify for compensation.
Is the Cartiva Implant Lawsuit a Class Action?
The Cartiva implant lawsuit is not a class action lawsuit. It is an MDL (i.e., a multidistrict litigation). In a class action lawsuit, the people filing the claim do not have individual cases, trials, or settlements. Instead, their claims would be merged into one group claim, and the resulting class-action settlement would be divided among each of the plaintiffs based on the terms of the agreement. In contrast, plaintiffs in an MDL have their cases decided on an individual basis. The Cartiva lawsuit is an MDL, so each plaintiff’s claims are separate, but they work together to coordinate some parts of their lawsuits.
Was There a Cartiva Implant Recall or Market Withdrawal?
There was a Cartiva implant recall and withdrawal from the market. On October 31, 2024, Cartiva initiated a Class II recall for the following synthetic cartilage implant (SCI) models:
- CAR-06-US (6mm)
- CAR-08-US (8mm)
- CAR-10-US (10mm)
- CAR-12-US (12mm)
The codes for these recalled Cartiva devices are CAR-06-US/00852897002328, CAR-08-US/00852897002021, CAR-10-US/ 00852897002038, and CAR-12-US/00852897002335. The Cartiva synthetic cartilage implant devices that were recalled included all lots distributed from July 2016 to October 2024. The Cartiva SCI implant recall was posted on the FDA’s website on December 4, 2024. However, Stryker issued a recall on its site on October 31, 2024.
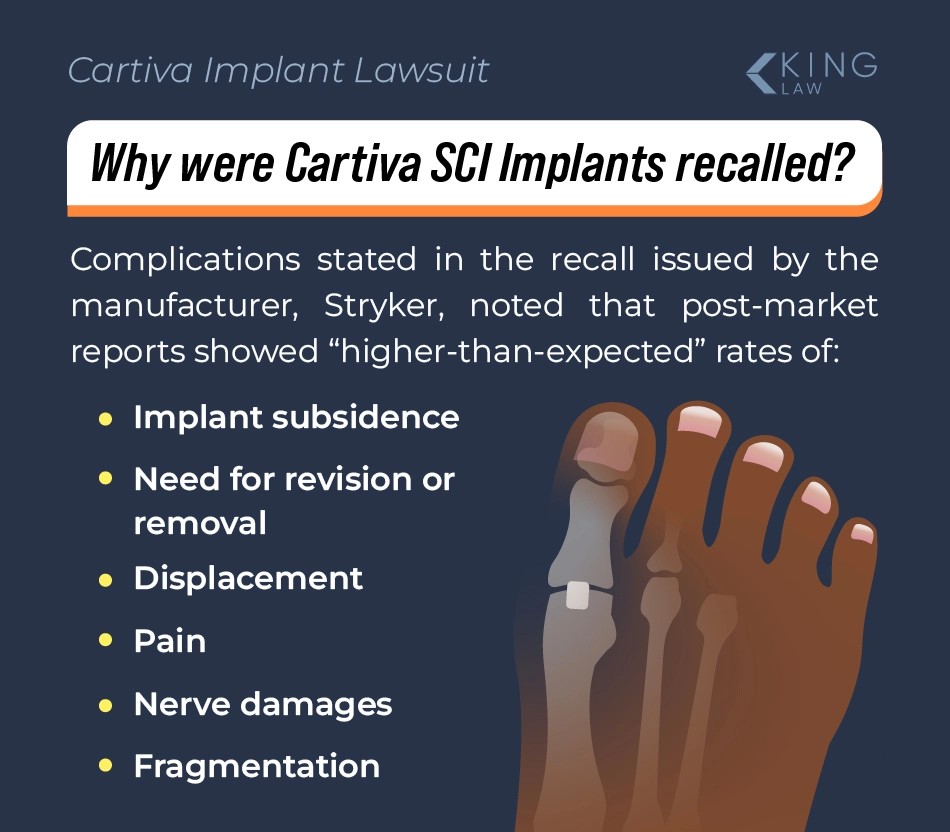
Why Was the Cartiva Implant Recalled in October 2024?
According to the FDA’s website, the Cartiva implant was recalled in October 2024 because patients were experiencing higher-than-expected rates of the following:
- Revision surgery (needing to have surgery to modify or remove the implant)
- Removal surgery (having surgery to remove the implant)
- Implant subsidence (the implant sinks or the toe sinks into the implant)
- Displacement (the implant moves out of place)
- Pain
- Nerve damage
- Fragmentation (the implant breaks or pieces of the implant break off)
The FDA’s website indicates the FDA determined that “device design” may be the cause for recalling the Cartiva implant. However, the FDA says it may change this as it learns more information about the recall.
What Are the Most Common Signs of Cartiva Implant Failure?
Not all implant failures happen immediately after the device is inserted into the body. Sometimes, device failures happen years or months after the implantation surgery. People who have Cartiva’s SCI implants should watch for symptoms of device malfunction or failure.
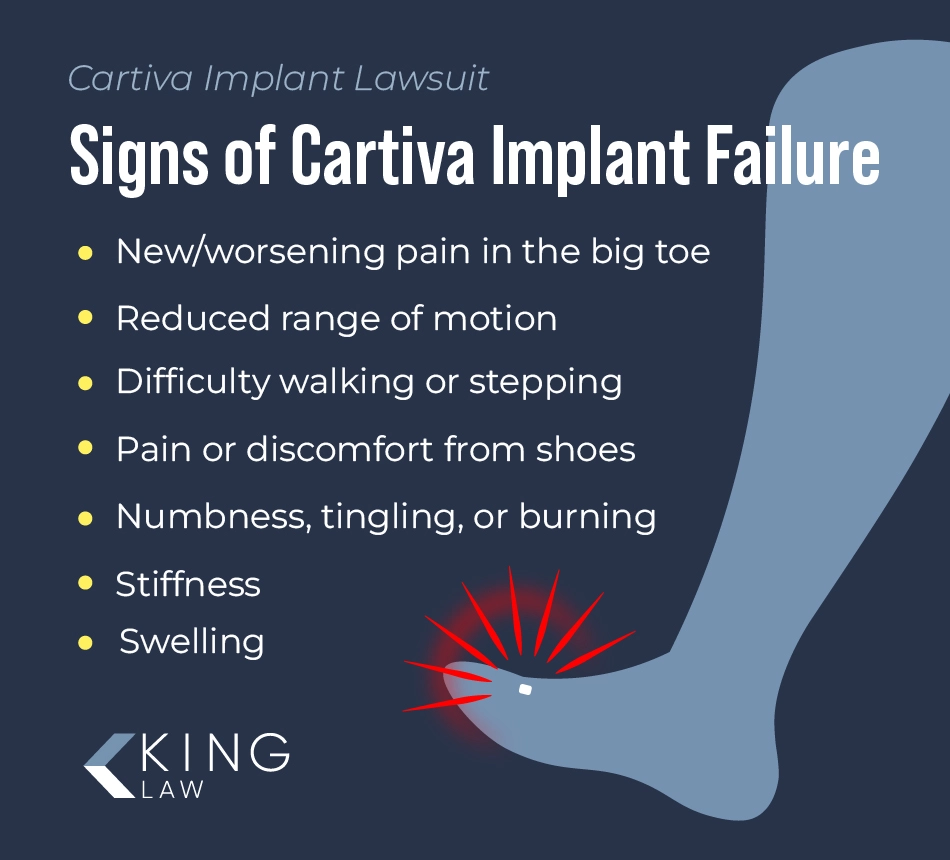
Cartiva implant failure signs reported by patients include:
- New or worsening pain in the big toe or surrounding bones
- Stiffness
- Reduced range of motion of the big toe
- Swelling
- Difficulty walking or stepping (e.g., problems pushing off with the big toe)
- Pain or discomfort when wearing shoes
- Numbness, tingling, or burning (nerve pain or irritation)
- The implant becomes displaced or loose
According to one study published in the European Journal of Orthopaedic Surgery & Traumatology, polyvinyl alcohol hydrogel implants (like the Cartiva synthetic cartilage implant) had a 27.9% complication rate and “concerning postoperative imaging findings” shortly after the person received the implant. Another post-marketing study published in the journal Foot and Ankle Surgery found that up to 38% of participants who received a Cartiva implant needed a revision surgery within 2 years. This was true when the implant was used to treat hallux rigidus (a form of arthritis).
What Are the Cartiva Implant Failure Symptoms Patients Mention Most Often?
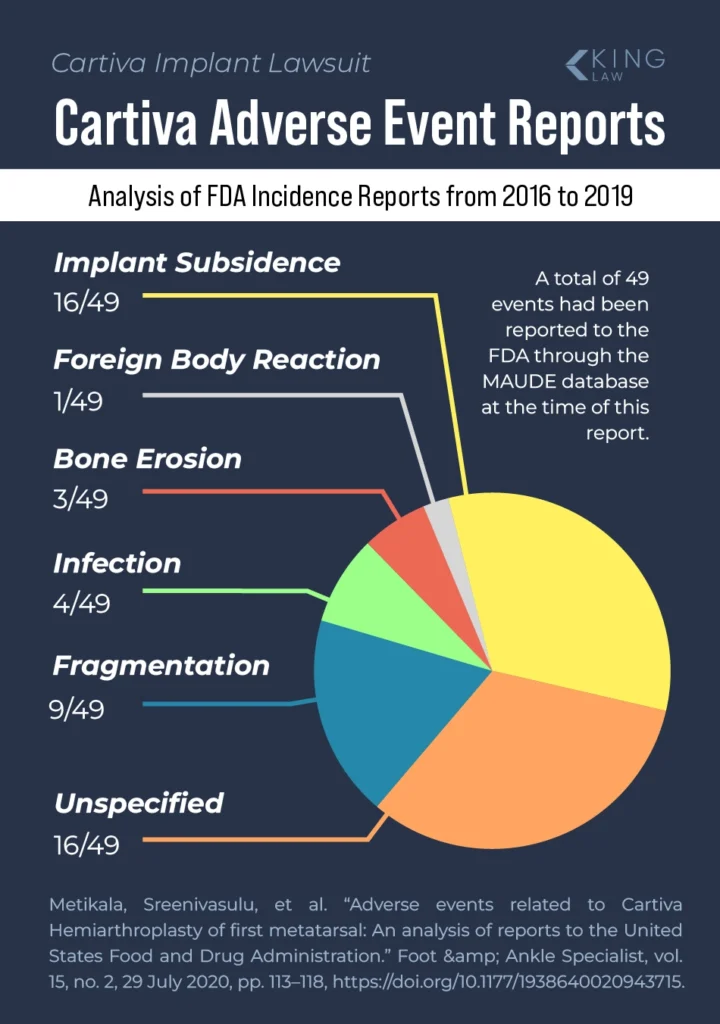
A study of the FDA’s adverse event reports for Cartiva found that, from 2016 through 2019, subsidence (the implant sinking into the bones) and “unspecified” adverse events were the most frequently reported complications, followed by fragmentation and infections. However, the researchers who conducted that study believed there were inconsistencies in how and when Cartiva-related adverse events are reported. As such, they said the dataset they used may not reflect the true number or type of Cartiva-related adverse events that patients experienced during that time.
In another study of 22 Cartiva patients, at least half experienced pain, stiffness, and limited range of motion within two years after having the surgery done.
How Does a Cartiva Implant Fail?
According to FDA reports and lawsuits, Cartiva implants may fail for a number of reasons, including:
- The implant may sink into the surrounding bone (called implant subsidence).
- The place where the bone and implant are in contact with each other may become loose or unstable.
- The implant may become displaced.
- Arthritis may form around the joint.
- Inflammation around the implant can occur.
These Cartiva implant complications may lead the surgeon to recommend another surgery to remove or revise (fix or adjust) the implant.
In its 2024 recall, Stryker advised physicians to monitor Cartiva patients for the following symptoms, which could indicate device malfunction:
- New or worsening symptoms of pain
- Difficulty walking
- Skin reactions
- Stiffness
- Swelling
- Weakness of the big toe joint
Why Does the Cartiva Implant “Sink” (Subsidence)?
Subsidence is when the Cartiva synthetic cartilage implant sinks into the bones of the big toe joint. Studies have suggested that subsidence is one of the leading complaints made by Cartiva patients. When implant subsidence occurs, it can cause pain, discomfort, and reduced range of motion. Some Cartiva patients who have experienced subsidence have had another surgery to revise or remove the implant.
Can the Cartiva Implant Cause Nerve Damage?
Nerve damage is one of a number of potential risks listed in the October 2024 Cartiva recall notice. Studies have also shown that some Cartiva patients still experience pain after they have the implant done. Cartiva patients have also reported experiencing neurovascular symptoms after receiving the implant. Some symptoms that Cartiva patients may experience include burning, numbness, tingling, or pain.
Can the Cartiva Implant Break, Fragment, or Displace?
Post-market studies have suggested that Cartiva implants can fragment, displace, break, or require revision or removal. For example, one study of FDA adverse reporting data from 2016 to 2019 found that fragmentation and bone erosion were among the complications reported involving Cartiva. Another study found that Cartiva patients in the study experienced worsening arthritis around the implant and a mismatch between the Cartiva implant size and the joint space.
What Does Real-World Evidence Say About Cartiva Failure Rates?
Real-world evidence discovered after Cartiva entered the market suggests that Cartiva implants may not provide better patient outcomes than arthrodesis. Arthrodesis involves fusing the joint in the big toe. When it was approved by the FDA in 2016, Cartiva was marketed as a non-inferior alternative to arthrodesis to improve pain and range of motion in patients with hallux rigidus. However, some post-marketing studies suggest that Cartiva has a higher-than-expected failure rate compared to arthrodesis. Some imaging studies also suggest that Cartiva patients may experience worsening arthritis or bone and joint changes after having the implant done.
Who Qualifies to File a Cartiva Implant Lawsuit?
To be eligible to file a Cartiva implant lawsuit with King Law, you should have:
- Had a Cartiva SCI device implanted between 2016 and 2024
- Had revision surgery due to subsidence (shrinkage), loosening, erosion and/or wear of the device
The above criteria are some of the eligibility requirements that our law firm has developed for our purposes. However, these may change, and the list does not include all complications we are investigating. You may still qualify to join the MDL even if your situation does not meet our firm’s eligibility criteria. Please contact our office to determine your eligibility.
What Proof Is Needed to File a Cartiva Implant Claim?
Some of the proof you can use to file a Cartiva implant claim includes the following:
- Medical records showing your doctor recommended you receive a Cartiva synthetic cartilage implant
- Documentation showing you received the implant (e.g., surgical records)
- Evidence of your Cartiva SCI device number
- Post-op notes showing that you experienced implant-related symptoms
- Imaging studies and diagnostic tests
- Evidence that you had surgery to remove or adjust the Cartiva implant
Your attorney can help you identify and gather records needed to support your potential Cartiva SCI claim.
What Medical Records Should I Request to Support My Cartiva Claim?
Some of the medical records that you or your attorney should request first include the original implant surgery report, post-op follow-up appointment records, and pre- and post-op imaging studies (e.g., radiology reports). You or your attorney should also locate surgical and medical records of procedures or treatments done to correct or remove the Cartiva synthetic cartilage implant. These records will help you tell the story about how you received the Cartiva implant and how it injured you.
How Can I File a Cartiva Toe Implant Lawsuit?
Filing a Cartiva toe implant lawsuit involves the following steps:
- Speaking to a product liability attorney during a free, confidential consultation.
- Gathering your records to prove that you received the Cartiva synthetic cartilage implant and believe you were harmed by it.
- Working with your lawyer to help them file your lawsuit against Cartiva, likely by joining the MDL.
- Your lawyer participates in settlement negotiations or takes your Cartiva synthetic cartilage implant case to trial.
Hiring a lawyer to help you through the legal process of filing a Cartiva claim can help you understand each step in the process. A qualified attorney can help you understand and navigate the legal steps involved in fighting for compensation. Most attorneys who handle Cartiva implant claims do not charge upfront legal fees to take on these cases. At King Law, we only take payment if we secure compensation for you.
How Long Does a Cartiva Implant Lawsuit Take?
How long a Cartiva implant lawsuit takes depends on many factors, such as the complexity of the person’s claim and whether they proceed with an individual claim or as part of the MDL. Some Cartiva synthetic cartilage implant cases may take up to a year or more to reach a resolution. The MDL was created in February of 2026, and MDLs often take multiple years to resolve.
Is There a Deadline to File a Cartiva Implant Lawsuit?
Yes. There is a deadline to file a Cartiva implant lawsuit. The deadline to file the lawsuit depends on the type of Cartiva claim you are bringing and which jurisdiction your claim is brought in. Each state has rules about when legal claims need to be filed, and it is important for you to understand and meet these deadlines. An attorney can help you determine what your filing deadline is for your Cartiva implant lawsuit.
Estimated Cartiva Implant Lawsuit Settlement Amounts
At this time, it is difficult to estimate how much each person will receive if they file a successful case as part of the Cartiva MDL. The payout range for Cartiva implant lawsuit settlements depends on the person’s individualized losses. Factors that can affect the final settlement value include the severity of their Cartiva-related losses, whether they can fully heal from the complications, and how their quality of life has been impacted. Types of compensation they might receive include money for medical expenses, physical therapy, pain and suffering, and lost income.
King Law Is Investigating Cartiva Toe Implant Claims
King Law is actively investigating Cartiva implant failure lawsuits. We are accepting Cartiva implant cases, including those involving implant failure or revision or removal surgery. We have vast experience handling complex medical device and product liability lawsuits. We take Cartiva synthetic cartilage implant cases on a contingency basis, meaning clients do not pay us upfront legal fees to represent them. Instead, we receive an agreed-upon percentage of the compensation package we win for them.
Contact a Cartiva Implant Lawyer Today
Call King Law today by dialing (585) 496-2648 to schedule a free consultation with a Cartiva implant lawyer. You can also contact us online. Our legal team provides no-cost Cartiva case reviews, and we have a wealth of resources to help us investigate and file your potential case.
Frequently Asked Questions (FAQs)
The Cartiva toe implant lawsuit claims the Cartiva synthetic cartilage implant may be defective and that Cartiva did not warn consumers about the risks.
Individuals are filing Cartiva implant lawsuits because they experienced new or worsening pain, bone loss, implant failure, nerve injury, and other complications, resulting in a revision surgery after getting the Cartiva synthetic cartilage implant.
Yes, there are lawsuits about the Cartiva SCI implants, which were recalled in 2024.
Cartiva subsidence happens when the implant sinks into the metatarsal bone, typically causing severe pain, stiffness, and device failure.
If you had to have fusion surgery after having a Cartiva toe implant removed, you may be eligible to file a lawsuit or join the MDL.
If you had surgery to remove or correct your Cartiva SCI implant, you may qualify for a lawsuit.
There is an MDL against Cartiva for toe implants, which is different from a class action.
Some of the reasons for the Cartiva implant recall in October 2024 included higher-than-expected rates of nerve damage, removal surgeries, revision operations, implant subsidence (sinking), displacement, and fragmentation.
According to the FDA’s website, the Cartiva toe implant was recalled due to a possible device defect and higher-than-expected complication rates.
Some common problems with Cartiva toe implants include pain and discomfort, not being able to wear shoes (especially heeled shoes), bone loss, fragmentation of the implant, and the implant moving out of place.
The failure rates of Cartiva toe implants vary. Some studies have suggested that Cartiva implant failure rates are low, while others suggest a much higher failure rate.
The evidence needed to file a Cartiva toe implant lawsuit includes records to prove that you received the Cartiva toe implant and experienced complications.
The length of time you have to file a Cartiva toe implant lawsuit depends on the type of case you are filing and what the applicable state filing deadlines are.
Compensation that may be available in a Cartiva implant lawsuit includes medical expenses, lost income, and pain and suffering.
If you receive a settlement, it will reflect your individualized losses and circumstances involving the Cartiva implant.
There is no set timeframe for settling a toe implant lawsuit with Cartiva. Each case moves at its own pace, depending on the complexity and strength of the person’s case, along with other factors.
Many people find hiring a lawyer to file a Cartiva toe implant lawsuit beneficial.
It depends. Many Cartiva implant lawyers do not charge upfront legal fees to handle cases involving implant complications or failure. Instead, they take a portion of your compensation, if you receive such.
List of Sources Used in this Article
Baumhauer, Judith F., et al. Prospective, Randomized, Multi-centered Clinical Trial Assessing Safety and Efficacy of a Synthetic Cartilage Implant Versus First Metatarsophalangeal Arthrodesis in Advanced Hallux Rigidus. Foot & Ankle International, 2016.
Fletcher, William R., et al. Mid-term Efficacy of the Cartiva Synthetic Cartilage Implant in Symptomatic Hallux Rigidus. Bone & Joint Open, 2024.
Judicial Panel on Multidistrict Litigation. Transfer Order – MDL No. 3172. U.S. Judicial Panel on Multidistrict Litigation, 2026.
“Clinical Outcomes of Synthetic Cartilage Implant for Hallux Rigidus”. ScienceDirect.
“First Metatarsophalangeal Joint Polyvinyl Alcohol Hydrogel Implant Hemiarthroplasty”. Foot & Ankle Orthopaedics.
“Orthopaedic Outcomes Study”. Archives of Orthopaedic and Trauma Surgery, 2024.
“Outcomes of Synthetic Cartilage Implant Hemiarthroplasty”. ScienceDirect.
“Patient Satisfaction Following Hallux Rigidus Treatment”. PubMed.
“Physical Function and Pain Interference Levels Following SCI Surgery”. Foot & Ankle Orthopaedics.
“Polyvinyl Alcohol Hydrogel Implant for Hallux Rigidus – Systematic Review”. European Journal of Orthopaedic Surgery & Traumatology, 2024.
“Surgical Outcomes of Synthetic Cartilage Implant Hemiarthroplasty”. PubMed.
“Synthetic Cartilage Implant Clinical Outcomes Study”. PubMed.
Stryker. Urgent Medical Device Recall – Cartiva Synthetic Cartilage Implant. Stryker Corporation, 2024.
U.S. Food and Drug Administration. Cartiva Synthetic Cartilage Implant – FDA Recall Database Entry. FDA.
U.S. Food and Drug Administration. Instructions for Use – Cartiva Synthetic Cartilage Implant. FDA, 2016.
U.S. Food and Drug Administration. Premarket Approval (PMA) – Cartiva Synthetic Cartilage Implant. FDA.
U.S. Food and Drug Administration. Summary of Safety and Effectiveness Data (SSED) – Cartiva Synthetic Cartilage Implant. FDA.

