See if You Qualify

Some people who take Ozempic have experienced severe side effects, such as stomach paralysis, ileus/intestinal blockages, gastroparesis, gallbladder issues, pancreatitis, and vision loss (NAION). This page is designed for people evaluating whether they qualify to file an Ozempic lawsuit or join one of the active federal MDLs.
Lawsuits filed against Ozempic’s manufacturer, Novo Nordisk, allege that the company did not properly warn patients or doctors about some of the serious side effects of the drug, leading to serious injuries for some people.
Enough people have claimed injuries for Ozempic that two mass litigations have been formed. These centralized litigations are called multidistrict litigations (MDLs), and they occur in federal courts. Additionally, lawsuits alleging blood clots and pulmonary embolism have been filed outside of the MDLs.
One Ozempic MDL is for people diagnosed with gastrointestinal injuries after starting Ozempic. The other MDL is for people who developed a type of vision loss called NAION (i.e., an eye stroke called non-arteritic anterior ischemic optic neuropathy). King Law is actively investigating both types of claims from people who took Ozempic and experienced these complications.
This page provides a detailed overview of the Ozempic lawsuit. It discusses what the Ozempic lawsuit is about, who can file a claim, how to file one, possible types of compensation, and the types of injuries Ozempic patients are experiencing. We also provide updates on the Ozempic lawsuit, key dates, and what medical researchers, doctors, and lawyers are saying about the Ozempic injuries.
About the Ozempic Lawsuit
News Updates for the Ozempic Litigation
King Law Featured in Ozempic Litigation News Coverage
Why Are Lawsuits Being Filed Against Ozempic?
Why Are There Two Ozempic MDLs?
Is There a Class Action Lawsuit Against Ozempic?
What Conditions May Qualify Someone for an Ozempic Lawsuit?
What Doctors and Attorneys Are Saying About Ozempic Litigation and Injuries
Ozempic Lawsuits Involving Vision Loss, Blindness, and NAION
Injuries in Ozempic Stomach Paralysis Litigation
King Law Criteria to File an Ozempic Lawsuit
What Are the Recoverable Damages in the Ozempic Lawsuit?
What Is the Deadline to File an Ozempic Lawsuit?
How Can I File an Ozempic Lawsuit?
How Much Is the Ozempic Lawsuit Going to Payout?
When Will the Ozempic Lawsuit Be Settled?
King Law Is Investigating Ozempic Claims Related to Severe Side Effects Allegedly Caused by Ozempic
Our Ozempic Legal Team at King Law
Contact an Ozempic Lawyer Today
Key Takeaways
Here’s some key information about the Ozempic lawsuits:- There are more than 3,200 claims across two MDLs filed since the litigation began in 2024 (the MDLs also include lawsuits for those injured by Wegovy, Mounjaro, and other GLP-1 drugs)
- Key legal claims against Novo Nordisk include failure to warn, negligence, negligent marketing, and product liability (for a pharmaceutical drug)
- Defendants are Novo Nordisk and Eli Lilly (which makes other GLP-1 drugs)
- There are two MDLs for the Ozempic injury claims:
- Number of pending claims in MDL 3094 as of March 2, 2026: 3,363
- Number of pending claims in MDL 3163 as of March 2, 2026: 54
- People can file individual Ozempic lawsuits or join the active MDLs
- The Honorable Karen S. Marston oversees both Ozempic MDLs
- The consolidated Ozempic lawsuits are pending in the Eastern District of Pennsylvania
- Ozempic is primarily prescribed for Type 2 diabetes management
- Some doctors prescribe Ozempic off-label for weight loss
- Trials in MDLs are scheduled for late 2026 and early 2027, but these dates may change.
- There have not been confirmed settlements yet.
If you think you qualify to file an Ozempic lawsuit, contact our team to learn and protect your legal rights.
News Updates For The Ozempic Litigation
March 9, 2026: Novo Nordisk Resolves Ozempic Knockoff Lawsuit with Competitor
Novo Nordisk has resolved a legal disagreement with U.S. telehealth company Hims & Hers Health, who Novo Nordisk says was selling unauthorized, compounded versions of Ozempic and other GLP-1 drugs. According to Hims, Novo Nordisk will sell Ozempic and one other GLP-1 on the competitor’s website for the same self-pay price on Novo Nordisk’s site. Many people taking Ozempic (a semaglutide) have suffered from severe gastrointestinal complications that they say they were not properly warned about. Some Ozempic patients have filed lawsuits over this, and many of these claims have been consolidated into a federal MDL.
March 5, 2026: FDA Issues Warning Letter to Novo Nordisk About Reporting Adverse Events for its GLP-1s
The FDA’s Center for Drug Evaluation and Research (CDER) has issued a warning letter to Novo Nordisk, Ozempic’s maker. The letter cites “objectionable conditions” regarding the company’s mandated reporting of adverse events for its GLP-1 drugs. Additionally, the FDA says Novo Nordisk “failed to develop written procedures” for reporting postmarketing adverse drug experiences (ADEs). The report cites two specific reporting infractions, one for a patient who experienced a stroke and another for a patient who committed suicide. Although Novo Nordisk has replied to FDA communications about these citations, the FDA says its replies fail to” determine whether your actions will effectively prevent similar violations in the future.” Novo Nordisk has 15 business days to provide the FDA with written communication regarding actions taken to prevent future reporting infractions.
February 24, 2026: Novo Nordisk Announces Plans to Cut Ozempic List Price by 35%, Personal Injury Lawsuits Continue
Novo Nordisk has said it intends to reduce Ozempic’s list price, starting January 1, 2027. According to Reuter’s, the U.S. list price for Ozempic will decrease by 35%. Ozempic can be purchased through self-pay and direct-to-consumer websites. The prices on those websites are much less than the current list price. The list price is the base cost of the medication without discounts, insurance contributions, and other services. Lowering Ozempic’s list price may help more people access the medication. However, some lawsuits claim that Ozempic has caused people to suffer painful, disabling, and life-threatening gastrointestinal issues. Some patients believe that Ozempic’s manufacturer, Novo Nordisk, should have told consumers more about the serious gastrointestinal risks of taking the medication.
February 2, 2026: Gastroparesis is Most Common Injury in Lawsuits Against Ozempic and Other GLP-1 Drugs
According to court documents, gastroparesis is the most frequently listed injury in complaints filed as part of the GLP-1 product liability litigation (MDL -3094), which includes Ozempic claims. In a hearing on January 13, 2026, Novo Nordisk’s attorney Katie Insogna gave a breakdown of injuries listed in lawsuits in the MDL. 75% of the lawsuits listed gastroparesis/stomach paralysis in the complaint, 18% listed ileus, 8% listed gallbladder complications, and 8% listed other severe gastrointestinal conditions. Novo Nordisk maintains that Ozempic and its other GLP-1 drugs are safe, and the company is defending against these allegations. People who have experienced one of these complications after taking Ozempic can talk to an attorney about filing a lawsuit related to their injuries.
January 15, 2026: FDA Issues Warning About Counterfeit Ozempic Found in U.S. Drug Supply Chain
Some Ozempic users may be using counterfeit Ozempic without knowing it, according to a December warning letter from the FDA. Consumers and pharmacies can identify the counterfeit drugs by products labeled with the lot number PAR1229. Citing safety concerns, the FDA and Novo Nordisk warn patients not to take Ozempic labeled with that lot number. This warning comes as more lawsuits are being filed over side effects linked to Ozempic. As of January 5, 2026, there are 3,063 active lawsuits against Novo Nordisk and other GLP-1 drug manufacturers, according to court documents.
January 2, 2026: Ozempic Hits Indian Market, Starting at Less Than $25 Per Week for Lowest Dose
As of December 20225, Novo Nordisk’s Ozempic is now available in India and will be sold as injectable pens. Doses will range from 0.25 mg to 1 mg, and the starting price for a 0.25 mg pen containing 4 doses will be $24.35 (8,800 rupees). India has the “world’s second-largest population of type-2 diabetes patients.” Obesity rates are also a growing concern in India, and, as in the United States, Ozempic may be used to treat diabetes or be prescribed off-label for weight loss purposes in India. However, as Ozempic use rises globally, so are the reports of severe complications from GLP-1s, including gastroparesis and pancreatitis. Patients in the United States are filing lawsuits to hold Novo Nordisk accountable for not telling them about the full range of risks of Ozempic.
December 15, 2025: “Ozempic Face” Added to List of Complications from GLP-1 Weight Loss Injection
Ozempic is known to cause a range of side effects. Another side effect of the drug may be a change in the patient’s appearance. Doctor Roy Cowley, an aesthetician based in the UK, warns that people who use Ozempic and lose weight may experience skin sagging and a hollowed expression. Called “Ozempic Face,” this side effect is caused by dramatic, sudden weight loss, which does not give the skin time to naturally shrink. Instead, the muscle and fat leave quickly, and the facial skin sags. Dr. Cowley says many Ozempic patients are displeased about how they look after taking Ozempic because of how it changes their face. Meanwhile, patients in the United States are suing Novo Nordisk for not telling them that Ozempic could cause them to develop blindness (NAION), pancreatitis, ileus, or gastroparesis.
December 2, 2025: Australian Government Issues Warning About Ozempic and Reports of Suicidality, Self-Harm
The Therapeutic Goods Administration in Australia (a government agency similar to the FDA in the U.S.) warns that a concerning number of patients who take Ozempic and other GLP-1s reported suicidal ideation and self-harm. The TGA and its partners did not find a direct, causal link between taking a drug like Ozempic and experiencing suicidal or self-harm tendencies. However, enough patients who took Ozempic reported these adverse mental health events that the TGA thought a label change was necessary. Patients in the U.S. are also suffering from other complications, including organ damage, and are filing lawsuits against Ozempic’s manufacturer, Novo Nordisk.
November 24, 2025: Ozempic Share Prices Fall After Clinical Trial Results and Mounting Lawsuits
Shares for Novo Nordisk, Ozempic’s owner, fell to their lowest level since 2021. The 10% drop in Novo Nordisk’s share price coincided with the announcement that an Ozempic pill did not slow the progression of Alzheimer’s disease. A pill containing semaglutide did not perform well in clinical trials, as Novo Nordisk hoped it would. Now, share prices are down as the company faces thousands of lawsuits for Ozempic side effects and a failed new application of its semaglutide drug.
November 14, 2025: Ozempic Side Effect Lawsuits Approach 3,000
Ozempic and other semaglutide injections for diabetes and weight loss have empowered many patients to shed stubborn pounds. However, thousands have suffered dearly after taking this GLP-1 injection, which landed them in the hospital for blindness, pancreatitis, gastroparesis, and other complications. To date, there are 2,914 lawsuits in the multidistrict litigation against Novo Nordisk (Ozempic’s manufacturer) and other pharmaceutical companies that make GLP-1s. The litigation is ongoing, and more patients and their surviving family members can file new Ozempic claims in state and federal court.
November 3, 2025: Ozempic Interferes With Medical Imaging Tests, Study Finds
A study has found that taking Ozempic and other GLP-1 receptor agonists can cause issues with medical imaging tests (like PET scans). The researchers found that the Ozempic shot can make the contrast solution used for PET scans show up in abnormal patterns. As a result, doctors may misdiagnose someone with cancer they do not have, incorrectly label the stage of the cancer, and lead to unnecessary tests. This finding adds to a growing list of complications experienced by Ozempic patients. Ozempic patients are also suffering from pancreatitis, gastroparesis, and vision loss, and they are filing lawsuits because of it.
October 29, 2025: Report Says 12% of Americans Have Used Ozempic or Another GLP-1 Drug
About 12% of Americans have used Ozempic or another GLP-1 drug, many of whom have experienced severe side effects. This is according to a national survey conducted by RAND, a nonprofit, nonpartisan research organization. That percentage is even higher among women ages 50 to 64, where 20% of that population has taken a GLP-1 agonist drug. Another 14% of Americans are interested in taking one. At least half people taking the drug have reported nausea as a side effect. Thousands of others have experienced more severe side effects from Ozempic. Those severe side effects have resulted in thousands of lawsuits being filed against Ozempic’s maker, Novo Nordisk.
October 22, 2025: More People Join Ozempic Group Litigation in October, As Board Members Resign
As of October 2025, there are now 2,809 active lawsuits due to serious side effects of Ozempic and other GLP-1 drugs. These lawsuits are part of the federal multidistrict litigation in Pennsylvania Courts. More people injured by Ozempic continue to join the lawsuit. This increase in filings comes at a time when Novo Nordisk, Ozempic’s maker, is already having difficulties with revenue and board members. Seven members of Novo Nordisk’s board of directors, including its chairperson and vice chairperson, are leaving the board. Novo Nordisk is facing fierce competition from compounded GLP-1 drugs. The loss of revenue and GLP-1 lawsuits could place the Danish company in difficult times.
October 15, 2025: New Analysis Links Ozempic to Acute Kidney Injuries
A recent analysis that examined FDA data found that Ozempic (semaglutide) is associated with acute kidney injury (AKI). The analysis, titled “Are All Incretin Therapies Equal for the Kidney? Comparative FAERS Analysis of Tirzepatide and Semaglutide,” looked at adverse event reporting data from the FDA. Of the 92,807 adverse events reported for semaglutide drugs, about 440 people experienced AKI. People who took tirzepatide drugs had a much lower incidence. Kidney injuries are one of the reasons Ozempic patients have filed lawsuits against Novo Nordisk.
October 1, 2025: Most Ozempic Users Stop Taking It Within First Year of Starting, Study Finds
A new study has found that most people who take Ozempic discontinue treatment within 1 year. The reasons for the change include high and sometimes unpredictable costs, troublesome side effects, and co-occurring health challenges. The study discovered that over 40,000 of the 70,000+ participants taking a GLP-1 drug, like Ozempic, quit within one year, and 42% of the participants stopped within nine months. Many people are experiencing complications that cause discomfort, pain, or are life-threatening. Lawsuits are being filed in state and federal courts to hold Novo Nordisk accountable for not warning people of the full scope of the risks of Ozempic.
September 16, 2025: New Study Links Ozempic Use and Vision Loss; Defendants Seek Consolidation of Lawsuits
Another study, published in the Journal of the American Medical Association (JAMA), has found that people who take Ozempic for type 2 diabetes are at an increased risk of vision loss. Patients in the study were twice as likely to develop vision loss if they were taking Ozempic. The type of vision loss associated with Ozempic use is called NAION (nonarteritic anterior ischemic optic neuropathy). The condition is usually permanent and causes blurred or lost vision. Currently, people who took Ozempic and developed NAION cannot join the multidistrict litigation (group lawsuit) for people harmed by the drug. However, in an unusual turn of events, defendants in the lawsuit have asked for NAION lawsuits to be consolidated into a different group lawsuit, within the same federal court as the first group lawsuit. This action could indicate that drug companies recognize that GLP-1 drugs like Ozempic can do permanent damage to a patient’s vision.
September 3, 2025: Nearly 500 More People File Ozempic Lawsuits
Between August and September of 2025, almost 486 new plaintiffs joined the group lawsuit for people harmed by Ozempic. This means there are 2,676 pending lawsuits in the federal litigation against Novo Nordisk and other GLP-1 manufacturers. People who experienced gastric and intestinal conditions, blood clots, vision loss, and other severe complications from Ozempic may still be able to file a lawsuit to seek compensation for their injuries.
August 21, 2025: Novo Nordisk to Pay an Estimated $2 Billion for Ozempic Lawsuits
According to a recent analysis, Novo Nordisk may have to pay an estimated $2 billion in compensation for the lawsuits being filed against it. People are experiencing life-threatening and life-altering complications after taking Ozempic, a medication originally created to help people moderate their blood sugar levels. In recent years, its popularity has exploded, and some people use it off-label for weight loss. Some of the side effects that patients are complaining of include sudden vision loss, pancreatitis, gastroparesis, and kidney problems. The total number of lawsuits against glucagon-like peptide-1 (GLP-1) drug manufacturers, such as Novo Nordisk, has grown to nearly 2,200, with more cases being filed monthly. Novo Nordisk could assume even more than $2 billion in liabilities due to Ozempic.
August 19, 2025: Judge in Ozempic Lawsuits Rules on Medical Tests Required for Plaintiffs Claiming Gastroparesis
Many people who have joined the federal group lawsuit for those harmed by Ozempic have said the drug caused their gastroparesis. Now, in an August 15, 2025, memorandum, Judge Karen M. Marston has clarified what medical tests plaintiffs must provide to prove their case in court. Plaintiffs who say they developed gastroparesis will need to present a “properly performed gastric emptying study (scintigraphy, breath
test, or WMC).” This means that plaintiffs will have to show, by way of a medical test, that they have gastroparesis. Plaintiffs’ attorneys had hoped that a diagnosis based on symptoms would be sufficient to file a case. However, this order provides people harmed by Ozempic with clear expectations and a clear path forward for pursuing a settlement and compensation.
August 18, 2025: Ozempic Lawsuits Will Move Forward Against Novo Nordisk
Judge Karen M. Marston has issued an important opinion in the multidistrict litigation (i.e., federal group lawsuit) against Novo Nordisk and Ozempic. Her opinion stated which expert testimonies could and could not move forward in the litigation. Her opinion also says that lawsuits against Ozempic’s manufacturer can move forward based on breaches or express warranty and implied warranty. Basically, this means that Novo Nordisk made specific promises to patients that Ozempic was safe and effective when they put their drug on the market. The lawsuits can proceed on the grounds that Novo Nordisk may have violated those guarantees. This is good news for people who say they were not properly warned about the dangers of Ozempic and then developed serious injuries.
August 15, 2025: Scientists Uncover Natural Remedy for Weight Loss and Blood Sugar Concerns
Ozempic and other GLP-1 agonists experienced an unprecedented surge in popularity once their weight-loss benefits were discovered. However, the injection, which also helps people manage their diabetes, is fraught with side effects, some of which are deadly or cause patients to become suddenly and permanently blind. A new study published in the journal Nature suggests that there are natural remedies that can help patients lower their blood sugar levels, rather than relying on potentially unsafe injections. Meanwhile, people continue to file lawsuits after suffering severe side effects from Ozempic. There are currently 2,190 pending lawsuits in the federal litigation against GLP-1 drug manufacturers.
August 1, 2025: Study Finds that Ozempic May Cause Hair Loss, Especially in Women
A preliminary study suggests that Ozempic may increase the risk of hair loss in some patients. This is yet another undesirable side effect of Ozempic. The study’s authors examined over 3,000 participants, none of whom had diabetes or were taking medication to lower their blood sugar. The Ozempic group had a significantly higher rate of hair loss compared to the non-semaglutide group. Men taking Ozempic had a 52% higher hair loss rate, while the rate for women taking Ozempic was double that of women in the non-Ozempic group. Ozempic is causing hundreds to experience a wide range of complications, and many have filed lawsuits because of those side effects.
July 17, 2025: EU Clears Ozempic for Heart Conditions, Even Though Reports of Side Effects Are Rising
Drug regulators in the European Union have given doctors the all-clear to prescribe Ozempic to treat peripheral artery disease, a type of heart condition. Peripheral heart disease makes it harder for people with diabetes to walk long distances. Recent trials showed that Ozempic injections helped some patients walk farther than they could before. These broadened prescribing criteria for Ozempic mean that more people will experience severe side effects from the drug. This would mean more lawsuits filed against Novo Nordisk for Ozempic-related injuries.
July 2, 2025: People Suffering Vision Loss From Ozempic May Have New Group Lawsuit Option
A group of people who suffer vision loss after taking Ozempic and Wegovy has requested that their lawsuits be consolidated in New Jersey state courts. In late June, 21 plaintiffs requested that their lawsuits be combined via a process called multicounty litigation. If the court panel approves their request, these plaintiffs will be able to bring a group lawsuit against Novo Nordisk, Ozempic’s manufacturer. If this consolidation moves forward, people who took Ozempic and developed a rare eye condition called NAION (nonarteritic anterior ischemic optic neuropathy) will have new legal filing options. People who experienced other harms from Ozempic can still join the federal group lawsuit.
June 10, 2025: Taking Ozempic Doubles a Person’s Risk of a Type of Vision Loss
The European Medicines Agency (EMA) has concluded that taking Ozempic doubles a person’s risk of developing a type of vision loss called nonarteritic anterior ischemic optic neuropathy or NAION. The EMA, which is similar to the FDA, reviewed non-clinical studies, clinical trials, post-marketing surveillance, and the medical literature regarding NAOIN and semaglutide drugs, like Ozempic. Although the risk is relatively rare, the number of people at risk is high because of the number of people who take GLP-1 drugs containing semaglutide. NAION is a painless and sudden vision loss that is typically irreversible. It is one of the reasons people are filing lawsuits against Ozempic’s manufacturer.
June 2, 2025: Parties Will Discuss How Gastroparesis Is Diagnosed in Upcoming Ozempic Hearing
An important issue in the lawsuits against Ozempic and Novo Nordisk is how gastroparesis is diagnosed in patients. Many of the consumers who have filed lawsuits claim that Ozempic slows or stops patients’ digestion (a condition called gastroparesis). The parties have talked to different medical experts about how doctors assess whether someone has this condition, and now they will discuss their findings in an upcoming hearing. The plaintiffs’ attorneys plan to present materials about differential diagnosis, which is a standard method some medical experts use to decide if a patient has gastroparesis. This is a critical issue because many patients’ legal complaints say that the active ingredients in Ozempic caused them to develop this condition.
May 12, 2025: More Patients Sue Because of Side Effects of Ozempic and GLP-1 Products
There are 1,809 lawsuits pending against the companies that made and sold Ozempic, the semaglutide medicine used to treat things like diabetes and obesity. Serious complications of Ozempic include gastroparesis (where the stomach empties too slowly), thyroid tumors, pancreatitis, and bowel blockages. Many patients have been hospitalized because of severe side effects. Some have lost their lives due to complications caused by the drug.
May 1, 2025: Nearly 25,000 Emergency Room Visits Attributed to Ozempic and Semaglutide Drugs
A new study found that many people end up in the emergency room due to complications from taking Ozempic and other semaglutide drugs. The study’s findings, which were published in the Annals of Internal Medicine, used data from the CDC’s NEISS-CADES project to extrapolate how many emergency room visits might be linked with semaglutide side effects. The study found that from 2022 to 2023, there were almost 25,000 visits. Most of these visits (69.3%) were due to severe gastrointestinal adverse reactions. About 37% of these visits required hospitalization. This data underscores how Ozempic and other semaglutide drugs can cause real harms to patients. Many of these patients have chosen to pursue lawsuits, claiming they were not properly warned of the dangers of these “wonder drugs.”
April 16, 2025: Expert Hearing Approaching in Federal Ozempic Lawsuit
There is an important hearing coming up in the massive multidistrict litigation against the companies that made Ozempic and other GLP-1 products. On May 14, 2025, the plaintiffs and the defendant corporations will meet to discuss a reliable diagnostic process for identifying gastroparesis. Gastroparesis, which is when someone’s gut becomes fully or partially paralyzed, is a severe complication of taking drugs like Ozempic. Discovering more details about the medical examination and treatment process for gastroparesis will help the people filing the lawsuits develop their arguments against the pharmaceutical companies.
April 1, 2025: MDL Against Ozempic and Other GLP-1s Top 1,600 in April
As of April 1, 2025, there are 1,685 active lawsuits pending against Novo Nordisk and other pharmaceutical companies that sold GLP-1 drugs like Ozempic. These drugs have been used to help people control their diabetes and lose weight in recent years. However, hundreds of consumers are experiencing severe side effects from taking Ozempic. Some people have experienced paralysis of their digestive system or lost their lives due to complications from using Ozempic. Many of these people or their surviving family members have filed lawsuits against Novo Nordisk for not properly warning them of the hazards of the drug.
March 20, 2025: People Who Filed Ozempic Lawsuits May Benefit From Two Developments in the Case
On March 18, 2025, the judge in the group Ozempic lawsuit, Judge Marston, held a court conference. During that conference, she discussed issues related to deposition protocol and the short-form complaints. Establishing the process for depositions is especially important when litigating large, corporate entities like Eli Lilly or Novo Nordisk. Attorneys representing people harmed by Ozempic will likely want more depositions, and the defense will likely want fewer. Plaintiffs’ attorneys want to understand what employees at these pharmaceutical companies knew, when they knew it, and how they came to know that information. Discussions about short-form complaints are promising for many plaintiffs because these complaints are much less burdensome than long-form complaints. If a short-form complaint is used, it would be accompanied by a master complaint, which would allow plaintiffs to use established facts for their case, instead of submitting these facts separately. These developments are likely a net positive for people harmed by Ozempic and other GLP-1 drugs.
March 3, 2025: Go-Forward Plan in Ozempic Lawsuits Discussed During Status Hearing
In a recent status conference for the Ozempic group lawsuit, Judge Karen Spencer Marston discussed a number of important milestones for GLP-1 lawsuits in 2025. The short-form complaint was discussed. This form will allow people harmed by Ozempic and other GLP-1 drugs to streamline documents supporting their injuries. At the conference, a date was also set for the Rule 702 hearing. This hearing, which will determine what expert testimonies will be relied on for the case, will take place on May 14, 2025. People who experienced severe digestive injuries and other serious conditions can still join the group lawsuit against GLP-1 manufacturers.
February 26, 2025: FDA Removes Ozempic From Drug Shortage List
On February 21, 2025, the FDA removed Ozempic from its drug-shortage list. Ozempic had been on this list since August 2022. The FDA received inventory and production data from Novo Nordisk, and that data made the FDA confident that the Ozempic shortage was over. Semaglutide drug compounders have already sued the FDA for this decision, saying it was a “reckless and arbitrary” decision. These events come as more people are filing lawsuits against Novo Nordisk after experiencing a range of severe side effects.
February 20, 2025: Ozempic’s Label Updated to Include Risks of Severe Injuries or Death Related to Pancreatic and Kidney Injuries
The U.S. FDA has posted an updated drug information label for Ozempic. The drug now carries a warning for severe pancreatitis and severe kidney injuries. The adverse reaction warning for pancreatitis includes fatal and non-fatal hemorrhagic or necrotizing pancreatitis. The risk for severe kidney injury is due to severe dehydration and volume depletion due to diarrhea and vomiting from Ozempic. These updates were made based on clinical trial data. Ozempic’s maker, Novo Nordisk, is already facing hundreds of lawsuits due to other severe side effects of taking semaglutide drugs.
February 3, 2025: More Than 100 New Lawsuits Join Litigation Against Ozempic and Other GLP-1 Drugs
As of February 3, 1,443 people have joined the multidistrict litigation (MDL) against Ozempic’s maker (Novo Nordisk) and other GLP-1 manufacturers. One of those who joined the MDL was a Michigan woman who says she experienced severe gastrointestinal side effects after taking Ozempic. The woman’s symptoms were so severe that she required hospitalization. Her lawsuit says Novo Nordisk did not adequately warn people of severe side effects and instead marketed Ozempic as a wonder drug. As these cases proceed through the MDL process, the next status conference will be held on February 24, 2025, at 2:00 p.m. The agenda for this conference will be posted at least 3 days in advance.
January 16, 2025: Key Trial Date Set in Ozempic Lawsuits
Many people who were harmed by Ozempic have joined the consolidated federal litigation for GLP-1 drugs (MDL 3094: GLP-1 Products Liability Litigation). A key date has been set in this trial. Currently, expert witnesses on both sides are gathering information to prove and disprove that GLP-1 drugs cause many of the diseases experienced by plaintiffs. There will be an evidentiary hearing on May 14, 2025. In this hearing, the judge will determine if the reports and evidence produced by the experts can be used in court. If the opinions of the plaintiffs’ experts are deemed admissible, this will be good news for people harmed by Ozempic and other GLP-1 drugs.
January 9, 2025 – People With Blood Clots or NAION From Ozempic Will Have Separate Legal Options
Some people who have been injured by Ozempic have joined MDL 3094: In Re: Glucagon-like Peptide-1 Receptor Agonists (GLP-1 RAS) Products Liability Litigation. Recently, the Judicial Panel on Multidistrict Litigation (JPML) was asked to add two injuries to the complaint—deep vein thrombosis (blood clots) and NAION (permanent vision loss from eye strokes). The JPML decided not to add these injuries to the existing MDL. This could be positive news for people suffering from these conditions, as a new MDL could be formed. DVT and NAION are serious conditions, and people who are diagnosed with these conditions due to their Ozempic use deserve to fight for compensation that is equivalent to their injuries.
January 2, 2025: More People File Ozempic Lawsuits Ahead of 2025
As of January 2, 2025, there are 1,331 pending lawsuits against GLP-1 drug manufacturers as part of federal, consolidated litigation. People who have filed these lawsuits (plaintiffs) say they were harmed by GLP-1 drugs like Ozempic. These plaintiffs say GLP-1 drugs led them to be diagnosed with gastric disorders, liver and pancreatic injuries, bowel conditions, or other injuries. Many people harmed by Ozempic have joined the multidistrict litigation (MDL). From December 2024 to January 2, 2025, 31 people joined the MDL.
December 19, 2024: Two New Studies Link Ozempic to NAION, Possibly Leading to Investigation by European Medicines Agency
The findings of two medical studies conducted by the University of Southern Denmark have led the Danish Medicines Agency (DMA) to ask for an investigation into the link between Ozempic use and the rare eye condition NAION. DMA will request an assessment from the European Pharmacovigilance Risk Assessment Committee (PRAC). NAION, which stands for nonarteritic anterior ischemic optic neuropathy, is an eye condition that affects the blood vessels in the eye and can lead to long-term vision loss. Both of the studies followed a large cohort of patients from Denmark and Norway who were treating their type 2 diabetes with Ozempic. The findings of both studies pointed to an increased risk of developing NAION. The DMA said the findings were strong enough that there should be an investigation into whether or not NAION should be listed as a possible adverse event when taking Ozempic.
December 18, 2024: FDA Updates Ozempic Warning Label to Include Pulmonary Aspiration as Adverse Reaction
The FDA updated the warning label for Ozempic, the GLP-1 drug used to treat diabetes and lose weight. The new label states that patients who use Ozempic and go under anesthesia or are sedated have a higher chance of getting food or liquid into their lungs, which is called aspiration. Aspiration can lead to serious complications or death. Because Ozempic causes delayed stomach emptying, patients may undergo sedation with partially digested food in their stomachs, even if they stopped eating when their physician asked them to. With the update, the FDA is urging patients taking Ozempic to talk to their doctors about the risks before being sedated.
December 12, 2024: Judge Approves Schedule for Upcoming Motions in Federal Ozempic MDL
On December 3, 2024, the federal Judge handling the massive case against the makers of Ozempic entered a scheduling order to set deadlines for the defendants’ motions to dismiss. It is common for defendants to file these types of motions. Having a set schedule for this motion gives the plaintiffs time to prepare their potential legal arguments to fight against the companies’ attempts to sidestep liability.
December 2, 2024: Ozempic and Other GLP-1 Lawsuits Reach 1,300 Active Plaintiffs
As of December 2, 2024, there are 1,300 pending lawsuits in the consolidated federal lawsuits against manufacturers of GLP-1 drugs such as Ozempic, which is manufactured by Novo Nordisk. The number of pending lawsuits in MDL 3094, Glucagon-like Peptide-1 Receptor Agonists (GLP-1 RAs) Products Liability Litigation has been steadily increasing as more people allege injuries such as gastrointestinal disorders, blood clots, and vision loss were caused by their use of Ozempic.
November 27, 2024: Researchers Warn of Kidney Injury Risk When Taking Ozempic
A recent research study suggested that some patients taking GLP-1 drugs like Ozempic may experience acute kidney injuries, such as acute interstitial nephritis (AIN). People taking GLP-1 drugs may also have elevated serum creatinine levels. Creatinine is a waste from muscles that healthy kidneys should be able to filter out in a person’s urine. Although GLP-1 drugs like Ozempic may have some renal benefits, the study’s authors stressed the need to monitor patients for adverse conditions like AIN and elevated serum creatinine levels.
November 26, 2024: Parties in Ozempic MDL Set Important Case Management Court Dates
On November 26, 2024, the Judge overseeing the Ozempic lawsuit entered an order setting key case management dates for the coming year. These dates allow the parties to discuss pending matters or introduce new ones. The order encourages the parties to meet before these court dates and says they can file a document suggesting agenda items up to one week beforehand.
November 22, 2024: Researchers Ramp Up Warnings About Ozempic’s Cancer Risks to Patients
Researchers continue to caution patients and medical professionals about the increased risks of using Ozempic, a glucagon-like peptide 1 (GLP-1) receptor agonist. Studies suggest a correlation between using GLP-1 drugs and an increased risk of developing thyroid cancers. The risk appears to increase with higher dosages or prolonged use of the type 2 diabetes drug. Researchers urge medical professionals to study the potential carcinogenic features of Ozempic more closely.
November 15, 2024: Plaintiffs’ Attorneys File Appearance in Ozempic Lawsuit Ahead of Expert Report Deadline
Attorneys for plaintiffs in the Ozempic MDL file their appearance before the deadline to submit expert reports supporting the injured consumers. Courts require people to file a document—called an appearance form—before they can participate in the case. The appearance form gives the Judge basic information about them, so it is on file, and the Judge knows whom to expect filings from.
November 7, 2024: Ozempic MDL Judge Sets New Deadline To File and Respond to Expert Reports
The judge handling the Ozempic multidistrict litigation (MDL 3094) signed a court order on November 5, 2024, setting critical deadlines for a round of expert reports and responses. The plaintiffs must submit their first batch of expert reports on November 18, 2024, unless they file for an extension. The defendant corporations—like Novo Nordisk—have until December 23, 2024, to file their initial expert reports.
November 1, 2024: Physicians Warn People Taking Ozempic of Skeletal Muscle Mass Loss and Call for More Research
Recent commentary published in JAMA warns that skeletal muscle mass (SMM) experienced while taking GLP-1 drugs may be of medical concern. SMM, which is called sarcopenia, likely occurs because of the rapid weight loss many people experience while taking Ozempic and other GLP-1 drugs. In their statement to JAMA, William J. Evans, PhD, and Steven Cummings, MD warn that sarcopenia in older adults may be especially concerning, as it can contribute to hip fractures, disability, and increased mortality. Dr. Cummings and Dr. Evans concluded that, “There is an urgent need for studies that accurately measure muscle mass, strength, and mobility during GLP-1 agonist–induced weight loss and regain of weight after they are stopped, especially in older people.”
October 28, 2024: FDA Issues Warning About Using Compounded Semaglutide Drugs for Weight Loss
In October, the FDA issued a warning to patients who take compounded GLP-1 drugs for weight loss. The FDA said that they have received 346 reports of adverse events associated with compounded semaglutide, the active ingredient in Ozempic. Many people are taking compounded GLP-1 for weight loss. These drugs are created at pharmacies, where pharmacists combine different drugs and additives. Some estimates say up to 2 million Americans have taken compounded GLP-1 drugs, which in many cases are completely legal. In their warning statement, the FDA said taking compounded GLP-1 drugs can be risky for patients because these drugs are not evaluated by the FDA for safety, effectiveness, and quality.
October 25, 2024: More Ozempic Lawsuits Filed as People Experience Severe Side Effects
As of October, there are 1,090 pending lawsuits in the Ozempic multidistrict lawsuit (MDL-3094 IN RE: Glucagon-like Peptide-1 Receptor Agonists (GLP-1 RAs) Products Liability Litigation). People who have experienced severe gastroparesis, organ injuries, blood clots, vision loss, and other complications from taking Ozempic continue to file lawsuits against Novo Nordisk and other GLP-1 manufacturers. We expect additional lawsuits to be filed in the coming months.
October 21, 2024: Judge Makes Critical Decision in Pre-Discovery Phase of Ozempic Lawsuit
The judge in the federal Ozempic lawsuit (MDL 3094) denied the plaintiffs’ motion to require the defendants to turn over specific marketing materials early in the case. One of the critical issues in the case is whether Ozempic’s warning labels gave people enough information about the potential risks and side effects of taking this weight-loss drug used to treat type 2 diabetes.
Plaintiffs argued that the aggressive and “unprecedented” marketing campaign for Ozempic essentially drowned out the information on the product’s warning labels. They asked the judge to require the defendants to turn over marketing materials from specific periods. After each side filed their arguments and talked to the judge during several hearings, the judge denied the plaintiffs’ motion to request this information at this time.
October 17, 2024: Dispute Over Discovery in Ozempic Lawsuit Continues
Lawyers in the Ozempic lawsuit continue to debate what information Novo Nordisk should be required to provide to the plaintiffs. The defense has successfully delayed the lawsuit by arguing that the judge should first determine whether the warning label precludes certain claims, such as gastroparesis. Plaintiff lawyers have requested marketing materials that the pharmaceutical manufacturer provided to doctors who prescribed Ozempic. This is a key issue regarding what discovery will be permitted at this stage. A decision on this discovery matter is expected within a few weeks.
October 4, 2024: Large Number of Plaintiffs File Ozempic Lawsuits in September
As of October 1, 2024, there were 1,090 cases pending in MDL 3092, Glucagon-like Peptide-1 Receptor Agonists (GLP-1 RAs) Products Liability Litigation. This MDL is the Federal consolidated lawsuit against the manufacturers of GLP-1 drugs, like Ozempic. From August to September, there was nearly a 20% increase in the number of lawsuits in the MDL. The GLP-1 litigation is a fast-growing lawsuit, and we expect tens of thousands of cases to be filed as more Americans take GLP-1 agonist drugs and experience harmful, serious side effects.
October 1, 2024: Increased Reporting of Ozempic Side Effects Will Corner Novo Nordisk to Address Failure to Warn Allegations
As media outlets continue to cover more stories about the side effects of Ozempic, Novo Nordisk’s public relations team will be cornered to maintain public trust and address these claims. Despite the reported side effects from Ozempic users, ad spend for the drugmaker remains substantial and continues to drive the use of its weight loss drugs through aggressive marketing campaigns. Lately, there’s been an obvious marketing push for Wegovy in TV commercials and print ads in doctors’ offices across the country. The growing awareness of these side effects should naturally drive more interest in the Ozempic lawsuits and fuel the failure to warn allegations cast against Novo Nordisk.
September 29, 2024: Increasing Media Coverage Highlights Severe Health Risks Linked to Ozempic
Mainstream media have started covering more stories from plaintiffs alleging that the drug Ozempic caused severe gastrointestinal issues, including gastroparesis and intestinal blockage. Numerous individuals have reported unbearable stomach pains, an inability to keep down solid foods, and prolonged vomiting and diarrhea. With more people using Ozempic, we’re now seeing larger-scale reporting of the drug’s side effects. Stories about Ozempic’s health risks have routinely been overshadowed by favorable coverage about its weight-loss benefits from large media outlets.
Our firm expects media reports of gastrointestinal issues to grow, along with cases of blood clotting and vision loss in semaglutide users.
September 25, 2024: Novo Nordisk Faces Senate Scrutiny Over Ozempic Pricing and Safety Concerns
Novo Nordisk, the company that manufactures Ozempic, has recently come under fire from the United States Government for its excessive pricing, with significantly lower costs for Europeans and other countries using the same drug. This month, the CEO of Novo Nordisk, Lars Fruergaard Jorgensen, appeared before the United States Senate Committee on Health, Education, Labor, and Pensions. He reiterated that “the full value of Ozempic and Wegovy can only be realized if patients can access them. Patients need affordability and access.”
While the Senate focuses on pricing, it may be overlooking a more important issue. Novo Nordisk and the government have failed to promote awareness of Ozempic’s potential risks, including conditions such as blindness, stomach paralysis, bowel blockages, and pulmonary embolisms. Lowering the cost of Ozempic may increase the population at risk for these serious health conditions, ultimately endangering more people with its dangerous side effects.
September 10, 2024: Woman Prescribed Ozempic Files Lawsuit for Failure to Warn
In a story first reported by CBS News, Juanita Gantt shared that her doctor recommended she try Ozempic and Wegovy due to her heightened risk of diabetes and ongoing challenges with weight loss. Like many patients, she initially felt fine while taking the medications, but her health took a sudden turn when her husband found her unconscious on the floor. During her stay in the hospital, doctors found that parts of her large intestine had died and required removal. Following surgery, she suffered cardiac arrest.
Gantt is now one of thousands of individuals suing Ozempic’s manufacturer, Novo Nordisk, alleging that the drug’s warning labels fail to properly warn patients about side effects related to gastroparesis, ileus, and bowel obstruction. In response, Novo Nordisk stated that the claims against Ozempic and Wegovy are unfounded and that the company will continue to defend itself against these lawsuits brought by former users.
September 1, 2024: Kentucky Woman Files Lawsuit Alleging Ozempic Caused Gastroparesis
On August 27, 2024, a 43-year-old Kentucky woman filed an Ozempic lawsuit, alleging that the GLP-1 drugs caused her gastroparesis, or stomach paralysis. The lawsuit claims that the plaintiff used both Ozempic and Trulicity at different times from 2021 to 2024 and that the defendants (Novo Nordisk and Eli Lilly) downplayed the seriousness of the side effects and should have better warned patients.
The complaint, filed in the Eastern District of Pennsylvania federal court, alleges that gastroparesis is an incurable condition that affects normal muscle movement in the stomach and causes symptoms such as nausea, vomiting, abdominal pain, abdominal bloating, severe dehydration, and other side effects.
The plaintiff demands a judgment against the defendants for pain and suffering, severe and permanent personal injury, healthcare costs, and medical monitoring.
August 25, 2024: Case Management Order in Ozempic Lawsuit Favors Defense Requests for Briefing
Case Management Order No. 18 in the Ozempic lawsuit grants the defense’s request for briefing on how the injury of gastroparesis must be claimed. The defense argues that a gastric emptying test is the only objective and reasonable method of diagnosis, while the plaintiff contends that symptoms alone can serve as the basis for the diagnosis. The plaintiffs’ lawyers have previously stated that 95% of the claims in the Ozempic lawsuit will allege gastroparesis.
The defense also requested motion dates to determine whether the warning labels will preempt some or all of the claims. Additionally, the defense sought discovery and motion practice on general causation—specifically, whether GLP-1RAs can cause intestinal and bowel injuries as alleged in the Ozempic lawsuits. The judge reserved judgment on this application. So far, the judge appears to lean in favor of the defense. There are currently 842 cases pending in the Ozempic MDL.
August 13, 2024: Judge Marston Issues Cost-Sharing Order for Ozempic Lawsuit
Judge Marston has issued an order detailing how the plaintiffs’ lawyers will share the expenses incurred in the Ozempic lawsuit. Case Management Order Number 17 establishes protocols for the lawyers to submit costs during the preliminary phases of the multidistrict litigation (MDL). MDLs require dozens of law firms to work together for several years and manage the litigation. Those firms are often partially compensated through Common Benefit Funds. Money is paid into these funds by the lawyers representing individual plaintiffs. These funds help cover expenses like legal research, expert witnesses, depositions, and other shared costs necessary for advancing the case for all plaintiffs. Common Benefit Funds are funded by a percentage of the lawyer’s portion of the settlement, not the client’s portion. The goal of these funds is to ensure the costs of the broader litigation effort are fairly distributed.
August 6, 2024: Judge Marston Issues Detailed Discovery Order in Ozempic Lawsuit
Judge Marston has issued a thorough discovery order in the Ozempic Lawsuit. The 19-page order, filed on July 31, 2024, details what documents will be produced by defendant Novo Nordisk in the Ozempic Lawsuit. The order requires that the parties meet and confer on several issues. The first important issue will be what custodians the plaintiffs and defendants agree will have relevant information for the Ozempic Lawsuit. Some of the people with important documents in their possession will later be questioned under oath at a deposition by plaintiff lawyers. We believe that there will be a heavy emphasis placed on the marketing department because of the aggressive and non-traditional marketing tactics employed by Novo Nordisk in selling the weight loss drug. The lawsuit includes failure to warn claims, as well as deceptive marketing claims by the plaintiffs.
August 2, 2024: Link Between Ozempic and Blood Clots, DVT, Pulmonary Embolisms Examined
A comprehensive meta-analysis that found a 266% increase in blood clots in people who use semaglutide drugs is gaining new traction. The article, published in the Endocrine Journal, looked at outcomes of patients involved in trials of semaglutide drugs. It found a substantially increased risk of deep vein thrombosis (DVT) for patients taking semaglutide drugs to treat type 2 diabetes. Patients with DVT are at risk of pulmonary embolisms, which can be fatal. As the number of people using semaglutide drugs rises, more people may be at risk for this dangerous type of blood clot.
August 1, 2024: Ozempic Lawsuit Filed by Florida Man in Pennsylvania Federal Court
A Florida man was the original plaintiff in the Ozempic lawsuit. The complaint titled Roderick Shirley v. Novo Nordisk was filed on December 15, 2023. The complaint was filed in the federal court for the Eastern District of Pennsylvania. The plaintiff alleges that the case was filed in the Pennsylvania district court because Wegovy pens are assembled there. The court document further alleges that Novo Nordisk failed to warn the plaintiff about the risks of Ozempic, demonstrating negligence and deception in marketing, and a design defect. The plaintiff alleges that he was diagnosed with deep vein thrombosis, also known as DVT, and a related pulmonary embolism when the blood clot traveled to his lungs. The injury resulted in surgery, a three-week hospitalization, and over two weeks in the ICU. The complaint alleges that the injury was the result of the use of Ozempic and Wegovy.
July 24, 2024: Plaintiffs in the Ozempic Lawsuit Required to Submit Detailed Fact Sheets
Plaintiffs in the Ozempic Lawsuit are now required to file a Plaintiff Fact Sheet. Case Management Order number 12, filed on July 14, 2024, stipulates that after filing a complaint, the plaintiff must then provide basic information to the defendant. The fact sheet requires the plaintiff to specify the type of GLP-1 RA they used and the injuries they allegedly suffered. The plaintiff must also state whether they are alleging economic damages or psychological injury. Plaintiff fact sheets are common requirements in multidistrict litigations. This is an important step in the lawsuit. For plaintiffs interested, we have access to the detailed, 17-page Ozempic Plaintiff Fact Sheet provided by the judge for viewing.
July 23, 2024: Rise in Ozempic Lawsuits Following Study on Vision Damage Risks
Ozempic lawsuits have been on the rise, especially since the publication of a previous study showing an increased risk for sudden vision damage. Allegations continue to surface that Ozempic can lead to multiple life-long conditions, such as gastroparesis and nonarteritic anterior ischemic optic neuropathy (NAION), dramatically impacting lives. We are still accepting cases for this litigation.
July 9, 2024: New Study Links Semaglutide to Increased Risk of Vision Problems
A new study by Dr. Jimena Hathaway, MD, MPH, published in the Journal of the American Medical Association, indicates that Semaglutide, the active ingredient in Ozempic, Wegovy, and Rybelsus, shows an increased risk of vision loss. The study found that in a pool of 16,827 patients, nonarteritic anterior ischemic optic Neuropathy (NAION) increased more than four times for people taking semaglutide compared to those who took another drug. The analysis was conducted from 2017 to 2023. This is a critical study in the Ozempic lawsuit because the label does not warn of this side effect. Eye doctors and lawyers have suspected vision problems for some time. We expect that the federal court lawsuit will soon include Ozempic vision loss cases, including blindness and blurred vision.
June 28, 2024: Study Reveals Potential Bone Density Reduction with GLP-1 Treatment
In a recent study, Simon Jensen, Ph.D., and others found that GLP-1 treatment, without exercise, could lead to reduced bone density. The study was published on June 25, 2024, in JAMA Network. The most common GLP-1 is Ozempic. The study follows speculation that the reduction in nutrient and water absorption by the body could have long-term health implications. So far, the Ozempic lawsuit has focused on gastrointestinal side effects like gastroparesis and bowel obstructions.
June 17, 2024: Ozempic Lawsuit Progresses with New Judicial Orders and Status Conference
The Ozempic lawsuit is again beginning to move. The first status conference with the new judge was held on June 10, 2024. The focus was on the logistics of moving the MDL forward. Since the conference, the judge has issued two mundane orders. First, lawyers who wish to file cases in the GLP-1 MDL will not have to pay a fee to be admitted pro hac vice. Second, the parties have filed a motion, seemingly with permission from the judge, regardless of privilege logs. Parties in lawsuits have to turn over relevant information to the other side in lawsuits like this one. However, sometimes there are privileges that can be asserted. If a document is not turned over due to privilege (like a communication with a lawyer), then the document is supposed to be logged, and the other side informed that it exists. This log is called a privilege log and is part of every case.
June 8, 2024: New Judge Appointed in Ozempic Lawsuit, Case Moves Forward
There is a new judge in the Ozempic Lawsuit. Case Management Order number 7 was filed on June 6, 2024, by Judge Karen Spencer Marston. Judge Marston will be the new judge overseeing the case. She was nominated for the federal bench following a lengthy career as an Assistant US Attorney. The next conference will be held in person in the Eastern District of Pennsylvania on June 10, 2024.
June 2, 2024: Ozempic Lawsuit Paused in Federal Court Awaiting New Judge Appointment
The Ozempic lawsuit pending in federal court is at a standstill until a new judge is appointed following the unexpected death of the previous judge. We also expect to see cases continue to be filed in New Jersey state court. Federal court is the appropriate venue when there are parties from two different states. Cases that will be brought against Novo Nordisk by residents of the State of New Jersey will probably have to be brought in New Jersey court. It is possible that other plaintiffs will file claims in a New Jersey state court as well.
May 22, 2024: Science Day Canceled in Ozempic Lawsuit, Case Reassignment Possible
Science Day has been canceled in the Ozempic Lawsuit. The Chief Judge of the Eastern District of Pennsylvania, Mitchell Goldberg, issued an order on May 21, 2024, vacating Case Management Order number 5 and canceling Science Day. The case must be reassigned. Perhaps Judge Goldberg will be the new judge on the case.
May 20, 2024: Judicial Shift in Ozempic Lawsuit After Judge Pratter’s Passing Impacts Proceedings
The Ozempic lawsuit is in a period of flux with the death of Judge Pratter. Judge Pratter was the presiding Judge in the lawsuit and was taking an active and aggressive role. The plaintiffs and defendants will now await a new judge to be appointed by the Judicial Panel on Multidistrict Litigation. Plaintiffs will hope for a more liberal plaintiff-friendly judge. The Eastern District of Pennsylvania is thought to be pro plaintiff, with several large verdicts in Philadelphia recently. The death of Judge Pratter may delay the case temporarily.
May 19, 2024: Novo Nordisk and Eli Lilly Outline Legal Strategy to Reduce Liability and Challenge Plaintiff Claims on Ozempic and Mounjaro
Novo Nordisk and Eli Lilly recently outlined their strategy in a defendant statement to reduce liability and minimize plaintiff recoveries. Both companies covered the benefits and advantages of Ozempic and Mounjaro and countered the plaintiffs’ focus on failure to warn for GLP-1RAs. The defense categorized the lawsuits into different injury classes, which puts pressure on the plaintiffs to provide specific evidence for each of the cases. They’re also pushing for plaintiffs to disclose information about unfiled cases, but that is not legally required. They also pushed for early motions to dismiss by requesting objective testing for “gastroparesis,” which can complicate the plaintiffs’ burden of proof.
May 3, 2024: Plaintiffs Respond to Defendants’ Position Statement in Ozempic Lawsuit
The plaintiffs filed a statement in the Ozempic lawsuit responding to the defendants’ “Statement of the Case”. The plaintiffs’ statement outlines the framework they will argue and attempts to dismiss the defendants’ arguments from their filing. The statement highlights a conflict between the two parties about what should be required to file a lawsuit. The defendant has argued that a plaintiff should be required to have a gastric emptying study to bring a lawsuit. The plaintiffs’ lawyers say that the test is not required. Instead, they assert that a doctor’s diagnosis of gastroparesis, stomach paralysis, or bowel injuries is sufficient, at least for the early stages of a lawsuit.
April 30, 2024: ‘Science Day’ Scheduled for the Ozempic Lawsuit
Judge Pratter issued Case Management Order number 5 in the Ozempic lawsuit and scheduled “Science Day” for June 14, 2024. Science Day will provide an overview of the medical and scientific issues relevant to the case. Plaintiffs and Defendants will have a designated time slot to present their positions to the Judge during Science Day. It would be interesting to know whether the plaintiffs’ lawyers will focus on Ozempic and other semaglutide drugs or split their time with some of the Eli Lilly drugs as well. Unfortunately, the Judge ruled that the record from Science Day will be confidential to the Court.
April 21, 2024: Drawing Parallels – Ozempic Lawsuit and Benicar MDL Settlement Analysis
Some guidance in the Ozempic lawsuit might be gained from a review of the Benicar MDL. Benicar was a common blood pressure medication in the early 2000s. Like the Ozempic case, the Benicar case alleged a failure to warn of serious gastrointestinal side effects, like chronic diarrhea and bowel obstructions. In 2017, the case settled for a reported $300 million to 2,300 plaintiffs. Other reports indicated the actual settlement value was closer to $360 million. The most serious cases are reported to have settled for over $500,000, while others received much less. The average, however, seems to have been well over $100,000. It would not surprise us if the settlement matrix in that case is a target for the lawyers in the Ozempic case.
April 20, 2024: Ozempic Lawsuit Is About Whether Users Knew of Potential Side Effects
The plaintiffs and defense have begun to submit their respective positions to the judge in the Ozempic lawsuit. Early court documents show that this case will be about whether the manufacturer properly warned of the side effects. Plaintiff lawyers say that gastroparesis and other bowel injuries are unwarned consequences of the use of Ozempic and similar drugs. The defense states that intestinal issues, some severe, are side effects that everyone knew about. Part of their defense is that the doctors knew of the side effects and had an obligation to inform the patients.
April 11, 2024: Ozempic Lawsuit Joint Defense Statement Filed by Novo Nordisk and Eli Lilly
Defendants Novo Nordisk and Eli Lilly have submitted a joint statement of the case in the ongoing Ozempic lawsuit. This document, filed in federal court on April 9, 2024, aims to outline the defense’s strategy over the coming months. At the time of filing, 117 plaintiffs had initiated lawsuits: 100 cases targeted Novo Nordisk, 8 targeted Eli Lilly, and 9 involved both defendants. According to the defense, GLP-1RA drugs are revolutionary medications with FDA-approved warnings, asserting that they are generally safe and have undergone extensive testing. The defense also claims to have insufficient information about both filed and potential cases. They note a rise in counterfeit products and assert that many plaintiffs alleging “gastroparesis” lack substantial evidence of their diagnosis. This filing provides a significant insight into the defenses planned for the Ozempic lawsuit.
April 10, 2024: Additional Cases Transferred to Eastern District of Pennsylvania
The April 4, 2024, conditional transfer order signed by Tiffaney D. Pete, Clerk of the Panel on Multidistrict Litigation, states that two additional cases were transferred to the United States District Court for the Eastern District of Pennsylvania and have been assigned to Judge Gene Pratter as part of the Ozempic Lawsuit (MDL No. 3094). The Order notes that 17 cases were transferred in the original transfer order on February 2, 2024. Since that time, 36 more cases have been added to the lawsuit. Like most MDLs, the filings have been slow to start with. Most law firms will wait for the lawyers to agree on a short-form complaint, then file their cases. A short-form complaint is a simplified pleading that gives the basics of each plaintiff’s complaint. We expect that the Ozempic complaint will include things like which drug was prescribed to the plaintiff, date of prescription, the diagnosed injury, the theory of liability against, and whether the claim is against Novo Nordisk or Eli Lilly.
April 1, 2024: Ozempic Manufacturer Novo Nordisk Sues Pharmacies for Knock-Offs
Ozempic manufacturer Novo Nordisk continues to pursue lawsuits against pharmacies that sell compounded semaglutide. The FDA has discouraged the National Association of Boards of Pharmacy from compounding Ozempic, Wegovy, and Rybelsus. The FDA further notes that different forms of semaglutide appear to violate federal law. On January 10, 2024, the FDA issued guidance acknowledging the risk and popularity of knock-off Ozempic. The biggest concern is “compounded” Ozempic. Novo Nordisk has settled several lawsuits against pharmacies providing substances that are not, in fact, Ozempic. Of course, the federal lawsuit alleges that even in its purest form, Ozempic can be dangerous, and the manufacturer is now properly warning of the risks associated with the drug.
March 23, 2024: Ozempic Lawsuit Leadership Structure Proposed
Plaintiff lawyers in the Ozempic lawsuit have submitted a motion for leadership structure to the court. This is no surprise. Court leadership needs to get to work on setting schedules and making requests for documents from the drug manufacturers Novo Nordisk and Lilly, formerly known as Eli Lilly. Our litigation partner, Daniel Nigh, Esq., is a proposed member of the executive committee. Daniel is an experienced dangerous drug lawyer, and we expect him to be on the frontlines of the litigation of the serious scientific issues in the Ozempic case. We expect the motion to be granted in short order by Judge Pratter.
March 21, 2024: Anticipating a Surge of Ozempic Lawsuits in New Jersey Courts
The Ozempic lawsuit will soon become the Ozempic lawsuits. We believe that a substantial number of Ozempic lawsuits will be filed in New Jersey state court, in addition to the cases being filed in federal court. We expect that law firms with a large volume of Ozempic cases will file some in state court as a way to hedge their bets against putting all their cases into one litigation, in front of one judge. The biggest risk in a large federal court tort case is a bad Daubert ruling. “Daubert” refers to a landmark case that sets the standard for what an expert can testify to. A bad Daubert ruling can lead to the dismissal of all cases. New Jersey is the most likely state court venue because it is the U.S. headquarters of Novo Nordisk, the Ozempic manufacturer. In addition, some lawyers who are not satisfied with their role in the federal court case may choose to file in state court instead. Several Ozempic cases have already been filed in New Jersey state court.
March 20, 2024: Status Conference Update in Ozempic Lawsuit
The first status conference was held in the Ozempic Lawsuit last Thursday. The conference began with the Judge asking for proposals about how the plaintiffs would organize the several lawyers from law firms around the country who are filing cases. Lawyers from Eli Lilly made an aggressive early play asking the judge when they could file for summary judgment. The court said that would be a long time down the road. Early estimates are that Eli Lilly will defend about 10% of the cases for their drug, Mounjaro. Some lawyers believe that the most serious injuries come from Ozempic and Wegovy. The comments seem to indicate that the case will include both manufacturers for the foreseeable future. Finally, the court indicated a “science day” where each side gets to present to the Judge about the science involved in this case. Science days help the Judge understand the case more fully as they rule on legal issues. We anticipate the Science Day will occur in the next three months in the Ozempic Lawsuit.
March 10, 2024: Important Decisions Upcoming In Ozempic Lawsuit
Judge Gene E.K. Pratter, the presiding Judge in the Ozempic lawsuit, will soon appoint lawyers to certain roles and set a discovery schedule. She will also make decisions about what cases are included in the lawsuit and what evidence is allowed to be used at trial. Judge Pratter might be considered a blue-blood judge. She graduated from Stanford and received her law degree from the University of Pennsylvania. The University of Pennsylvania law school is currently ranked #4 in the United States. She was a partner in the law firm of Duane Morris from 1975, until her nomination to the federal bench by George W. Bush. She was confirmed by the Senate on June 15, 2004. She is a member of the adjunct faculty at the University of Pennsylvania. Her resume is that of an experienced and intelligent judge. We expect the Ozempic lawsuit to be one of the largest and most complex in the history of the United States. It is no mistake that the case was assigned to Judge Pratter. Local attorneys know her as a no-nonsense type. We expect Judge Pratter to be impressive this week at her first status conference in the Ozempic litigation and show the lawyers that she is going to run a tight ship.
March 2, 2024: Upcoming Ozempic Lawsuit News
There hasn’t been much news in the Ozempic lawsuit since it became official and was consolidated in the Eastern District of PA. We’ll get our initial look at how the Ozempic lawsuit is shaping up at the first status conference scheduled with Judge Gene E.K. Pratter on March 14, 2024. We continue to see Ozempic everywhere on TV and social media. You cannot go very long without being fed its so-called greatness on mainstream news or the countless influencers on Facebook, Instagram, and others. While it is portrayed as a miracle weight loss drug, there is the dark side of its serious side effects like stomach paralysis affecting thousands of people.
February 28, 2024: Behind the Scenes of the Ozempic Lawsuit
The Ozempic lawsuit is in the critical early stages of the Multidistrict Litigation. The politics of mass tort litigation is difficult to understand. Simply put, there are different factions in almost every case. Law firms want power and authority to control the decisions in the case because they have a lot of resources invested in getting the case to become an actual lawsuit. The Ozempic lawsuit highlights the differences between some of the “old guard” lawyers and some of the big firms that are entering mass torts. One big firm filed for consolidation of the Ozempic lawsuit, allegedly without much warning to other law firms. Other lawyers then were forced to rush to get cases filed and have the right to be heard at the JPML hearing. The responding lawyers asked for the case to be transferred to Philadelphia, and the panel granted their request. There is some conflict between law firms. At this point, we will see if the different factions can work together to propose a group of lawyers to drive the case forward for the next several years. The judge will appoint a lead counsel and plaintiffs’ leadership committee in the next several months. The group will then proceed with discovery and learn how Novo Nordisk developed Ozempic, Wegovy, and Rybelsus. Most importantly, we will learn what they knew about the risks when they failed to warn of stomach paralysis and intestinal blockage.
February 24, 2024: Ozempic Manufacturer Files Lawsuits Targeting Knockoffs
Ozempic Manufacturer Novo Nordisk is not only being sued in federal and state courts for injuries caused by Ozempic (semaglutide), they are filing their own lawsuits too. Novo Nordisk owns the patent for semaglutide, and no other manufacturer is allowed to produce it. The company has filed at least 12 lawsuits against entities they say are selling knockoff Ozempic. Novo Nordisk has aggressively targeted pharmacies and online drug sellers for “compounding.” Novo Nordisk maintains that they are the only company allowed to sell semaglutide and allege that they do not know what is contained in the compounded drugs being sold online. The company states that the compounded drugs are not FDA-approved.
February 23, 2024: Ozempic Manufacturer Has Faced Many Lawsuits
Ozempic manufacturer Novo Nordisk is no stranger to lawsuits. The company has recently been in the news because Ozempic lawsuits are popping up everywhere. Earlier this month the federal court cases were centralized in Philadelphia for efficiency. A new case filed in New Jersey State Court, Tinsley v. Novo Nordisk, also suggests that some lawyers think it will be advantageous to stay outside the class action-style lawsuit and proceed with a New Jersey state court Ozempic case. It is worth noting that the New Jersey case is a gallbladder case, and we believe gallbladder cases will not be part of the federal court lawsuit.
It should be noted that the United States Department of Justice filed a lawsuit against Novo Nordisk nearly 15 years ago for fraudulently and aggressively marketing their diabetes drugs. Novo Nordisk settled that case brought by Loretta Lynch, United States Attorney for the Eastern District of New York. In 2017, the State of California settled with the Ozempic manufacturer for promoting a type 2 diabetes drug outside of what it was approved to be used for. In that case, the company targeted children for the diabetes drug, even though the FDA had not approved the drug for use in children. Similarly, the current lawsuit alleges that Novo Nordisk aggressively marketed Ozempic using inappropriate tactics and has promoted a diabetes drug for a use inconsistent with FDA approval.
February 21, 2024: Ozempic Manufacturer Failed to Warn of Health Risks
The Ozempic lawsuit will be largely focused on what the manufacturer knew about the side effects of using Ozempic and when. Plaintiffs’ lawyers have brought the lawsuit. So, they will have the burden to prove that Novo Nordisk did something wrong. The most likely claim to be successful is a failure to warn of the dangers associated with the drug. Many lawsuits have been filed alleging that the manufacturer of Ozempic knew of the risk of gastroparesis, ileus, and bowel obstruction. The allegations also include aggressive and deceptive marketing. On October 5, 2023, an important study, published in JAMA and authored by Mohit Sodhi, indicated an increased risk of gastroparesis, bowel obstruction, and pancreatitis for those using GLP-1 agonists. Ozempic is the trade name for semaglutide. The study used a cohort of over 5,000 randomly selected individuals from the PharMetrics Plus for Academics Database of over 16 million prescription drug users in the United States. The Ozempic lawsuits began being filed around the United States shortly after this study. By December, an application was made to consolidate all Ozempic lawsuits in a federal class action type, called a Multidistrict Litigation (MDL). The application was granted, and we are in the early stages of the Ozempic MDL, centered in Philadelphia. The first status conference before the Judge is forthcoming. The first milestones in the case will be selecting lead counsel, filing a master complaint, and establishing a preliminary discovery schedule for document production.
February 16, 2024: First Status Conference set in Ozempic lawsuit.
Just a week after the Judicial Panel on Multidistrict Litigation created MDL 3094 (the official Ozempic Lawsuit) and transferred all cases to the Eastern District of Pennsylvania, the Hon. Gene E.K. Pratter has scheduled the first status conference for Thursday, March 14, 2024. Among the topics to be discussed are: Organization and Process for selecting plaintiff’s lead counsel, scheduling and frequency of future status conferences, filing procedures for motions, responses, complaints, and other documents, and the creation of plaintiff fact sheets. Perhaps the most important agenda item is in discussion on whether to keep this just an Ozempic case or to include other manufacturers such as those who made Rybelsus, Wegovy, Trulicity, and Mounjaro.
February 15, 2024: Ozempic Lawsuit Injuries Coming Into Focus
With the Ozempic lawsuit just weeks old, we are refining which injuries will likely be strong cases. It is important to understand that the basis of the lawsuit is the failure to warn about the potential for serious side effects caused by using Ozempic and similar drugs. There is solid support for gastroparesis, stomach paralysis, gastrointestinal obstruction, and ileus (bowel obstruction).
February 4, 2024: Ozempic Lawsuit becomes official
There is now an active Ozempic Multidistrict Litigation, as we expected. This is similar to an Ozempic Class Action Lawsuit. The case will be known as MDL 3094 In Re: Glucagon-Like Peptide-1 Receptor Agonists (GLP-1 RAs) Products Liability Litigation. The case will be heard in the Eastern District of Pennsylvania federal court. Hon. Gene E. K. Pratter will be the Judge. This venue is generally considered a plaintiff-friendly venue. There have recently been large verdicts in the Philadelphia area against Monsanto in the Roundup litigation. The other big question that was left open during the January 25th hearing was who would be included in the case. Eli Lilly, the manufacturer of Mounjaro, has been included in the Ozempic federal court lawsuit. Eli Lilly has done their best to distance themselves from Novo Nordisk. The Judicial Panel on Multidistrict Litigation (JPML) seems to have been conflicted on whether or not to include them. The order specifically states that if Judge Pratter decides it is not a good decision, he has options to separate the defendants. For now, Mounjaro is part of the Ozempic lawsuit. The first important question for Judge Pratter will be, who is going to lead this litigation for the plaintiffs’ side.
February 1, 2024: Decision on the Future of Ozempic Lawsuit Looms
The Judicial Panel on Multidistrict Litigation is set to rule on how the Ozempic Lawsuit will proceed. At hand remains the question as to whether the lawsuit will move forward to include only the Ozempic brand itself, which is manufactured by Novo Nordisk, or if other similar drugs such as Ely Lilly’s Mounjaro will become part of the suit. A decision from the JPML is expected in February.
January 25, 2024: Pivotal Day For Ozempic Lawsuit
The Ozempic lawsuit is at a critical stage today. The Judicial Panel on Multidistrict Litigation will hear arguments in Santa Barbara, CA, with respect to the application brought by plaintiffs to create a multidistrict litigation. It is nearly certain that they will grant the application because the Ozempic manufacturer does not object. There are two questions remaining, however. First, will drug manufacturer Ely Lilly be included in this case. They make the drug Mounjaro, which is similar but different from Ozempic. Some lawyers think it is better to keep them separate. Finally, where will the case be located? The application requested the Western District of Louisiana. There are also applications for the lawsuit to be based in federal district courts in California, North Carolina, and Philadelphia.
January 20, 2024: Manufacturers of Ozempic and Mounjaro take differing positions in Lawsuit
The future of the Ozempic/Mounjaro litigation is still undecided. After an application by plaintiffs to organize the case in a Multidistrict Litigation (MDL), the two largest defendants, Novo Nordisk and Eli Lilly, are split on their response to the application. Novo Nordisk, the manufacturer of Ozempic, has supported the application for consolidation but requests the case be heard in the Middle District of North Carolina or the Southern District of California. The Ozempic response was filed with the Panel on December 29, 2023. The different venues here are surprising. The middle of North Carolina is probably a play at a lower average income jury pool, as a way to limit exposure and perhaps a move towards more conservative jurors that would not be as likely to support drug use for weight loss. California seems like a risky request. California verdicts can be some of the highest in the country. The defense must feel that the jury pool would think that it was common knowledge there were risks associated with the use of Ozempic.
Eli Lilly, the pharma giant and manufacturer of Mounjaro has asked the JPML to deny the plaintiffs’ motion for consolidation of the lawsuits. The Ozempic manufacturer probably thinks it will be too difficult and expensive to defend cases in virtually every federal court in America. The Mounjaro manufacturer is likely trying to distance itself from Ozempic, which has been largely used and abused off-label.
January 14, 2024: Application Made to Organize Ozempic/Mounjaro Lawsuits Into a Class Action-Style MDL
A recent application by attorneys of those injured by the side effects of drugs like Ozempic and Mounjaro was submitted to the Judicial Panel for Multidistrict Litigation (JPML). The application asks the panel of judges to consider referring all Ozempic and similar lawsuits nationwide in a single federal district court. The benefits of consolidation into a multidistrict litigation (MDL) include efficiency in the pre-trial discovery process, cost-effectiveness for all parties, and fairness among all plaintiffs with similar injuries. An Ozempic MDL has similarities to an Ozempic class action lawsuit, although the main benefit for plaintiffs is that an MDL allows for individualized settlements based upon the severity of each person’s case. The application includes the drugs liraglutide, dulaglutide, semaglutide, and tirzepatide. Those drugs are marketed as Saxenda, Trulicity, Ozempic, Rybelsus, Wegovy, and Mounjaro. King Law is currently investigating cases where individuals used these drugs and were further diagnosed with gastroparesis, stomach paralysis, gastrointestinal obstruction, and ileus.
January 7, 2024: Ozempic lawsuits increase in frequency following a recent study
Published in The Journal of the American Medical Association (JAMA), the study found Ozempic and other drugs with Glucagon-Like Peptide-1 Receptor Agonists are linked to pancreatitis, bowel obstruction, and gastroparesis. The study by Mohit Sodhi and colleagues looked at a random sample of 16 million patients from 2006 to 2020. Ozempic lawsuits are popping up all over the country. The general claims are that Ozempic was not properly studied before its release, there were not proper warnings about the effects of Ozempic, and Ozempic is being used off-label. Off-label use of medical devices and drugs like Ozempic is a common allegation in a lawsuit. Drugs are studied and approved for certain uses. In the case of Ozempic, the medication is approved for the treatment of type 2 diabetes. We anticipate much of the Ozempic litigation will center around the marketing practices that encourage use for weight loss, instead of what the FDA said the drug could be safely used for.
November 18, 2023: Ozempic Lawsuit Allegations Grow Internationally
In addition to a possible class action-type lawsuit in the United States against the manufacturers of the Diabetes drug Ozempic, a Canadian law firm has now filed a similar suit in British Columbia. The lawsuit alleges the same type of failure to warn of side effects such as Gastroparesis, Stomach Paralysis, Ileus, Gallbladder issues, and Gastric Intestinal Obstruction.
November 18, 2023: Ozempic Class Action MDL Lawsuit Could Come Summer 2024
A Class Action lawsuit could come as early as summer 2024 against the manufacturer of the popular drug Ozempic. The allegations are that the drug, which helps regulate type 2 diabetes and assist in weight loss, causes severe gastrointestinal problems. Those who have experienced these injuries claim that pharmaceutical companies Novo Nordisk and Eli Lilly failed to warn patients of the adverse side effects from Ozempic (semaglutide).
October 2023: JAMA Research Letter Warns of Possible Ozempic Risks
A research letter filed in the Journal of the American Medical Association alleges the risk of adverse gastrointestinal events associated with the use of GLP-1 agonists that are used not only to treat diabetes but also off-label for weight loss. The research concludes that there is an increased risk of pancreatitis, gastroparesis, and bowel obstruction with the use of the drugs.
August 2023: Lawsuits Begin To Be Filed By People Harmed by Ozempic
The first lawsuit is filed against the makers of Ozempic and its sister drug Wegovy alleges a failure to adequately warn consumers about the potential risk of harm associated with using the drugs. The Ozempic lawsuit, filed against pharmaceutical giants Novo Nordisk and Eli Lilly, alleged that the drugs, both Glucagon-like peptide 1 (GLP-1) agonists, caused severe gastrointestinal issues.
King Law Featured In Ozempic Litigation News Coverage
King Law closely follows the Ozempic litigation and has been featured in news coverage about Ozempic cases. For example, the firm was featured in a New York Post story about one of King Law’s Ozempic clients who lost his vision shortly after starting Ozempic. King Law focuses its practice on mass torts, including drugs and medical devices that are alleged to have caused mass injuries. We have helped many clients secure favorable outcomes in mass tort lawsuits.
About King Law
King Law is a reputable personal injury and mass tort firm. Our team is client-focused, and we are trained to help people throughout complex civil lawsuits, like those filed about Ozempic. We put our clients first in every aspect of our representation by seeking to understand their lived experiences and crafting a legal strategy that can lead to compensation. Our firm has nationwide reach, meaning we can represent clients across the United States.
Why Are Lawsuits Being Filed Against Ozempic?
Ozempic patients are suing Novo Nordisk because they experienced severe complications that they believe they should have been properly warned about. According to legal filings and public reports, the severe side effects of Ozempic that were not properly addressed on past warning labels include:
- Vision loss (caused by NAION, a type of eye stroke)
- Gastrointestinal issues (e.g., ileus, gastroparesis, and bowel obstructions)
- Gallbladder problems
- Pancreatitis
- Blood clots and deep vein thrombosis
Multiple scientific studies have suggested that these side effects, while relatively uncommon, may be more likely to occur if a patient is taking Ozempic or another GLP-1 drug compared to some other common diabetes drugs. Legal complaints accuse Novo Nordisk of failing to disclose these risks to Ozempic patients and doctors. Lawsuits also allege that Novo Nordisk was not transparent about the safety of Ozempic.

What Are The Key Allegations In Ozempic Lawsuits?
Some of the key allegations in the Ozempic lawsuits include the following:
- Failure to warn and inadequate labeling: Lawsuits contain allegations that Novo Nordisk did not adequately warn Ozempic patients about some of the severe gastrointestinal and vision complications they might experience.
- Negligent misrepresentation: Lawsuits accuse Novo Nordisk of overstating the potential benefits of Ozempic while minimizing or obscuring some of its risks.
- Defective design of Ozempic: Some lawsuits allege that Ozempic’s design is unsafe and puts people at a higher risk of complications than necessary, given the drug’s purpose.
- Negligence and gross negligence: Lawsuits allege that Ozempic’s manufacturer was negligent in how it marketed Ozempic and how it described its potential risks to consumers and doctors.
At their core, these lawsuits accuse Novo Nordisk of misleading and hiding information from patients about what could happen if they take Ozempic. Patients were prescribed Ozempic to help them manage their type 2 diabetes. Many of the patients filing lawsuits say that, had they known about the scope of Ozempic’s risks, they may not have taken Ozempic.

Why Are There Two Ozempic MDLs?
There are two Ozempic MDLs because the injuries alleged in lawsuits are very different from each other. One MDL focuses mostly on injuries to the stomach, intestines, and other organs in the abdomen. The other MDL focuses on a specific injury to the eye. The JPML (courts) decided to make two separate litigations because the injuries involved will require different medical evidence, experts, and causation theories.
The two Ozempic MDLs were created to help manage the two groups of claims that people have been filing. The first Ozempic MDL (MDL No. 3094) was created in February 2024 and is about gastrointestinal complications (e.g., ileus, pancreatitis, and gastroparesis) that the drug allegedly caused. Judge Marston oversees the MDL in the Eastern District of Pennsylvania.
The second Ozempic MDL (MDL No. 3163) was created in December 2025 for claims that Ozempic use caused NAION (an eye stroke that can cause vision loss). Judge Marston also oversees the Ozempic vision loss MDL in the Eastern District of Pennsylvania. The NAION MDL for Ozempic was created because many people were filing NAION and vision loss claims, and that injury was largely different from the injuries alleged in the first MDL. The courts believed that making an MDL for these Ozempic claims could streamline the litigation process.
Ozempic Safety Timeline: FDA Warnings, Studies, And Lawsuits
December 2025: Multidistrict litigation was formed for claims that Ozempic caused patients to develop NAION (non-arteritic anterior ischemic optic neuropathy). At the time the MDL was formed, at least 30 Ozempic vision-loss claims were consolidated into the federal group lawsuit. Sources indicate that at least 40 more Ozempic NAION cases were pending in state courts, too.
May 2025: Post-marketing research suggests that GLP-1s like Ozempic are associated with an increased risk of deep vein thrombosis (a type of blood clot). According to an observational study, patients who took Ozempic or another GLP-1 were 1.65 times more likely to develop deep vein thrombosis than patients in the control groups. That same study suggested that taking a GLP-1 for 1.5 years or more was associated with a 2.32 times higher chance of a deep vein thrombosis.
August 2024: A study (Risk of Nonarteritic Anterior Ischemic Optic Neuropathy in Patients Prescribed Semaglutide) is published and suggests a link between taking a semaglutide like Ozempic and an increased risk of developing NAION. The study found that participants with Type 2 diabetes who were prescribed semaglutide were 4.28 times more likely to develop NAION. Participants who took semaglutide for obesity and weight loss were 7.62 times more likely to develop NAION. The study’s authors said more research is needed to fully understand and confirm the risk of NAION among people taking semaglutide drugs.
February 2024: The United States Judicial Panel on Multidistrict Litigation approved the creation of a federal MDL to consolidate the gastrointestinal claims against Ozempic and other GLP-1 drugs. The MDL was consolidated in the federal Eastern District of Pennsylvania and was overseen by Judge Pratter until her passing. Judge Marston has taken her place and is currently presiding over the Ozempic gastrointestinal lawsuits.
October 2023: Researchers conducted a large observational study of about 5,400 patients who took semaglutide, liraglutide, or bupropion-naltrexone for weight loss. Previous studies found that patients who took GLP-1s for diabetes had a higher likelihood of experiencing biliary (gallbladder) diseases, pancreatic issues, bowel obstructions, and gastroparesis (stomach paralysis). The researchers wanted to determine the risk ratios for patients who took these medications for weight loss. According to the results, taking a GLP-1 (such as Ozempic) for weight loss was associated with a higher risk of pancreatitis, bowel obstruction, and gastroparesis than bupropion-naltrexone.
2022: An analysis called Gastrointestinal adverse events associated with semaglutide: A pharmacovigilance study based on FDA adverse event reporting system describes post-marketing adverse events for the first three years that semaglutide (Ozempic) was on the market. During that time period, around 5,400 patients reported about 11,300 adverse events. Of those, 45 different categories of gastrointestinal issues were reported, including pancreatitis, constipation, nausea, vomiting, and diarrhea. A number of these adverse events resulted in hospital stays and, rarely, death.
2017: The FDA approves Novo Nordisk’s Ozempic (semaglutide, a glucagon-like peptide-1 receptor agonist) to treat type 2 diabetes in eligible patients. At the time, Eli Lilly’s Trulicity was the only GLP-1 on the market. In its approval notice, the FDA says that it will require Novo Nordisk to conduct additional postmarketing safety tests to confirm Ozempic’s safety and efficacy. It is common for the FDA to require drug companies to conduct these types of post-marketing studies.
Note: The studies listed above show associations, not definitive proof of causation. The courts and juries will weigh in on causation based on scientific evidence provided by experts and evidence presented in court proceedings.
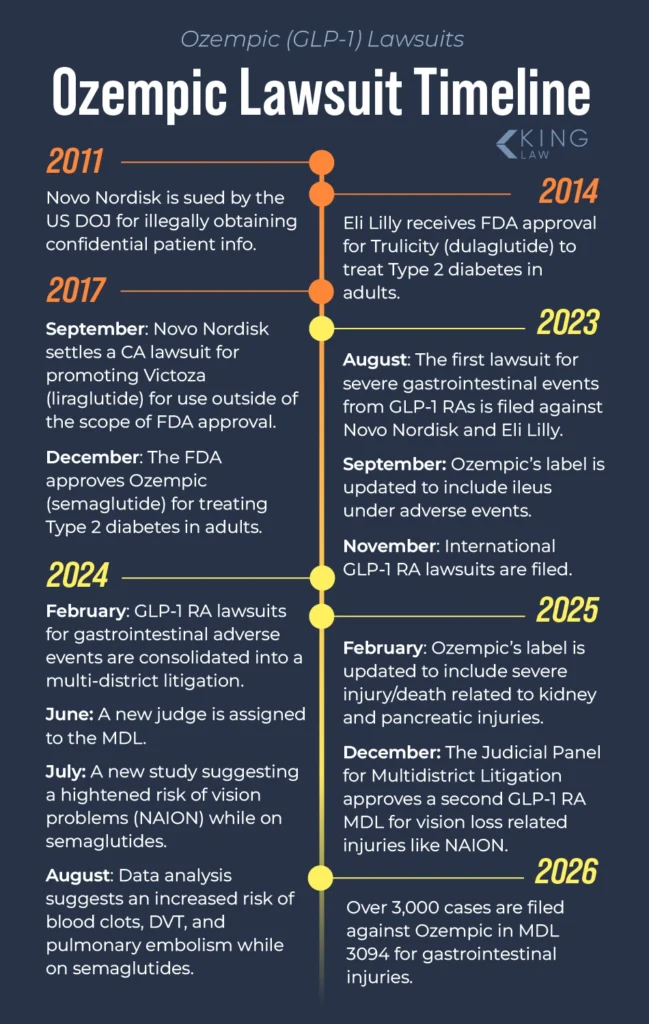
Past Important Dates In Ozempic Litigations
Here are some of the important milestones for the Ozempic litigation:
- August 2, 2023: One of the first Ozempic lawsuits was filed in Louisiana. The lawsuit also alleged injuries from Mounjaro. It listed Novo Nordisk and Eli Lilly as defendants.
- December 15, 2023: Another Ozempic lawsuit was filed in the Eastern District of Pennsylvania.
- February 2, 2024: The Ozempic MDL was created and is overseen by Judge Pratter until Judge Marston took over the case. 18 lawsuits were originally consolidated into the Ozempic MDL.
- March 14, 2024: First case management conference held in the first Ozempic MDL.
- May 17, 2024: Judge Pratter passes away.
- June 6, 2024: Judge Marston is assigned the Ozempic MDL after Judge Pratter’s passing.
- June 14, 2024: Original Science Day date, as scheduled by Judge Pratter. This date was later rescheduled.
- September 4, 2024: Science Day held in the Ozempic lawsuit. This proceeding allows experts for both parties to explain the science behind allegations in the lawsuits.
- August 2025: Judge Marston enters an order requiring gastroparesis claims to be supported by a gastric emptying study diagnostic test performed at the time the patient experienced symptoms.
- December 2025: JPML creates MDL for the Ozempic NAION vision loss claims.
Is There A Class Action Lawsuit Against Ozempic?
There is no single, nationwide class action lawsuit against Ozempic. Instead, Ozempic lawsuits are being filed individually in state and federal courts as product liability and wrongful death claims. However, many of these individual claims have been consolidated into two separate multidistrict litigations (MDLs) against Ozempic.
An MDL allows many Ozempic patients to coordinate parts of their lawsuits, such as discovery, trial preparation, and settlement negotiations. However, people who join the MDL can still take their case to trial and seek compensation for their individual losses from Ozempic.
What Conditions May Qualify Someone For An Ozempic Lawsuit?
Different law firms have different criteria for Ozempic lawsuits. In order to file with King Law, here are the conditions that may qualify someone for an Ozempic lawsuit:
- Deep vein thrombosis (DVT)
- Gallbladder cancer
- Gallbladder removal
- Gastroparesis (must have had a gastric emptying study to diagnose)
- Ileus/bowel obstruction (w/surgery)
- Necrotizing pancreatitis
- Non-arteritic anterior ischemic optic neuropathy (NAION)
- Pancreatic cancer
- Pulmonary aspiration
- Pulmonary embolism
We encourage you to contact our office if you took Ozempic and experienced complications like those listed above. You may still be eligible for representation with our firm, even if your Ozempic-related injury is not listed above. Our attorneys can investigate your case and file a lawsuit if we believe your diagnosis is related to Ozempic. Our intake team can review your medical history at no cost and tell you whether your case meets current MDL requirements.
What Doctors And Attorneys Are Saying About Ozempic Litigation And Injuries
Doctors are expressing caution and concern about Ozempic and the potential side effects. For example, David Kessler, MD, who used to run the FDA, wrote an article about GLP-1 drugs for Harvard Magazine. In that article, he wrote that delayed gastric emptying (the digestive system holding food in the stomach for longer) is the likely main way that GLP-1s like Ozempic reduce overeating.
By keeping food in their stomach longer, the person feels full sooner and for longer, which makes them less likely to overeat. Kessler also wrote that obesity is a metabolic disorder that causes the body to create too much fat tissue. Kessler suggests that medications like Ozempic promise long-term weight-loss benefits, but they may not address the underlying metabolic disorder that may be driving the person’s excess weight.
Attorneys are also concerned about how Ozempic has been marketed, prescribed, and portrayed to the public. For example, legal filings accuse Novo Nordisk of downplaying and hiding the truth about the gastrointestinal risks people could experience after taking Ozempic. Legal complaints also allege that Ozempic’s marketing materials may mislead the public into thinking that Ozempic patients will keep the weight off if they stop taking the medication.
However, a July 2025 study published in eClinicalMedicine notes that people who stop taking Ozempic and other GLP-1s tend to regain the weight they had lost. In that study, patients taking a semaglutide drug like Ozempic regained 68% of the weight lost after 1 year off Ozempic. That same study noted that taking a GLP-1 was associated with a higher rate of gastrointestinal side effects, pancreatitis, and pancreatic cancers, though other studies have not confirmed this.
Ozempic Lawsuits Involving Vision Loss, Blindness, And NAION
Patients are suing Novo Nordisk because they partially lost their vision after taking Ozempic. They believe that Ozempic caused them to experience an eye stroke (called NAION) in the vessels that supply blood to the optic nerve. NAION (non-arteritic anterior ischemic optic neuropathy) can cause sudden, painless, and irreversible blurry vision or blindness, usually in one eye.
A July 2024 study found that people who were taking semaglutide were about 4 times more likely to develop NAION compared to the control group. Many of the patients who have experienced blindness after taking Ozempic have said that they wish they had been told that this could occur.
About NAION:
- Vision loss in one or both eyes
- Blurry vision or dark clouds in the visual field
- Painless
- Permanent
- No treatment for NAION-related vision loss
Source: Brigham and Women's Hospital
Injuries In Ozempic Stomach Paralysis Litigation
As of February 2026, Ozempic’s warning label warns of “severe gastrointestinal adverse reactions” for some patients. However, lawsuits say these warnings were not and continue to be inadequate to properly inform patients of potential side effects.
Ozempic patients are suing Novo Nordisk after they experienced a range of stomach paralysis and gastrointestinal complications. Here is a list of some of the injuries listed in Ozempic lawsuits and scientific studies that researched those injuries in people who take GLP-1 drugs:
- Ileus: Intestinal blockages were 3.5 times more likely in patients taking Ozempic or another GLP-1RA, according to one study. (Source)
- Gastroparesis: Slowing or stopping of the gut (also called delayed gastric emptying) was between 3 to 6 times more likely among people who took Ozempic or other GLP-1s, one study suggested. (Source)
- Gallbladder problems: One survey study found that patients who took a semaglutide, like Ozempic, were significantly more likely to experience pancreatitis, ileus, and gastroparesis than the control group. (Source)
- Pancreatitis: Studies suggest that Ozempic use has been associated with an increase in reports of acute pancreatitis, which can be fatal in some cases. (Source)
- Intestinal obstructions: One study found that participants who took an incretin-based drug (e.g, a GLP-1 like Ozempic) were over 4.5 times more likely to report having an intestinal obstruction than those taking other diabetes medications. (Source)
In their lawsuits, some Ozempic patients say they were not adequately warned about these risks. Some Ozempic patients who experienced these complications had to be hospitalized to treat these conditions. Other Ozempic patients have suffered long-term impairments because of these stomach injuries.
Stomach-related symptoms of Ozempic:
- Ileus (the bowel muscles stop working and do not move food through the system)
- Acute gallbladder disease
- Gallbladder problems
- Pancreatitis
- Intestinal obstruction
- Nausea
- Vomiting
- Diarrhea
Source: FDA
King Law Criteria To File An Ozempic Lawsuit
King Law’s eligibility criteria for filing an Ozempic lawsuit include the following:
- The client used Ozempic or another GLP-1 drug
- The client developed a complication that research has shown may be a result of Ozempic use
- The client has seen a medical professional to diagnose and treat their injury
- The client’s doctors have formally diagnosed them with a condition that lawsuits allege is related to Ozempic use
- The client had to undergo treatment to address the condition
These criteria have changed over time as King Law has learned more about the complications that Ozempic patients might have experienced. The judges who have overseen the two Ozempic MDLs have also issued updated requirements for medical evidence that people must provide for their cases. The most effective way to find out if you qualify to file an Ozempic lawsuit is to contact our office directly.
What Injuries May Qualify Someone For An Ozempic Lawsuit?
King Law is currently investigating claims from people who experienced the following conditions:
- NAION
- Deep vein thrombosis (DVT)
- Gallbladder cancer
- Gallbladder removal (prior to March 2022)
- Gastroparesis (requires a gastric emptying study)
- Ileus/bowel obstruction (w/surgery)
- Necrotizing pancreatitis
- Pancreatic cancer
- Pulmonary aspiration
- Pulmonary embolism
However, these conditions are not exclusive to the MDLs. King Law will examine the information you provide you with possible legal options, including an MDL if there is one for your specific injury.
Typically, patients would have to have formal diagnostic tests to prove that they experienced one of these complications from Ozempic. For example, as of August 2025, Ozempic patients filing a gastroparesis claim must have a gastric emptying study performed.
The vision loss MDL for Ozempic patients is new, so the filing requirements could change in the future. An Ozempic attorney on our team can help you understand what the updated criteria are and if you meet those after experiencing vision loss after taking a GLP-1 drug.
Questions Your Attorney May Ask When You Want To File An Ozempic Lawsuit?
Some of the factors that may help strengthen your Ozempic claim with King Law include the following:
- Did you take Ozempic for at least 3 months?
- Was your condition diagnosed by a specialist, such as a gastroenterologist or ophthalmologist?
- Did your diagnostic team perform imaging studies and endoscopies (where appropriate)?
- Did you have a history of gastroparesis?
- Did your doctor perform a gastric emptying diagnostic study?
- Did your symptoms improve after you stopped taking Ozempic?
Some of the above tips for strengthening Ozempic cases do not apply to all claims. For example, an Ozempic patient who developed NAION and lost their vision in one or both eyes could still file a lawsuit even if their vision did not improve after they stopped taking Ozempic. Collaborate with a seasoned Ozempic attorney to find ways to strengthen your specific claim.
What Medical Evidence Can Your Attorney Use To Build Your Ozempic Claim?
Medical records that matter when crafting your Ozempic claim depend on the type of complication you experienced. Generally, Ozempic claims fall into four categories: gastrointestinal issues, blood clots, aspiration, and vision loss.
For gastrointestinal claims, medical records that you might need include the following:
- Gastric emptying study (required for gastroparesis claims)
- Endoscopy
- Colonoscopy
- Intestinal imaging studies
- GI specialist notes
- ER records
If you want to file an Ozempic vision loss claim, here are some of the medical documents you might want to locate:
- OCT imaging
- Visual field testing
- Ophthalmology diagnosis
- NAION confirmation
Finally, Ozempic patients who experienced blood clots may use the following records to support their claims:
- CT angiogram
- Doppler ultrasound
- Hematology reports
Keep in mind that the judge could issue orders that change what proof someone needs to file a case. For example, in August 2025, the Ozempic MDL judge entered an order saying that patients filing a gastroparesis claim must prove that their doctors used a gastric emptying study to diagnose them with gastroparesis. Requirements for other Ozempic-related claims may also change. For that reason, consulting an attorney is a good idea to make sure you understand and can protect your legal rights.
Why Does Medical Proof Matter In An Ozempic Lawsuit?
Medical proof is important in any lawsuit alleging injuries from a drug. In an Ozempic lawsuit, having a qualifying diagnosis and tests supporting that diagnosis can help your attorney build a strong case. People with strong, legally defensible cases may have a better chance of securing compensation.
What Are The Recoverable Damages In The Ozempic Lawsuit?
Ozempic patients may seek compensation for losses they incurred due to Ozempic-related complications. For example, this could include:
- Reimbursement for lost wages and paid or unpaid time off
- Diagnostic tests
- Medical bills
- Surgeries or other treatment
- Prescriptions/medication
People who file successful lawsuits may also qualify for pain and suffering, loss of quality of life, punitive damages, and other forms of compensation. Compensation packages in Ozempic cases might cover past and future medical expenses and lost wages. However, each Ozempic settlement is calculated based on that specific patient’s losses.
What Is The Deadline To File An Ozempic Lawsuit?
The deadline to file an Ozempic lawsuit depends on the applicable statute of limitations. Statutes of limitations are determined by state law, and these rules change based on the type of case the person is filing. The most reliable way to discover the filing deadline for your Ozempic case is to call a reputable attorney.
How Can I File An Ozempic Lawsuit?
The steps to file an Ozempic lawsuit include the following:
- Contact a reputable law firm and speak with its intake team.
- Retain an attorney to represent you during your Ozempic lawsuit.
- Help your attorney locate and gather your Ozempic-related medical records.
- Your attorney writes and files a legal complaint against Ozempic’s manufacturer, Novo Nordisk.
- Your attorney files or transfers your Ozempic case into the applicable MDL or files a claim outside of the MDL.
- Your attorney requests evidence during the discovery process of your Ozempic case.
- Your Ozempic attorney creates a strong settlement negotiation strategy.
- If your attorney cannot secure a fair out-of-court settlement, they prepare to take your Ozempic case to trial.
Teaming up with a reputable attorney is beneficial because trained lawyers understand how Ozempic lawsuits work. They know what to expect during the lawsuit process and can prepare for each step along the way. They can also track the Ozempic court filings and latest legal analysis to help craft an effective strategy for your case.
How Much Is The Ozempic Lawsuit Going To Payout?
The Ozempic lawsuit is ongoing, so there is no exact or guaranteed payout amount. However, our legal partners estimate that some Ozempic patients could receive significant compensation in severe cases. However, this is a speculative estimate based on severe injuries in other pharmaceutical litigations. Plaintiffs are not guaranteed any amount of compensation from a lawsuit.
If a plaintiff is offered a settlement from Novo Nordisk, the final settlement value of their Ozempic lawsuit depends on the person’s damages, including their medical bills, injury severity, and likelihood of fully recovering from those injuries. Consult an attorney to find out what your settlement package might be if you file an Ozempic lawsuit.
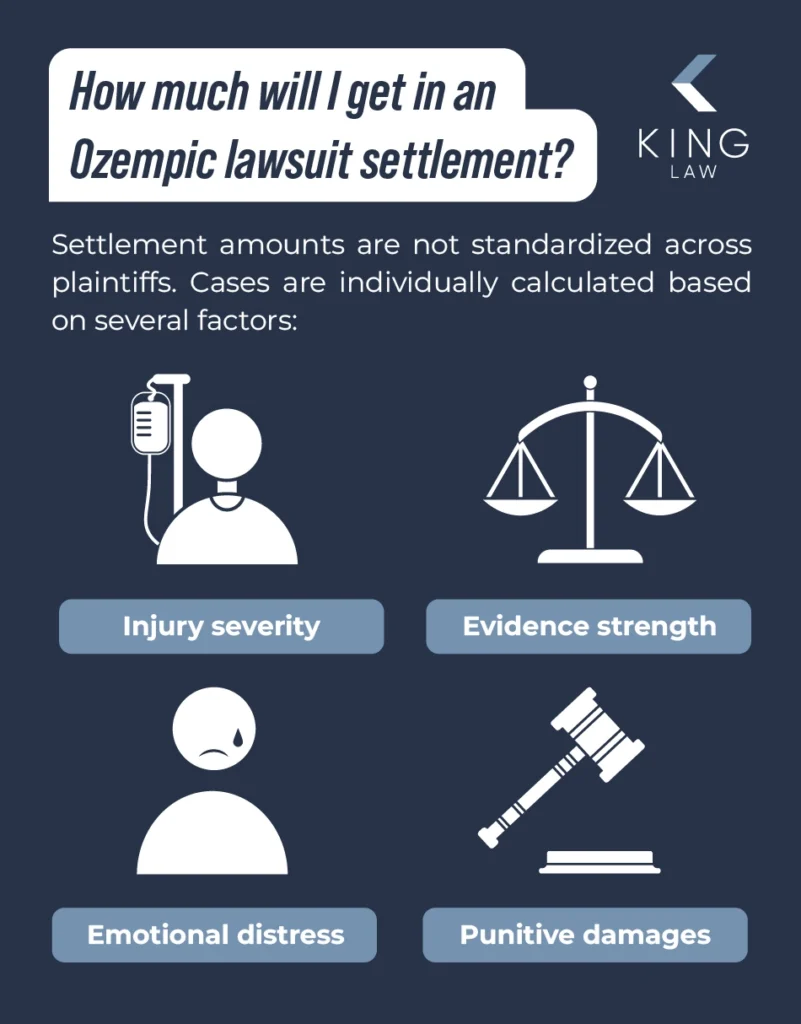
When Will The Ozempic Lawsuit Be Settled?
There is no set timeline for when Ozempic lawsuits settle or when a global settlement in the MDLs might occur. The Ozempic MDLs are still in the discovery phases, and there have not been any trials yet.
Some legal scholars think settlement negotiations for the Ozempic cases could begin in 2027. However, this is purely speculative. Currently, Bellwether trials are expected to begin in late 2026 or early 2027, but these dates may change. An attorney can help you stay informed about the status of the Ozempic lawsuits and how you can protect your rights.
What Factors Might Ozempic Settlements Depend On?
There are many factors that could impact someone’s Ozempic settlement. For example, having a strong Ozempic case supported by reliable, relevant evidence can help someone increase their chances of receiving a settlement. Additionally, someone who experienced a permanent loss or was hospitalized may qualify for a higher payout.
Proof of lost income or earning potential due to the Ozempic-related complication may also influence the person’s settlement value. If someone has a history of stomach-related problems prior to taking Ozempic, this may make it harder for them to prove their case. However, an attorney can still help them seek the compensation they deserve for their Ozempic-related losses.
Find a way to convert the image below into snappy text or a bulleted list.
King Law Is Investigating Ozempic Claims Related To Severe Side Effects Allegedly Caused By Ozempic
King Law is actively investigating Ozempic claims being brought by people who experienced severe side effects after taking this semaglutide drug. We have extensive experience fighting for patients who think they have been harmed by pharmaceutical drugs, like Ozempic. We are prepared to take steps to seek justice on your behalf.
Our Ozempic Legal Team At King Law
Contact An Ozempic Lawyer Today
If you suffered from gastroparesis, blood clots, vision loss, or a condition listed above after taking Ozempic, call our firm today at [phone] to talk to our intake specialists. We may be able to help you secure compensation against Novo Nordisk. We provide free case reviews, and we do not charge any upfront legal fees to meet with or hire us. You only pay our firm if we are able to secure compensation for you. Contact us to learn more.
Frequently Asked Questions (FAQs)
List Of Sources Used In This Article
American Heart Association Journals. “Glucagon-Like Peptide-1 Receptor Agonists and Cardiovascular Outcomes.” Journal of the American Heart Association, 2024.
Brigham and Women’s Hospital. “Non-Arteritic Anterior Ischemic Optic Neuropathy (NAION).” Brigham and Women’s Hospital.
BMJ. “Glucagon-Like Peptide-1 Receptor Agonists and Adverse Events.” BMJ, vol. 392, 2024.
Food and Drug Administration. “FDA Approves Novo Nordisk’s Ozempic for Type 2 Diabetes.” U.S. Food and Drug Administration, 2017.
Food and Drug Administration. “Ozempic (Semaglutide) Prescribing Information.” U.S. Food and Drug Administration, 2025.
Government of the United Kingdom. “Semaglutide (Wegovy, Ozempic, and Rybelsus): Risk of Non-Arteritic Anterior Ischemic Optic Neuropathy.” Drug Safety Update.
Harvard Medical School. “What Doctors and Patients Should Know About GLP-1 Drugs.” Harvard Medicine Magazine.
Journal of the American Medical Association. “Association of GLP-1 Receptor Agonists With Gastrointestinal Adverse Events.” JAMA, 2023.
JAMA Ophthalmology. “Semaglutide and Risk of Nonarteritic Anterior Ischemic Optic Neuropathy.” JAMA Ophthalmology, 2024.
Judicial Panel on Multidistrict Litigation. “Transfer Order for In re: GLP-1 Receptor Agonists Products Liability Litigation (MDL No. 3094).” United States Courts, 2024.
PubMed Central. “Adverse Gastrointestinal Effects of GLP-1 Receptor Agonists.” National Library of Medicine.
PubMed Central. “Glucagon-Like Peptide-1 Receptor Agonists and Optic Nerve Ischemia.” National Library of Medicine.
PubMed Central. “Ocular Complications Associated With GLP-1 Receptor Agonists.” National Library of Medicine.
PubMed Central. “Emerging Safety Signals for Semaglutide and Vision Loss.” National Library of Medicine.
Reuters. “FDA Approves Novo Nordisk Diabetes Drug Ozempic.” Reuters, 5 Dec. 2017.
Reuters. “Lawsuits Claiming Ozempic and Other GLP-1 Drugs Led to Blindness Become Second Mass Tort.” Reuters, 15 Dec. 2025.
Reuters. “Ozempic Litigation in Limbo After Judge’s Death.” Reuters, 6 June 2024.
U.S. District Court for the Eastern District of Pennsylvania. “In re: GLP-1 Receptor Agonists Products Liability Litigation (MDL No. 3094).” United States Courts.
U.S. District Court for the Eastern District of Pennsylvania. “In re: GLP-1 Receptor Agonists – NAION Litigation (MDL No. 3163).” United States Courts.
U.S. District Court for the Eastern District of Pennsylvania. “Judge Gene E.K. Pratter – Biographical Profile.” United States Courts.





