See if You Qualify

The Wegovy pill is an oral GLP-1 (semaglutide) drug used for weight loss that is new to the market. The recently approved pill has drawn legal scrutiny as patients experience severe side effects. Lawsuits involving the Wegovy pill raise new questions about how its risks were studied and how those risks were disclosed and communicated to patients.
People who are filing Wegovy pill lawsuits allege that the drug’s manufacturer, Novo Nordisk, did not adequately warn them about some potential side effects. Prescribing information and warnings for the Wegovy pill and injection are on the same drug pamphlet. This means patients and doctors may have to parse data, leaving many patients wondering if they were properly informed about the possible risks of the Wegovy pill.
People who took the Wegovy pill and believe they suffered from serious complications can talk to a qualified lawyer about filing a lawsuit. They may be eligible to file a legal claim against the Wegovy pill’s manufacturer, Novo Nordisk.
About the Wegovy Pill Lawsuit
Wegovy Pill Lawsuit Updates and Case Developments
Why Is the Wegovy Pill Facing Legal Scrutiny?
What Legal Claims Are Being Investigated Against Novo Nordisk?
What Medical Conditions Are Being Reported After Taking the Wegovy Pill?
How Does Oral Semaglutide Affect the Body?
What Makes the Wegovy Pill Different From the Injection?
What Warnings Are Provided About the Wegovy Pill?
Could the Wegovy Pill Cause Long-Term or Permanent Damage?
Who May Be Eligible to File a Wegovy Pill Lawsuit?
What Types of Compensation May Be Available in Wegovy Pill Cases?
Are Wegovy Pill Lawsuits Being Consolidated Into an MDL?
How Does the Wegovy Pill Compare to Other GLP-1 Lawsuits?
What Steps Should I Take to File a Wegovy Pill Lawsuit?
How Long Do I Have to File a Wegovy Pill Lawsuit?
Wegovy Pill Lawsuit Settlements and Payout Amounts
King Law Is Investigating Wegovy Pill Lawsuits
Contact a Wegovy Pill Lawyer Today
Wegovy Pill Lawsuit Updates and Case Developments
The updates below explore the regulatory, medical, and legal developments involving the new Wegovy weight-loss pill made by Novo Nordisk. This page focuses specifically on lawsuits involving the oral Wegovy pill. For information about lawsuits involving the injectable version, see our Wegovy lawsuit page.
April 2, 2026: Novo Nordisk Says Trial Data Shows Wegovy Pill Outperforms Orforglipron (Foundayo)
According to Novo Nordisk, the maker of the Wegovy pill, its drug adoutperforms the active ingredient (i.e., orforglipron) in Eli Lilly’s Foundayo. These two weight-loss pills are battling for market share. According to Novo Nordisk, people who took the Wegovy pill lost more weight and were more likely to stay on the drug than people taking orforglipron. This information was gathered as part of the OASIS 4 and ATTAIN-1 clinical trials.
February 5, 2026: FDA Says Some Wegovy Pill Advertisements Are Misleading
The FDA has issued a warning letter to Novo Nordisk after it determined that the company’s January 2026 Wegovy pill TV advertisements were misleading. According to the February 5, 2026, notice, the FDA believed that some of the statements in the TV advertisement could lead consumers to believe that the Wegovy pill is safer and more effective than other weight-loss drugs. Novo Nordisk is facing dozens of lawsuits filed by patients who claim they took a GLP-1 and suffered from side effects they do not believe they were warned about.
January 5, 2026: Wegovy Pill Officially Enters U.S. Pharmacies
Novo Nordisk’s new Wegovy pill became available in U.S. pharmacies on January 5, 2026, weeks after its approval was announced. The Wegovy pill will first be offered in doses of 1.5 mg, 4 mg, 9 mg, and 25 mg. The Wegovy pill may be taken once a day. The starting price for the Wegovy pill for some patients will be around $149. However, Novo Nordisk has said it plans to increase this price in mid-April 2026. Some patients who took the Wegovy injections have filed lawsuits after they allegedly suffered from complications they say they were not told could happen. People who take the Wegovy pill may also qualify to file a legal claim if they experience severe complications.
December 22, 2025: Novo Nordisk Announces FDA Approval of New Wegovy Weight-Loss Pill
Novo Nordisk has announced that the FDA approved its new Wegovy pill for weight loss and other limited purposes. Novo Nordisk manufactures the injectable GLP-1s, such as Wegovy and Ozempic, as well as one other GLP-1 pill (Rybelsus). However, according to Novo Nordisk, the Wegovy pill is the first oral GLP-1 approved for weight loss. The company says the clinical trial of the Wegovy pill (a semaglutide) showed that the Wegovy pill’s safety was “comparable to previous trials with semaglutide for weight management.”
Why Is the Wegovy Pill Facing Legal Scrutiny?
The Wegovy pill is facing legal scrutiny because the GLP-1 drug can cause people to experience severe side effects, and patients question whether or not they were properly warned. The Wegovy pill was the first semaglutide weight-loss pill introduced to the U.S. market. It was available before Eli Lilly’s Foundayo, another semaglutide weight-loss tablet.
Oral semaglutide absorption can vary based on patient behavior and physiology. So, some medical professionals and attorneys are examining whether this variability could affect how risks are experienced or identified. Additionally, some medical and legal experts are concerned that there could be implications associated with the race to be “first to market.” Those implications could possibly affect patients who take the Wegovy pill.
What Legal Claims Are Being Investigated Against Novo Nordisk?
Some Wegovy pill lawsuits may accuse Novo Nordisk of failing to warn people about the severe risks that semaglutide may pose. Under regulatory law, Novo Nordisk has a responsibility to be transparent with the FDA, prescribers, and consumers about the Wegovy pill’s severe risks and potential benefits.
If Novo Nordisk does not follow the laws about disclosing the Wegovy pill risks, it may be sued. Other legal claims that may be brought include allegations that the Wegovy pill was defectively designed and that Novo Nordisk misrepresented the safety profile of its oral semaglutide.
What Medical Conditions Are Being Reported After Taking the Wegovy Pill?
Currently, the pill and injectable versions of Wegovy share the same prescribing information and label, with some noted differences for the tablet version. Some people who have taken Wegovy (semaglutide) pills have reported severe complications that they say they were not warned about.
Possible complications associated with taking the oral Wegovy tablet that could be cited in lawsuits include:
- Gastroparesis (stomach paralysis)
- Ileus or bowel obstruction
- Acute and necrotizing pancreatitis
- Gallbladder problems (including gallbladder disease and removal)
- Deep vein thrombosis (DVT)
- Pulmonary embolism
- Pulmonary aspiration
- Acute kidney injury
According to legal documents and public reports, some of the people who have experienced these adverse events required surgery, hospitalization, or long-term treatments. Others may have died from complications they allegedly suffered after taking Wegovy.
How Does Oral Semaglutide Affect the Body?
Oral semaglutide in the Wegovy pill activates GLP-1 receptors in the body, which is one of the ways humans feel hunger or fullness. GLP-1 is a hormone that is produced naturally in the body, and it also slows down gastric emptying (the movement of food from the stomach to the intestines).
Introducing a humanmade GLP-1 (semaglutide) into the body may disrupt how the body normally empties the stomach and digests food. Some studies have suggested that disrupting the body’s natural digestion cycle may lead to complications, including vomiting, gastroparesis, bowel obstructions, and nausea. Studies have also noted that taking semaglutide may be associated with a potentially
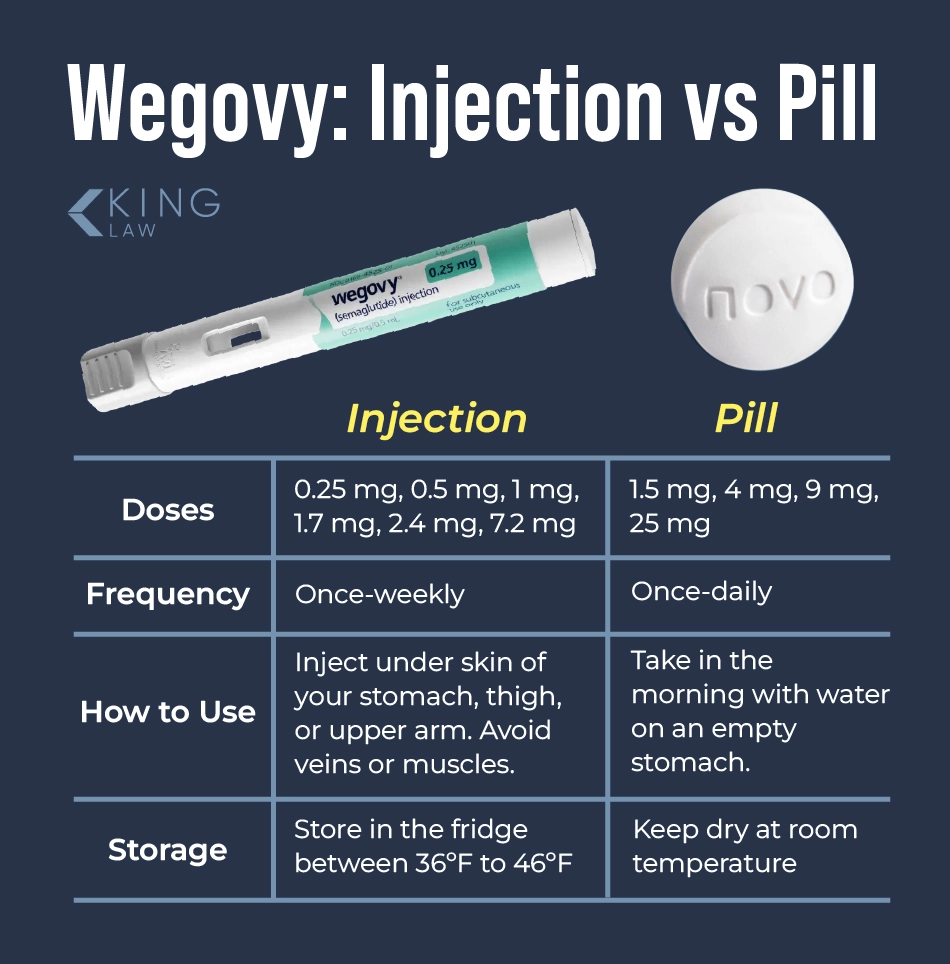
What Makes the Wegovy Pill Different From the Injection?
The Wegovy pill is an oral semaglutide drug made by Novo Nordisk. It is a glucagon-peptide receptor agonist (GLP-1 RAs or GLP-1s). The Wegovy pill and injection both have the same active ingredient (semaglutide), but they come in different doses and have different instructions for when to take them.
The Wegovy pill is digested and absorbed in the stomach. Its method of absorption is different. According to research published in the Springer medical journal, how long someone has been fasting, how much water they take with the pill, and their current body weight all can affect how much semaglutide is bioavailable in their system.
According to the prescribing label, the Absolute bioavailability of semaglutide is “estimated to be approximately 1% to 2% following oral tablet administration.” The maximum concentration of semaglutide is achieved one hour after someone takes the pill as directed.
Unique Characteristics of the Wegovy Pill
According to FDA prescribing information, here are some ways the Wegovy pill is unique:
- Wegovy pills are taken once a day on an empty stomach as opposed to being injected once a month.
- The pill is directly digested by the stomach as opposed to entering subcutaneous fat and entering the bloodstream.
- The pill must be taken on an empty stomach, unlike the injection, which may be administered on a full or empty stomach.
- The initial dosing and escalation schedules are different.
The current Wegovy pill is made in doses of 1.5, 4, 9, or 25 mg per day. The recommended maintenance dosage of Wegovy tablets is 25 mg. The Wegovy pill is to be taken once daily for cardiovascular risk reduction and weight reduction in adults. The differences in how Wegovy is put into and absorbed by the body may affect the side effects that people experience.
The active ingredient in the Wegovy pill is semaglutide. This is the same active ingredient present in injectable GLP-1 drugs like Wegovy, Ozempic, and Rybelsus, which are currently approved to treat diabetes.
What Warnings Are Provided About the Wegovy Pill?
The Wegovy pill boxed label includes the following warnings:
- Acute pancreatitis
- Acute gallbladder disease
- Acute kidney injury
- Severe gastrointestinal problems
- Increased risk of thyroid tumors
The Wegovy pill warning label may be updated by the FDA in the future. Some Wegovy pill lawsuits claim that Novo Nordisk was not clear enough about the gastrointestinal and other risks people might experience when taking oral semaglutide. For example, the March 19, 2026, warning label for the Wegovy pill does not list gastroparesis (stomach paralysis) or ileus (bowel blockage) as a potential complication.
According to prescribing information, severe gastrointestinal reactions were reported in 2% of people who took Wegovy tablets in clinical trials. Acute gallbladder disease was reported in 2.5% of patients who took Wegovy tablets, while it was only reported in 1.6% of Wegovy injection-treated patients.
Could the Wegovy Pill Cause Long-Term or Permanent Damage?
Some of the complications that past Wegovy pill patients claim to have experienced may cause long-term or permanent damage. In some situations, the complications may continue even after the person stops taking the Wegovy pill.
For example, acute pancreatitis can sometimes lead to chronic pancreatitis or recurrent acute pancreatitis (RAP). One research study (
Who May Be Eligible to File a Wegovy Pill Lawsuit?
Someone may be able to file a Wegovy pill lawsuit with our firm if they took the Wegovy pill and experienced severe and unexpected complications, including:
- Gastroparesis
- Deep vein thrombosis (DVT)
- Gallbladder cancer
- Gallbladder removal (prior to March 2022)
- Ileus/bowel obstruction (w/surgery)
- Necrotizing pancreatitis
- Vision loss from non-arteritic anterior ischemic optic neuropathy (NAION)
- Pancreatic cancer
- Pulmonary aspiration
- Pulmonary embolism
Sometimes, you may need to have a specific test done (e.g., a gastric emptying study) to prove that you were diagnosed with the above conditions. People may also qualify to file a lawsuit if they believe their loved one died because of Wegovy pill complications.
The above criteria were developed by our firm and may change in the future. You may still qualify to file a Wegovy pill lawsuit even if your situation is different from what we have listed here.
What Types of Compensation May Be Available in Wegovy Pill Cases?
Some people who develop severe complications after taking the Wegovy pill may qualify to request the following compensation:
- Medical expenses to cover the costs of diagnosis, treatment, and maintenance
- Lost wages or earning potential
- Pain and suffering
- Treatment-related travel expenses
- Loss of quality of life
Surviving family members may be able to receive wrongful death damages (e.g., funeral and burial costs) if their loved one passes away from Wegovy pill-related complications.

Are Wegovy Pill Lawsuits Being Consolidated Into an MDL?
Some Wegovy lawsuits have been consolidated into two MDLs (multidistrict litigation) to streamline some of the legal process for gastrointestinal or vision claims. Many of those lawsuits centered on the injectable forms of Wegovy and other GLP-1s, but they have also included at least one oral GLP-1.
The Wegovy pill is new, and patients may file lawsuits if they believe they suffered from Wegovy-related complications that they were not warned about. These lawsuits may be included in the existing GLP-1 multidistrict litigation, or a new MDL may be created for the oral versions of Wegovy and other GLP-1s.
Our attorneys are closely following this litigation. We will meet with each client and try to file their case in the way that presents the most potential advantages to them, whether that is by joining a centralized legal action at the state or federal level or filing outside of a centralized action.
How Does the Wegovy Pill Compare to Other GLP-1 Lawsuits?
Novo Nordisk and at least one other GLP-1 manufacturer are facing lawsuits filed by past patients who took a GLP-1 like Wegovy and suffered severe gastrointestinal complications. Some of these lawsuits accuse Novo Nordisk of failing to warn patients about the risks associated with Wegovy and its other GLP-1 drugs. It is likely that the same allegations and arguments will be produced in lawsuits alleging injuries from oral Wegovy tablets.
What Steps Should I Take to File a Wegovy Pill Lawsuit?
Filing a Wegovy pill lawsuit involves taking the following steps:
- Schedule a free case evaluation with a reputable Wegovy pill attorney.
- Collaborate with your lawyer to gather evidence to support your Wegovy pill complication claim.
- Your attorney writes and files a complaint against Novo Nordisk, which manufactures the Wegovy pill.
- Your Wegovy pill lawyer guides settlement discussions to help maximize your compensation potential.
- Your attorney takes your Wegovy pill case to trial if settlement talks are unsuccessful.
Wegovy pill patients may find it helpful and reassuring to partner with an attorney who can protect and assert their legal rights.
Evidence Needed to File a Wegovy Lawsuit
Here is some of the evidence someone will need to file a Wegovy pill lawsuit:
- Pharmacy and medical records that show you were prescribed the Wegovy pill
- A record of initial dosing and increases (e.g., how many 1.5 mg, 4 mg, 9, mg and 25 mg doses were taken)
- Medical records related to all diagnosed conditions possibly related to the Wegovy Pill
- Medical bills and other financial losses related to your diagnosis
You can reach out to an attorney even if you do not have all of this information. Our team can advise you on how to obtain records and file requests on your behalf.
How Long Do I Have to File a Wegovy Pill Lawsuit?
The amount of time you have to file a Wegovy pill lawsuit depends on several factors, including the state law that applies to your case. If you file outside of the applicable statute of limitations, you may lose your right to recover compensation for your Wegovy pill-related losses.
States have their own laws that designate timelines for different types of lawsuits, such as personal injury, product liability, and wrongful death claims. Some of these statutes of limitations change on an individual basis if a legal exception applies. People who believe they may qualify to file a Wegovy pill lawsuit may benefit from consulting a lawyer to learn their deadlines.
Wegovy Pill Lawsuit Settlements and Payout Amounts
Wegovy pill lawsuits do not have an average or standardized payment amount, like one would receive in a class action settlement. However, each case’s result (including the payout) may reflect the individual patient’s losses, the applicable liability laws, the strength of their case, and other factors. A knowledgeable attorney can help patients estimate the value of their settlement if they win their case.
King Law Is Investigating Wegovy Pill Lawsuits
King Law is actively investigating claims where Wegovy pill patients suffered from complications that they may not have been warned about. We are prepared to conduct free case evaluations to determine if injured patients who took the Wegovy pill were told to expect gastroparesis, pancreatitis, blood clots, vision loss, and other adverse events. We encourage Wegovy pill patients to call us to learn more.
Contact a Wegovy Pill Lawyer Today
Contact our office today by calling 585.496.2648 or filling out a form if you took the Wegovy pill and experienced complications. You may be entitled to compensation from Novo Nordisk. King Law’s team is prepared to help you discover and protect your legal rights. Our consultations are cost and risk-free.
Frequently Asked Questions (FAQs)
List of Sources Used In This Article
King Law used the following sources when writing this article about Wegovy pill lawsuits. We apply rigorous editorial standards to create content for our visitors.
Drug Approval Package: Application No. 218316. U.S. Food and Drug Administration, 2026.
GLP-1 Receptor Agonists: Clinical Review. National Institutes of Health, 2022.
GLP-1 Receptor Agonists and Associated Outcomes. National Institutes of Health, 2025.
MDL No. 3094 Case Information “In re: Glucagon-Like Peptide-1 Receptor Agonists (GLP-1 RAs) Products Liability Litigation (MDL No. 3094).” U.S. District Court for the Eastern District of Pennsylvania, 2024.
Labeling for Application No. 218316. U.S. Food and Drug Administration, 2026.
News and IR Materials. Novo Nordisk, n.d.
Novo Nordisk to Sell Wegovy Pill in U.S. for Self-Pay Patients Starting at $149 per Month. Reuters, 5 Jan. 2026.
Overgaard, Rasmus V., et al. “Clinical Pharmacokinetics of Oral Semaglutide”. Clinical Pharmacokinetics, 2021. PubMed Central (PMC).
Safety and Regulatory Information Document U.S. Food and Drug Administration, 2026.
Wegovy Prescribing Information. Novo Nordisk, n.d.
Wegovy Pill Demonstrated Greater Weight Loss Study PR Newswire, 2026.