Complete the form below to see if you qualify for a Oxbryta Lawsuit

People are filing Oxbryta lawsuits after experiencing vaso-occlusive crisis (VOCs) and injuries associated with this condition. Oxbryta (voxelotor) is a pharmaceutical drug that was intended to treat hemolytic anemia caused by sickle cell disease (SCD). But, in September 2024, the drug was voluntarily withdrawn from the U.S. and international markets. During postmarketing clinical trials, it was revealed that Oxbryta presented an increased risk of vaso-occlusive crisis and death. Some people who took Oxbryta to treat their SCD experienced these severe side effects. As more information about the dangers of Oxbryta emerges, injured patients and their families are filing lawsuits against Pfizer, the drug’s manufacturer. If you have experienced severe side effects after taking Oxbryta, our firm is ready to answer your questions.
Oxbryta Lawsuit Overview
People who experienced severe side effects and injuries after taking Oxbryta (Voxelotor) are starting to file lawsuits against Pfizer. These lawsuits allege the drug manufacturer failed to properly warn patients with sickle cell disease about the dangers of taking the drug. Instead of Oxbryta helping them fight the anemia associated with sickle cell disease (SCD), many people faced severe side effects such as:
- Increased VOCs
- Increased deaths
- Increased blood clots
- Increased serious vascular events
- Pediatric deaths
- Organ damage secondary to VOCs (kidney/liver)
- Stroke
These side effects were discovered in two registry-based studies from U.S. post-clinical trials and the European Medicines Agency (EMA). Due to these severe side effects, Pfizer initiated an international, voluntary withdrawal of Oxbryta on September 25, 2024. The next day, the U.S. FDA issued a warning statement about Oxbryta to patients, caregivers, and healthcare professionals. The notice advised physicians to immediately stop prescribing Oxbryta and told patients to talk to their physicians immediately about discontinuing the drug.
Pfizer’s previous patient information label for Oxbryta does not warn physicians or patients about the possibility of VOCs, stroke, organ failure, or increased risk of death. Pfizer updated the warning label in August of 2023 and did not list these severe side effects. Patients who experienced these side effects are pursuing legal action against Pfizer for not properly warning them about the dangers of Oxbryta. These side effects had serious impacts to patients and their families, and attorneys are fighting to get people injured by Oxbryta and their surviving family members the compensation they deserve.

Oxbryta Lawsuit – 2026 Update
April 23, 2026: Status Report Due in Oxbryta Personal Injury and Class Settlement Cases
Several people who took Oxbryta for sickle cell anemia symptoms became sick or died, such as from vaso-occlusive crises. Some believe these complications were potential side effects that Oxbryta patients were not warned about by Oxbryta’s manufacturer or their doctors. Some lawsuits have been filed against Oxbryta’s manufacturer over this matter, and settlements may be underway. The Judge overseeing some of these federally-filed Oxbryta cases has asked the parties to submit a status report on May 22, 2026, and has suggested that the parties intend to ask the judge to review the preliminary terms of the settlement soon.
April 16, 2026: Parties in Oxbryta Lawsuits Once Again Ask Judge for More Time to Finalize Settlement Terms
Some patients who took Oxbryta to treat symptoms of sickle cell anemia experienced higher-than-expected rates of vaso-occlusive crises (VOCs) and other complications. Because of this, Pfizer recalled Oxbryta in 2024. Some people who took Oxbryta and suffered complications filed lawsuits because they believed they should have been warned about the risks. Several of those Oxbryta lawsuits have been coordinated in federal court, and the parties have asked the judge for additional time to finalize settlement terms.
March 19, 2026: New Status Hearing Date Set in Coordinated Oxbryta Lawsuits
The judge overseeing the coordinated Oxbryta lawsuits has approved another delay in the status hearing originally scheduled for earlier this year. Near the end of 2025, the parties had told the judge they had agreed to settle the Oxbryta disputes and needed more time to finalize the terms. Since that time, they have asked for several extensions. The judge has agreed to reschedule the status hearing to April 23, 2026, and asks that the parties submit a joint case management statement by April 16, 2026.
March 12, 2026: Lawyers in Oxbryta Lawsuits Ask Judge for More Time to Finalize Settlement Terms
The lawyers representing individuals and families who were impacted by complications allegedly caused by Oxrbyta have asked to reschedule an upcoming status hearing. Oxbryta was prescribed to many people to treat sickle cell anemia-related symptoms. However, it was voluntarily withdrawn from the market in 2024 after there were concerning reports about Oxbryta patients experiencing an increase in vaso-occlusive crises, which can be fatal. Lawyers representing some of the people suing Pfizer and Global Blood Therapeutics said they have agreed to settle some of these cases but are still finalizing the terms. The parties have asked for another 30-day extension of these Oxbryta cases, which the judge says they might grant if the parties agree to withdraw pending motions, which is common when the parties enter a settlement.
March 3, 2026: Parties in Oxbryta Lawsuits To Discuss Progress on Settlement Discussions and Next Steps on March 19th
A hearing has been scheduled for March 19, 2026, in a lawsuit involving Oxbryta, according to a text-only docket entry. The parties had announced a potential settlement agreement, but they said more time was needed to continue and finalize those discussions. According to the docket notation issued by the clerk, the parties have been asked to turn in a Joint Case Management Statement by March 12, 2026. Oxbryta patients who believe they experienced unexpected complications after taking the now-recalled medication are encouraged to talk to a lawyer about whether they qualify for compensation.
February 6, 2026: Parties Request More Time to Continue Settlement Discussions in Oxbryta Cases
The parties in the Oxbryta lawsuit have asked the judge for a 30-day extension to continue settlement talks. Late last year, the parties signaled that they had decided to settle but needed more time to finalize the terms. A hearing was scheduled for February 12, 2026. However, that date may be extended another 30 days. Some people who took Oxbryta to treat sickle cell anemia symptoms experienced complications that were life-threatening, and they or their surviving loved ones have filed lawsuits to seek compensation from Pfizer and Global Blood Therapeutics. Oxbryta patients or their surviving loved ones who have not already filed a claim may be eligible to do so. They are encouraged to talk to an attorney about their situation.
February 4, 2026: Parties Prepare for Upcoming Status Conference as Settlement Talks Continue
Some patients with sickle cell anemia were given Oxbryta to help treat their symptoms. However, a number of these patients experienced severe complications that resulted in hospitalizations and even death. Oxbryta has been recalled due to higher-than-expected complication rates, and some patients and their surviving loved ones are suing Global Blood Therapeutics and Pfizer for failing to warn them about some of Oxbryta’s risks. Some of these lawsuits have been transferred to federal court. As of December 2025, settlement talks are underway, and the parties are expected to update the judge about these discussions on February 12, 2026.
January 6, 2026: Settlement Underway for More People Alleging Injuries from Oxbryta
A man took Oxbryta after it was prescribed to him by his doctor to treat his sickle cell anemia-related symptoms. He experienced complications shortly afterward, which his medical team believes were due to Oxbryta. He and several other former Oxbryta patients filed a class action lawsuit against Pfizer and Global Blood Therapeutics on December 23, 2024. According to court filings, a potential settlement of the case is in the works, and the parties in his Oxbryta class action lawsuit have asked for a 60-day extension to continue negotiating terms. The judge granted this request, and the parties indicated that they expect to provide an update by February 2026. Other Oxbryta patients who have suffered complications can reach out to an attorney to learn if they may qualify for a lawsuit and be able to seek a settlement.
January 4, 2026: Woman Dies After Taking Oxbryta for Sickle Cell Anemia Symptoms
The estate of a woman who passed away is pursuing a lawsuit against the makers of Oxbryta. The case involves a woman who died after taking Oxbryta to treat her sickle cell anemia-related symptoms. One of her close relatives, who is also the administrator of her estate, filed a lawsuit in hopes of holding Pfizer and Global Blood Therapeutics accountable. The family believes that Pfizer and Global Blood Therapeutics were aware of the elevated risks associated with Oxbryta and should have shared this information with patients. The complaint accuses the Oxbryta companies of 10 counts, including failing to warn, strict liability, wrongful death, and negligence. Although this lawsuit was filed in 2024 and amended in 2025, it is still active in San Francisco courts under Case No. CGC-24-621022.
December 18, 2025: California Woman Sues After Husband’s Death Due to Oxbryta Complications
Laura Christine Matteliano-Madu has filed a lawsuit against a hospital and Global Blood Therapeutics after her husband, Nbubuisi Madu, died. In 2021, Nbubuisi started taking Oxbryta as part of a clinical trial for the treatment of sickle cell disease, which Nbubuisi was diagnosed with when he was seven. Several years after he started taking Oxbryta, Nbubuisi suffered from urgent side effects, including a life-threatening vaso-occlusive crisis, which, in layperson terms, is when pieces of blood cells get stuck in tiny veins in the body. Laura claims that these complications and alleged mistakes made by the hospital caused Nbubuisi’s death. She also believes that Pfizer, Global Blood Therapeutics, and several others are responsible for not warning her husband of the risks and for not taking better care of him in the hospital.
December 3, 2025: Pfizer, Global Blood Therapeutics Settle Another Oxbryta Lawsuit
A man named Maurice Frazier filed a lawsuit after Oxbryta harmed him. His attorneys have reached an agreement to settle his Oxbryta lawsuit against Global Blood Therapeutics and Pfizer. Maurice was prescribed Oxbryta to help treat his sickle cell anemia. However, he suffered from several vaso-occlusive crises (VOCs), which involved painful blockages that prevented blood and oxygen from reaching different parts of his body. Maurice believes that Pfizer and Global Blood Therapeutics knew about the increased risk of VOCs but did not tell patients taking Oxbryta. The upcoming settlement, which is still being finalized, should help compensate Maurice for the immense medical bills and personal trials he went through because of Oxbryta complications.
November 17, 2025: Oxbryta Ban in the EU Renewed Over Continued Safety Concerns
Sickle cell anemia treatment Oxbryta was recalled in the EU in September 2024 because safety data showed that people who took Oxbryta had a higher risk of blood clots and other complications. EU authorities have decided to extend this Oxbyrta ban after further trial data showed that more patients who took Oxbryta died compared to those who did not take Oxbryta, particularly if they were children. Patients in the United States are filing lawsuits against Pfizer and other Oxbryta manufacturers for not warning them that the drug was likely to cause blood clots and other complications.
November 4, 2025: Woman Receives Oxbryta Settlement on Behalf of Herself and Her Husband’s Estate
Dianna Ford’s attorneys have reached a settlement with Pfizer and Global Blood Therapeutics, makers of the defective Oxbryta. Oxbryta was hailed as the potential gold standard for treating sickle cell anemia. However, patients (such as Dianna’s deceased husband, Bruce) who took Oxbryta had a higher chance of developing blood clots, strokes, and related complications. Patients and their surviving loved ones are filing lawsuits against Oxbryta’s makers to seek much-deserved compensation for the injuries and deaths Oxbryta has caused.
October 17, 2025: One Plaintiff’s Oxbryta Case Reaches a Settlement As More Lawsuits Are Filed
Tirrell and Latoya Allen have reached a potential settlement with Oxbryta’s manufacturers, Pfizer and Global Blood Therapeutics. They expect to finalize the terms of settlement in the coming months, and it is likely that the Allens’ payout amount will be confidential. Tirrell developed blood clots and had a stroke shortly after taking Oxbryta to treat his sickle cell anemia. The compensation he will receive from the settlement will help pay for his medical bills and reimburse him for his other losses.
October 3, 2025: Number of Oxbryta Complication Cases Grows Because of Now-Withdrawn Sickle Cell Anemia Drug
Oxbryta patients continue to discover that their blood clots and other medical conditions are due to the sickle cell anemia drug they took. Oxbryta was pulled from the shelves. However, many patients who experienced complications are standing up against Pfizer because of the losses they endured due to this unsafe medication. A recent court filing reveals that at least 18 Oxbryta lawsuits are pending in federal and state courts, and this number is expected to grow.
September 2, 2025: Oxbryta Lawsuits Gain Momentum as Pfizer’s Second Sickle Cell Drug Fails Safety Testing
Thousands of people with sickle cell anemia took Oxbryta to help improve their symptoms and quality of life. Unfortunately, many patients experienced high rates of blood clots and other complications, including death. Oxbryta was pulled from the shelves due to safety concerns, and Pfizer has been testing a replacement sickle cell treatment. However, that new drug also resulted in higher-than-expected blood clot rates during clinical trials. Oxbryta patients and their surviving loved ones are filing lawsuits to recover compensation for their losses.
August 1, 2025: New Oxbryta Lawsuit Joins the Fight Against Pfizer and Global Blood Therapeutics
Dianna Ford has brought a lawsuit against Oxbryta’s manufacturer, Global Blood Therapeutics, and its parent company, Pfizer. Dianna’s husband, Bruce, took Oxbryta to treat his sickle cell anemia. Tragically, Oxbryta caused Bruce to develop painful blood clots, suffer from a stroke, and die. Dianna seeks to hold Oxbryta’s manufacturers accountable for creating a deadly drug and not warning her and Bruce of the risks.
July 2, 2025: Oxbryta Lawsuit Progresses as More Plaintiffs File Individual Claims
Tirrell Allen’s case against Global Blood Therapeutics continues as he and the defendant corporation submit an update to the court. In this joint statement, the parties explain the basis of the lawsuit: Tirrell took Oxbryta to treat his sickle cell anemia, and he experienced a high number of blood clots and a stroke within a month afterward. Tirrell’s legal team continues to gather evidence to support his claim for compensation.
June 3, 2025: Another Oxbryta Lawsuit Joins Federal Cases Against Global Blood Therapeutics and Pfizer
Another Oxbryta lawsuit has been added in the fight against Global Blood Therapeutics and Pfizer because of its unsafe sickle cell anemia treatment. This drug caused dozens of patients to experience organ damage and blood flow blockages that were often life-threatening. In a recent court order, Judge Thompson, who is overseeing many current Oxbryta lawsuits, noted it may be appropriate to consolidate these actions into a multidistrict litigation.
May 13, 2025: Parties in Oxbryta Lawsuits Update Judge on Number of Cases and Mediation Status
The parties in several Oxbryta lawsuits submitted a joint status update to Judge Trina L. Thompson. According to the document, there are at least seven cases against Global Blood Therapeutics and Pfizer because of complications from Oxbryta, a sickle cell anemia medication that has now been withdrawn (which has similar implications to a recall). The parties are choosing a mediator to help resolve the dispute, even as more consumers file new lawsuits.
May 1, 2025: People Who Experienced VOCs on Oxbryta Still Have Options to File a Lawsuit
Although Oxbryta has been withdrawn (an action similar to a recall) from the U.S. market, many people who used the sickle cell disease experienced devastating consequences. There is still time for people who suffered VOCs or organ damage while taking Oxbryta to talk to an attorney. If someone lost a family member who was taking Oxbryta, they may still have options to file a lawsuit on behalf of that family member.
April 11, 2025: Oxbryta Judge Adds Another Oxbryta Lawsuit To Their Docket
Judge Trina Thompson has signed an order transferring another Oxbryta lawsuit to her docket, the lawsuit of Tirrell Allen. Judge Thompson has been overseeing Mr. Allen’s claims against Global Blood Therapeutics and Pfizer. The parties (lawyers on both sides) are in the process of filing and resolving pre-trial motions, including an upcoming motion to dismiss. Another plaintiff asked the judge to manage their case as well, which can encourage consistent and efficient rulings. The judge granted this request. Having these lawsuits appear before the same judge can be very beneficial to plaintiffs.
March 18, 2025: People Can File Lawsuits After Realizing Their Injuries Might Have Been Caused by Oxbryta
Oxbryta was removed from the market over dangerous side effects, including an increased risk of vaso-occlusive crises (VOCs). However, some people who took this sickle cell disease treatment may not realize that their kidney damage, heart damage, liver damage, or strokes were caused by Oxbryta-related VOCs. If patients are just learning Oxbryta put them at an increased of these conditions, there may still be time to file a lawsuit against the manufacturers.
March 3, 2025: Trial Date Set in Man’s Oxbryta Lawsuit Against Pfizer and Global Blood Therapeutics
A federal judge approved a scheduling order in a case filed by a plaintiff, Tirrell Allen, against the companies that manufactured the sickle-cell anemia drug, Oxbryta. The trial is scheduled for June 7, 2027, and may last between 12 and 15 days. Numerous experts, doctors, and corporate staff may be questioned at trial about how the drug was made, tested, prescribed, and used. People who experienced VOCs after using Oxbryta can still file lawsuits.
February 12, 2025: 4 Months Out From Global Withdrawal, Oxbryta Lawsuits Can Still Be Filed
It has been more than 4 months since Pfizer issued its global withdrawal (recall) of Oxbryta, its sickle cell anemia treatment. After the drug was found to increase the risk of vaso-occlusive crises (VOCs) in many people, the drug was pulled from the market. Even though Oxbryta is no longer available, people who were harmed by it can still file lawsuits. That statute of limitations has not expired, and people who were harmed by Oxbryta likely have viable cases. People who lost a loved one to this drug may also still have legal options.
January 14, 2025: Oxbryta Lawsuits Say Pfizer and GBT Failed Patients at Multiple Points
People are continuing to file Oxbryta lawsuits after suffering severe side effects associated with the sickle cell disease (SCD) drug. It is now known Oxbryta increased the risk of vaso-occlusive crises (VOCs) in SCD patients. Lawsuits filed against Pfizer and Global Blood Therapeutics (GBT) say the companies knew about this risk and failed to take the proper actions to warn patients. People who used Oxbryta (voxelotor) say Pfizer and GBT failed patients in multiple capacities. If patients had received proper information or warnings about the increased risk of VOCs, they may not have taken Oxbryta. In failing to properly inform patients, it is likely these companies caused severe injuries and deaths. Lawsuits allege that despite trial and other data demonstrating severe risks, these companies kept the drug on the market and did not alert the proper authorities of their findings.
December 12, 2024: More Than Half of Patients in Voxelotor Study Did Not Follow Through on Drug
In a study published in the American Society of Hematology’s journal Blood, more than half of the patients involved in the Oxbryta (voxelotor) study did not participate in all steps. 31 of 60 of the study either did not initiate the drug, did not return for follow up labs at time of analysis, or refused to take the drug once approved. Another 9 patients only returned for 1 month of follow-up labs. This left a small portion of patients for the study’s authors to base their findings on.
December 2, 2024: Oxbryta Lawsuits Accuse Manufacturers of Reckless Conduct With Their Sickle Cell Disease Treatment
In a lawsuit filed in November, attorneys representing a man harmed by Oxbryta make several accusations against Pfizer and Global Blood Therapeutics, who manufactured Oxbryta (voxelotor). The complaint filed in California federal court says the “Defendants’ conduct…was reckless. Defendants risked the lives of consumers and users of the products.” Additionally, the lawsuit says the plaintiffs had knowledge of safety problems associated their sickle cell disease treatment, yet they suppressed this knowledge from the general public.
November 21, 2024: Illinois Man files Oxbryta Lawsuit After Experiencing Several Complications
An Illinois man has filed a lawsuit against Global Blood Therapeutics, Inc. and Pfizer, Inc. after he experienced severe injuries after taking Oxbryta. He filed his complaint in the U.S. District Court in Northern California on November 7, 2024. Tirrell Allen started using Oxbryta in August of 2024. He had been diagnosed with sickle cell disease (SCD) as a child. Immediately after starting the drug, he had pain, swelling, increased vaso-occlusive crises (VOCs), and other debilitating symptoms. In September of 2024, he suffered a stroke and had to be hospitalized. Allen’s attorneys say that at the time he used the drug, he was unaware of the higher risks of VOCs and strokes because, “Defendants failed to warn, instruct, advise, educate, or otherwise inform Oxbryta users and prescribers about the risk of VOCs and/or death.” At the time Allen’s complaint was filed, he was still hospitalized.
November 15, 2024: British Medical Agency Had Concerns About Oxbryta Years Before Market Withdrawal
Pfizer withdrew Oxbryta (voxelotor) from global markets at the end of September. However, there were growing concerns about the drug and its performance in clinical trials for years before this withdrawal. England’s National Institute for Health and Care Excellence (NICE) had concerns throughout the approval process for Oxbryta. In July of 2023, the agency rejected voxelotor for use by Britain’s National Health Service. The reason they cited for their decision was that clinical trial evidence did not support cost effectiveness. NICE published new draft guidance for the drug in February of 2024, but the drug would only be on the market for a few months before it was voluntarily withdrawn by Pfizer.
November 7, 2024: People Injured by Oxbryta Begin Filing Lawsuits After Being Harmed by Sickle Cell Disease Drug
Patients and surviving family members are starting to file lawsuits after they or their loved ones were injured by Oxbryta. These cases cite injuries such as increased vaso-occlusive crises (VOCs), organ failure, and death for patients with sickle cell disease who were taking the drug. Our firm has been intaking several Oxbryta clients from around the country. The most common complaint is an increase in VOCs leading to hospitalization. We have also heard from several patients that found out they were in the early stages of organ failure because of blood tests that showed poor kidney and liver function. Studies have also shown an increased risk of death and stroke when using Oxbryta. If you have used Oxbryta and would like to discuss the side effects you experienced, please call our office or click on “See If You Qualify” to submit a form.
November 4, 2024: Oxbryta Side Effects Include Increased VOCs, Organ Failure, and Risk of Death
Oxbryta complaints include death, increased vaso-occlusive crises (VOCs), and organ failure. Our firm has been taking on several Oxbryta clients from across the country. The most common complaint is an increase in crises leading to hospitalization. We have also heard from several patients who discovered they were in the early stages of organ failure due to blood tests showing poor kidney and liver function. Studies have also shown an increased risk of death and stroke when using Oxbryta. If you have used Oxbryta and would like to discuss the side effects you experienced, please call or fill out a form here.
November 1, 2024: European Medicines Agency (EMA) Recommends Suspension of Oxbryta for SCD Treatment
The EMA’s human medicines committee (CHMP) made the recommendation to suspend marketing authorization for Oxbryta a treatment for sickle cell disease (SCD). They took this action after the active ingredient in Oxbryta (voxelotor) was shown to increase the risk of vaso-occlusive crises (VOCs) and death. As part of the recommendation the EMA said doctors should take the following actions: stop prescribing Oxbryta to new patients; contact patients currently taking Oxbryta and ask them to stop taking the drug and find a new treatment; and monitor patients for adverse events. Similarly, the EMA is advising SCD patients who take Oxbryta to contact their physicians immediately.
October 31, 2024: Oxbryta Lawsuits Expected Soon, Details Still Unclear
Many legal experts think Oxbryta lawsuits will be filed soon. We have not seen any complaints yet, as the withdrawal is only a few weeks old. We expect the lawsuits to be filed in federal court. It is unclear how many cases will be filed since Sickle Cell Disease is relatively rare and this drug was not used for very long. Estimates suggest that about 100,000 Americans are affected by Sickle Cell Disease. It is uncertain whether enough lawsuits will be filed to allow for multidistrict litigation, a class action-style lawsuit.
October 26, 2024: Oxbryta Clinical Trial Data Shows High Death Rate in Children Ages 2-15
The phase 3 clinical trial of Oxbryta that was supposed to end in January of 2025 was halted by the voluntary market withdrawal of Oxbryta. However, data from that study likely motivated the decision for that withdrawal. In the study, entitled, HOPE Kids 2 trial (NCT04218084), 8 children who were given Oxbryta died, compared with two deaths in the placebo group. This discovery likely contributed to Pfizer’s decision to pull Oxbryta from the market.
September 26, 2024: Pfizer Issues Global Withdrawal of Sickle-Cell Drug Oxbryta Due to Safety Concerns
Due to Oxbryta leading to an increased risk of serious vascular events and death, Pfizer has voluntarily withdrawn the drug, which is used to treat anemia caused by sickle cell disease. As part of the voluntary recall/withdrawal, Pfizer is suspending distribution and discontinuing all active clinical trials and expanded access programs, due to the dangers the drug poses to people with sickle cell disease.
August 8, 2022: Pfizer Acquires Oxbryta Manufacturer Global Blood Therapeutics, Inc. for $5.4B
Pfizer, Inc. and Global Blood Therapeutics, Inc. (GBT) entered into an agreement where Pfizer would acquire GBT for $5.4 billion. GBT is a biopharmaceutical company that developed Oxbryta (voxelotor) in hopes of serving underserved patient communities, especially those affected by sickle cell disease.
December 17, 2021: FDA Gives Accelerated Approval for Oxbryta Prescriptions for Patients Age 2 to 11
The FDA approved the use of Oxbryta (voxelotor) tablets in pediatric sickle cell patients ages 4 to 11. As of 2019, the drug has received accelerated approval for patients age 12 and older.
November 1, 2020: GBT Consultant Publishes Research Paper About the Effectiveness of Voxelotor
A consultant working for the manufacturer of Oxbryta (GBT) served as the lead author in a study published in Blood, the Journal of the American Society of Hematology. The study, titled, “A phase 1/2 ascending dose study and open-label extension study of voxelotor in patients with sickle cell disease,” concluded that, “oxelotor was well tolerated with no treatment-related serious adverse events and no evidence of tissue hypoxia.” Time would prove otherwise.
November 25, 2019: FDA Grants Accelerated Approval for the Use of Oxbryta for Patients 12 and Older
The FDA granted accelerated approval to Oxbryta (voxelotor), who was owned by Global Blood Therapeutics, Inc (GBT). This approval was for sickle cell patients age 12 and older. At the time, the side effects listed were headache, diarrhea, abdominal pain, nausea, rash, fatigue, and pyrexia.
December 3, 2018: FDA Accepts GBT’s Proposal for Accelerated Approval Pathway for Oxbryta
Global Blood Therapeutics, Inc., submitted a proposal to the FDA for an accelerated approval pathway for voxelotor for the treatment of sickle cell disease. The FDA approved this proposal on December 3, 2018, setting the drug on the way to sickle cell patients.
May 2017: FDA Clinical Trials for Oxbryta Conclude
The FDA’s clinical trials for Oxbryta as used to treat patients with sickle cell disease concluded in May 2017. The trials evaluated the safety, tolerability, pharmacokinetic, and pharmacodynamic effects of the GBT440, which would become Oxbryta.
About the Oxbryta Lawsuit:
What Is Oxbryta and How Does It Work?
Oxbryta’s FDA-Approval and Market Presence
Oxbryta Side Effects and Health Risks
Oxbryta Linked to Vaso-Occlusive Crises (VOCs) and Death
Oxbryta Manufacturer Announces Voluntary Global Withdrawal
Eligibility Criteria to File an Oxbryta Lawsuit
Recoverable Damages in the Oxbryta Lawsuit
How to File an Oxbryta Lawsuit
Oxbryta Lawsuit Settlement and Payout Amounts
What Is Oxbryta and How Does It Work?
Oxbryta (voxelotor) is a prescription drug that was used to treat people with sickle cell disease (SCD). It offered a new approach to treating SCD, being a hemoglobin S polymerization inhibitor. It was designed to stop cells from sickling by addressing the root cause of SCD.
It came in tablet or liquid form and did not have a generic alternative. The drug was intended to decrease complications from SCD. SCD is a group of red blood cell disorders, which are inherited from a parent.
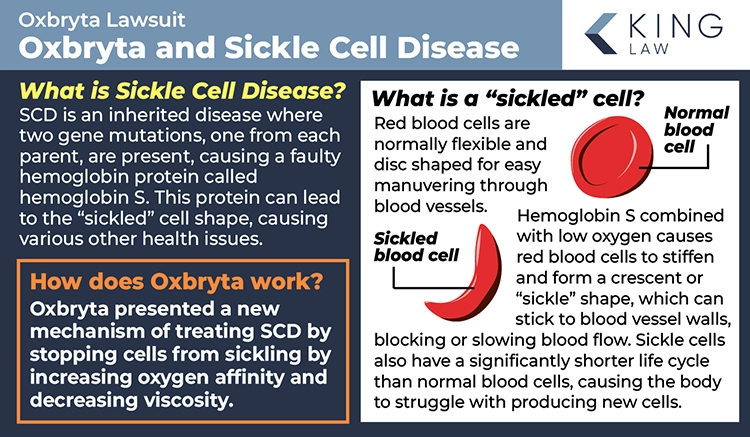
What is Sickle Cell Disease?
Sickle cell disease (SCD) is an inherited red blood cell disorder. When someone has SCD, their hemoglobin is abnormal and their red blood cells become hard and sticky and morph into a c-shape (a sickle shape). Because of these abnormalities, when red blood cells travel through blood vessels, they can get stuck and limit blood flow. The decrease in circulation can lead to severe problems for people with SCD, including infection, acute chest syndrome, and stroke. Additionally, these abnormalities lead to early red blood cell death.
Oxbryta was intended to prevent these serious complications in people with SCD. The medication was supposed to decrease sickle-shaped red blood cells from being formed by inhibiting hemoglobin polymerization. Oxbryta was often prescribed at the same time as the drug hydroxycarbamide, which is an oral chemotherapy drug that is used to reduce painful episodes and the need for blood transfusions in people with SCD.
Oxbryta was supposed to keep red blood cells from sickling, reduce blood viscosity (stickiness), and keep red blood cells from changing shape (deformability). The active ingredient in the drug (voxelotor) binds to hemoglobin, stabilizing it to prevent clumping and maintaining normal red blood cell shape and flexibility. This process helps red blood cells flow more easily through blood vessels, reducing complications associated with sickle cell disease.
Oxbryta’s FDA-Approval and Market Presence
Oxbryta’s road to FDA approval began in the early 2010s, but the drug was officially approved by the FDA on November 25, 2019. At that time, it was approved for adults and children age 12 and older. The United States was the first to approve the drug in the global market. Oxbryta gained its FDA approval via the accelerated approval process. It was granted this accelerated approval because of the results of the phase 3 GBT-HOPE clinical trial.
The results of the HOPE trial showed initial promise. The randomized, placebo-controlled trial involved 274 participants with SCD. The study concluded that Oxbryta (voxelotor) increased hemoglobin levels and reduced hemolysis (the breakdown of red blood cells). The results supported the idea that Oxbryta had disease-modifying potential.
Oxbryta gained a second accelerated approval from the FDA in December of 2021. This time, the drug was approved to treat SCD patients age 4 and older. It was approved based on the results of the HOPE-KIDS 1 trial.
Oxbryta also gained approvals in many other countries. The European Medicines Agency (EMA) granted Oxbryta the Priority Medicines (PRIME) designation and designated the drug as an orphan medicinal product for treating patients with SCD. Between 2019 and 2024, Oxbryta received approval in 35 different countries.
The FDA’s Accelerated Approval Process and Oxbryta
In order for Oxbryta to be prescribed to SCD patients in the United States, Global Blood Therapeutics (GBT) sought approval under the FDA’s accelerated approval process. This approval process means Oxbryta got faster approval. It was granted approval because initial data from the GBT-HOPE clinical trials showed promise in treating a serious disease, sickle cell disease.

Oxbryta Side Effects and Health Risks
Oxbryta has many possible side effects and health risks. Some side effects are mild and common, while others are less frequent but more severe.Common side effects of Oxbryta for adults include:
- Headaches
- Diarrhea
- Nausea/abdominal pain
- Fatigue
- Pyrexia (fever)
- Rash
The most common side effects of OXBRYTA in children ages 4 to 12 include:
- Fever
- Vomiting
- Rash
- Stomach-area (abdominal) pain
- Diarrhea
- Headache
Less-common but more serious side effects of Oxbryta include:
- Severe skin rash
- Serious allergic reactions that can lead to anaphylaxis, difficulty breathing, swelling of the face or throat, and rapid heart rate
Blood clots that present as chest pain; shortness of breath; or pain, swelling, or warmth in the leg
Oxbryta Linked to Vaso-Occlusive Crises (VOCs) and Death
As data from post-clinical trials came to light, it became apparent that Oxbryta likely increased the risk of vaso-occlusive crises (VOCs) and death, two things the medication was supposed to decrease for patients. Oxbryta has now been associated with an increased risk of VOCs, a severe and painful complication of sickle cell disease (SCD) caused by blocked blood flow due to sickled red blood cells.
Data from two phase-3 clinical trials, as well as interviews with people taking Oxbryta, led to startling findings about outcomes for patients taking Oxbryta. Data indicated that people were dying or experiencing VOCs at rates that questioned the drug’s safety.
Pfizer and the scientific community are currently exploring whether Oxbryta played a direct role in these deaths. This led to Pfizer issuing a voluntary withdrawal, saying the risks of Oxbryta likely outweigh the benefits of taking the drug.
“HOPE Kids 2” Study Shows High Rate of Pediatric Deaths
The “Study to Evaluate the Effect of GBT440 on TCD in Pediatrics With Sickle Cell Disease (HOPE Kids 2 Study)” was a post-clinical trial, Phase 3 study being monitored by the FDA. The study was a randomized, double-blind, placebo-controlled study that examined outcomes for children ages 2 to 15 who were taking Oxbryta (voxelotor) for SCD.
The study was supposed to conclude on January 28, 2025. However, early findings likely contributed to Pfizer to issuing the voluntary withdrawal. During the course of the study, eight children who were taking Oxbryta died, compared with only two children in the placebo group.
Voxelotor’s RESOLVE Study
Another study examined the outcomes for SCD patients who were taking Oxbryta to help resolve their leg ulcers. The Phase-3 study was called, “Resolution of Sickle Cell Leg Ulcers With Voxelotor (RESOLVE).” In that study, whole participants were 12 and older, 8 of the 88 patients died.

Oxbryta Manufacturer Announces Voluntary Global Withdrawal
On September 25, 2024, Pfizer initiated a global withdrawal of Oxbryta. Pfizer stated it was voluntarily withdrawing all lots of Oxbryta for the treatment of SCD in all markets where it is approved. Additionally, Pfizer announced it was suspending all clinical trials and worldwide expanded access programs. It made this decision to protect patient safety.
The reason for this withdrawal was information from the post-market clinical trials. Pfizer said that information gathered from these trials indicates the risks of Oxbryta now outweigh the drug’s benefits. Particularly, Pfizer cited that the increased incidence of vaso-occlusive crises (VOCs) and fatal events led to its decision to pull the drug off of the market. Pfizer is investigating these incidents, along with regulatory authorities from around the globe.
FDA Issues Oxbryta Warning to Patients and Healthcare Professionals
Oxbryta was not truly recalled. Instead, Pfizer issued a global market withdrawal. As soon as this occurred, the FDA issued a warning to Oxbryta users. On September 26, 2024, the U.S. FDA issued a warning statement about the dangers of Oxbryta. The statement alerted all patients, caregivers, and healthcare professionals about the Oxbryta (voxelotor) market withdrawal. It advised patients to contact their physicians immediately to discuss new treatment options for their SCD. In the statement, the FDA also encouraged anyone who had experienced a VOC or other severe side effect to report it to the Adverse Event Reporting System (FAERS).
Oxbryta’s Post-Marketing Monitoring Leads to International Withdrawal
After Oxbryta gained separate accelerated approvals from the FDA in 2019 and 2021, the clinical benefits of the drug still had to be proven. During these post-market clinical trials, researchers reported on serious dangers associated with Oxbryta. In the HOPE Kids 2 trial (NCT04218084), which looked at SCD patients from ages 2 to 15, 8 Oxbryta patients died, compared to 2 deaths in the placebo group. Additionally, two real-world registry studies also showed a higher rate of vaso-occlusive crisis in patients with sickle cell disease receiving Oxbryta. These observations led to the withdrawal of Oxbryta from the market.
European Medicines Agency (EMA) Recommends Suspension of Oxbryta
On September 26, 2024 the European Medicines Agency (EMA) recommended the suspension of Oxbryta. The EMA’s human medicines committee (CHMP) made this recommendation so they could review emerging data. The EMA started reviewing trial data in July 2024. The EMA’s said about the withdrawal, “overall, these data raise serious concerns about the safety of Oxbryta; due to the increased uncertainties it therefore recommended that the authorisation, marketing and supply of the medicine be suspended until all the available data have been assessed in the ongoing review.”
Eligibility Criteria to File an Oxbryta Lawsuit
People who took Oxbryta to treat their sickle cell disease (SCD) and then experienced severe complications may be eligible to file a lawsuit. That case would be filed against the drug’s manufacturer, Pfizer. To be able to file a claim, a patient must prove that they:
- Were prescribed Oxbryta to treat complications from SCD
- Experienced a vaso-occlusive crisis (VOC) during or after Oxbryta treatment
- Experienced organ damage or a stroke
- Suffered a fatal event during or after using Oxbryta
If a patient passed away due to complications from taking Oxbryta, a surviving family member is also eligible to file a lawsuit on their behalf.
If you meet the eligibility requirements for filing a lawsuit, you and your attorney will look at multiple factors, including:
- Duration of Oxbryta usage
- Time elapsed between starting or stopping Oxbryta and the occurrence of VOC or fatal event
- Serious complications such as organ damage or stroke
If you or a family member experienced any of these frightening complications, you may be able to file a claim against Oxbryta’s manufacturer.

Evidence Needed to File an Oxbryta Lawsuit
If you decide to file a lawsuit after being harmed by Oxbryta, your legal team will work with you to gather evidence for your case. Information you will likely be asked to gather includes:
- Medical records that show you have SCD and were prescribed Oxbryta by your doctor
- Prescription records from your pharmacy establishing how long you took Oxbryta
- Financial records such as medical bills, evidence of lost income, and other financial records related to your injury
- Documentation that shows you experienced vaso-occlusive crises (VOCs), organ damage, stroke, or death during or after your Oxbryta treatment
- Treatment records that show what treatments you endured related to your injuries
- Proof of hospitalization or surgery related to your injuries
Additionally, you and your legal team will work together to compile information on how your injuries from Oxbryta impacted your life. You may discuss and document how complications affected your quality of life, ability to work, and relationships.
Recoverable Damages in the Oxbryta Lawsuit
When you file an Oxbryta lawsuit for yourself or on behalf of a loved one, you will be filing a case in civil court and seeking damages. That means that if your case is successful, you can recover damages in the form of financial compensation. You may be able to receive compensation for things like:
- Past medical bills
- Future medical bills
- Prescription drug bills
- Compensation for lost wages
- Diminished earning capacity
- The physical pain you endured
- Loss of enjoyment of life and emotional suffering
- Permanent disability
- Loss of consortium (changes in your relationship with your partner)
- Wrongful death-related expenses (e.g., funeral and burial expenses)
In addition to receiving compensation for these wrongs, you may also be awarded punitive damages. If your case is successful at trial, Pfizer may be found to have been reckless or negligent and have to pay additional compensation.
How to File an Oxbryta Lawsuit
In order to file a lawsuit against Oxbryta’s manufacturer, you need to work with an attorney. That attorney should be experienced in product liability and personal injury lawsuits. There are certain steps in the lawsuit process, your legal team will help you through each of these steps:
- Initiate legal process with a law firm and sign a retainer
- Confirm eligibility
- Be assigned a lawyer familiar with pharmaceutical litigation
- Gather and prepare the necessary documentation
- File the lawsuit
- Complete the discovery phase
- Possibly engage in settlement negotiations
- Possibly participate in a trial
During step 1, you will reach out to a law firm and tell them you are interested in filing a case. During step 2, a legal team will confirm your or your loved one’s diagnosis of a VOC, fatal, event, or severe organ damage. During step 3, you will be assigned a lawyer, who will help you gather the evidence and documentation necessary for step 4. Step 5 is filing the lawsuit, likely in a federal court. As your lawsuit progresses, your case will go through the discovery phase. During this phase, your attorney will gather expert testimony as well as personal testimony and work to review files provided by the defendant (Pfizer). Both sides (plaintiff and defendant) will exchange relevant information, including your medical records, expert testimonies, and depositions. Your lawyer will also work to secure expert testimonies about the link between Oxbryta and VOCs or fatal events.
Once the discovery phase is completed, you may receive a settlement offer. Depending on the outcome of that offer and your attorney’s advice, your case may go to trial. During the trial, it will be determined if your case is successful and how much compensation you will receive.
Both sides will present their evidence, and a verdict will be made based on the case.
After the trial is complete, there may be additional steps. You or the defendant could appeal the decision. If they don’t and your case is successful, you will collect your awarded damages.

File an Oxbryta Recall Claim
Because Oxbryta has been withdrawn from the market, it is expected that Pfizer will face lawsuits after acknowledging their drug caused harm. If you or a loved one has experienced severe complications after taking Oxbryta, you should file a claim as soon as possible to protect the viability of your case.
Oxbryta Lawsuit Settlement and Payout Amounts
Settlement amounts for Oxbryta lawsuits will likely vary. Factors such as the severity of a patient’s injuries (like VOCs or fatal outcomes) and how those injuries impacted their life will play a role in determining settlement amounts., Because there is the possibility of punitive damages in an Oxbryta lawsuit, settlements could be high. Some settlements could range from $50,000 to $1.5 million or more, though this will vary widely by individual circumstances and there is no guaranteed amount.
The specifics of each case will be reviewed and factor into the settlement. The severity of the injuries, how much time was lost from work, medical costs, and real-life impacts will all be considered when determining a settlement. Severe, life-threatening, or life-ending complications tend to result in higher payouts.
There is no certain way to predict what settlements or trial awards will be in Oxbryta lawsuits. But, experienced attorneys can provide estimates.
Estimated Settlement Ranges:
- Moderate Cases (non-fatal VOCs or health complications): $50,000 to $200,000 per plaintiff.
- Severe Cases (multiple VOCs, significant health decline): $200,000 to $500,000 per plaintiff.
- Fatal Cases (wrongful death claims): $500,000 to $1.5 million or more per plaintiff, depending on circumstances and impact on the family.
The amount of each settlement could greatly vary from the estimates above. Each case will be evaluated based on individual factors and circumstances. Consulting an attorney who is experienced in product liability and pharmaceutical law will help the chances of a successful case and maximum compensation.
Oxbryta Lawsuit Statute of Limitations
State-specific statutes of limitations determine how much time someone has to file an Oxbryta lawsuit. A state’s statute of limitations sets a time limit for how long someone has to file a case after being injured by Oxbryta. Most statutes are 1 to 3 years, so filing a lawsuit quickly is an important step in securing compensation. If you or a loved one experienced VOCs, organ damage, or death after taking Oxbryta, you should consult with an attorney as soon as possible. If you wait too long, you may lose your right to file a lawsuit. Early consultation with a legal team will ensure your claim is filed on time.
Statutes of limitations are different for wrongful death claims. If your loved one passed away and you think their death was caused by complications from Oxbryta, you should contact an attorney as soon as possible.
There are specific reasons the statute of limitations may be extended. If someone was incapacitated or a minor, they may have more time to file a case. If someone recently discovered their injuries were likely caused by Oxbryta, they also may have more time to file a case. If you are unsure if you have a case, contact our legal team immediately to discuss your eligibility.
Contact an Oxbryta Lawyer
At King Law, we are a team of experienced product liability attorneys. We specialize in representing people who were harmed by drugs that were supposed to help them. Our team specializes in Oxbryta lawsuits related to vaso-occlusive crises (VOCs) and other serious complications. We have extensive experience in handling pharmaceutical litigation. Our team delivers personalized and prompt service. We get to know you and your case so that we can work to secure you the best compensation available. We are committed to advocating for those affected by Oxbryta. We promise to handle your case with expertise and care, from gathering evidence to navigating settlement negotiations. There are no up-front costs for our services, and we only get paid if your case is successful.
Frequently Asked Questions (FAQs)
Sources and Citations
“About Sickle Cell Disease.” Centers for Disease Control and Prevention, 9 Aug. 2022.
“Accelerated Approval.” U.S. Food and Drug Administration.
Bryson, Steve. “EU Agency Confirms Continued Suspension of SCD Therapy Oxbryta.” Sickle Cell Anemia News, 21 Oct. 2025.
“A Phase 1/2 Ascending Dose Study and Open-Label Extension Study of Voxelotor in Patients with Sickle Cell Disease.” Blood, vol. 133, no. 17, 2019, pp. 1865–1875.
“A Phase 3 Randomized Trial of Voxelotor in Sickle Cell Disease.” The New England Journal of Medicine, vol. 381, no. 6, 2019, pp. 509–519.
“A Phase 3 Study to Evaluate the Efficacy and Safety of GBT440 in Subjects with Sickle Cell Disease (GBT-HOPE).” ClinicalTrials.gov, identifier NCT03036813.
“A Study to Evaluate the Safety and Pharmacokinetics of GBT440 in Pediatric Participants with Sickle Cell Disease (HOPE-KIDS 1).” ClinicalTrials.gov, identifier NCT02850406.
“Data and Statistics on Sickle Cell Disease.” Centers for Disease Control and Prevention, 17 May 2023.
“FDA Adverse Event Reporting System (FAERS) Public Dashboard.” U.S. Food and Drug Administration.
“FDA Approves Drug to Treat Sickle Cell Disease in Patients Aged 4 Up to 11 Years.” U.S. Food and Drug Administration, 17 Dec. 2021.
“FDA Approves Voxelotor for Sickle Cell Disease.” U.S. Food and Drug Administration, 25 Nov. 2019.
“FDA Is Alerting Patients and Health Care Professionals About the Voluntary Withdrawal of Oxbryta from the Market Due to Safety Concerns.” U.S. Food and Drug Administration, 26 Sept. 2024.
“GBT440 in Subjects with Sickle Cell Disease.” ClinicalTrials.gov, identifier NCT02285088.
Gaston, Jeanne L., et al. “Voxelotor: A Hemoglobin S Polymerization Inhibitor for the Treatment of Sickle Cell Disease.” International Journal of Molecular Sciences, vol. 22, no. 3, 2021, article 1572.
“MARAC Statement: Pfizer’s Voxelotor (Oxbryta®) Withdrawal.” Sickle Cell Disease Association of America, 28 Sept. 2024.
“NICE Requests More Evidence on Sickle Cell Disease Treatment Voxelotor.” The Pharmaceutical Journal, 27 July 2023.
“Oxbryta – Patient Information Leaflet.” Pfizer.
“Oxbryta – Referral.” European Medicines Agency, 2024.
“Pfizer Voluntarily Withdraws All Lots of Sickle Cell Disease Treatment OXBRYTA® (voxelotor) from Worldwide Markets.” Pfizer, 25 Sept. 2024.
“Pfizer’s Sickle Cell Woes Continue with Late-Stage Failure of Osivelotor.” FirstWord Pharma, 15 Aug. 2025.
“Resolution of Sickle Cell Leg Ulcers with Voxelotor (RESOLVE).” ClinicalTrials.gov, identifier NCT05561140.

