Complete the form below to see if you qualify for a Olympus Scope Lawsuit

Patients who suffered serious infections or sepsis after undergoing endoscopic procedures are filing lawsuits, claiming device manufacturers, like Olympus, are liable for their injuries.
Reusable endoscopes are used in many minimally invasive procedures to examine organs and perform internal operations. However, contaminated endoscopes can transfer potentially harmful bacteria between patients, leading to life-threatening conditions like sepsis. Although these infections are relatively rare on a per-procedure basis, persistent design and reprocessing issues with many of these complex devices have raised ongoing patient-safety concerns.
This page will discuss how endoscope-related infections occur, some of the devices of concern, and how to file a claim if you have suffered such an injury. This page is part of our broader investigation into Olympus scopes lawsuits, including infections allegedly caused by colonoscopes and duodenoscopes.
About the Endoscopy Infection Lawsuit
Endoscopy Infection Lawsuit News and Updates
What Is an Endoscopy Infection Lawsuit?
How Can Contaminated Endoscopes Cause Sepsis?
Which Endoscope Manufacturers Have Been Implicated in Infection Outbreaks?
What Evidence Links Endoscopes to Patient Infections?
What Types of Infections Are Associated With Contaminated Endoscopes?
What Are the Legal Theories Behind an Endoscopy Infection Lawsuit?
Who Can File an Endoscopy Infection Lawsuit?
What Are the Recoverable Damages in an Endoscopy Lawsuit?
How Is King Law Investigating Endoscopy Infection Claims?
Start a Free Consultation for an Endoscopy Infection Lawsuit
Endoscopy Infection Lawsuit News and Updates
January 2026: FDA Issues Class 1 Recall on Olympus Endoscope Accessory
The FDA issued a Class I recall on the ViziShot 2 FLEX, an aspiration needle manufactured by Olympus, after the device was linked to 40 injuries and one death. Devices with UDI 00821925043060, manufactured prior to May 12, 2025, pose a reasonable risk of causing serious injury in patients. The manufacturing flaw can cause the device to eject during a procedure, damaging internal tissues. A Class 1 recall is the FDA’s most serious type of recall, reserved for products that are deemed to pose a substantial risk of causing a serious adverse event.
June 2025: The FDA Issues Import Alerts on Multiple Olympus Devices, Including Automated Endoscope Reprocessors
The FDA issued an import alert on Olympus medical devices manufactured at the company’s Aizu facility, citing quality regulation violations. The import alert gives the FDA the authority to restrict future shipments of the named devices into the U.S. Devices named in the recall include automated endoscope reprocessors. While the alert does not force providers to stop using named Olympus devices they already own, the FDA does advise strictly following labeling and reprocessing procedures to lower the risk of adverse events.
2022-2023: The FDA Issues Two Warning Letters to Endoscope Manufacturer
The FDA’s Center for Devices and Radiological Health (CDRH) issued two warning letters concerning Olympus and its subsidiary, Aizu Olympus Co. following facility inspections. Despite its ongoing efforts to address issues related to endoscope reprocessing at Olympus, the CDRH found Olympus to be out of compliance with reporting and quality system requirements. Among the CDRH citations: Olympus did not develop MDR procedures, and failed to submit medical device requirements (MDRs) within the required timeframes.
2018-2019: Olympus Pleads Guilty To Distributing Misbranded Medical Devices, Pays $85 Million in Fines
Olympus pleads guilty to federal charges that it distributed misbranded medical devices and failed to report adverse events related to one of its endoscopes. The Newark Federal court fined the medical device manufacturer $80 million and an additional $5 million in asset forfeiture. Olympus had not filed adverse event reports after the TJF-Q180V was linked to “superbug” outbreaks in its European markets. Also pleading guilty is Olympus senior regulatory executive, Hisao Yabe, who had overseen the company’s Olympus Quality and Environment Division. Yabe would later be sentenced to a year of probation and a $5,000 fine.
December 12, 2018: Washington Family Secures Settlement in Endoscope Infection Case Against Olympus and Hospital
A family in Washington has won a wrongful death settlement in an endoscope infection case they brought against Olympus. The lawsuit alleges that their loved one developed a fatal superbug they believe was caused by bacteria trapped in the endoscope used during the procedure. According to their lawsuit, the endoscope’s design prevented the hospital from thoroughly disinfecting the device before using it on their loved one. The family won part of their case in the jury trial, and a new trial had been ordered after it was discovered that Olympus did not give the family access to important records like it was supposed to. The family reached a $6+ million settlement with Olympus and the hospital that performed the endoscope-assisted procedure.
2018: Study Finds Endoscopic Infection Rates Significantly Higher Than Expected
A John Hopkins
2006: Reprocessing Errors, Biofilm Blamed for Endoscope Infections
Insufficient cleaning and drying are blamed for Salmonella and Pseudomonas bacterial infections in a comprehensive review of medical literature titled,
What Is an Endoscopy Infection Lawsuit?
An endoscopy infection lawsuit refers to civil litigation where the plaintiff claims they developed a serious infection due to a contaminated endoscope. The complex design of some reusable endoscope devices can allow infection-causing pathogens to remain on and inside the scope in some cases, even when reprocessing guidelines are strictly followed.
The design of some endoscopes may make the devices very difficult to sterilize. People have filed lawsuits against endoscopy manufacturers.
Legal theories cited in endoscopy infection lawsuits include:
- Design defect
- Failure to warn
- Manufacturing defect
- Negligent reprocessing
- Failure to adopt safer technology (e.g., disposable components)
Many of these cases involve the medical device manufacturer Olympus, which was recently the subject of an FDA import alert due to ongoing quality control issues. The FDA has advocated for a “safer alternative design” for some endoscopes, including pushes for disposable components and single-use endoscopes.
Types of Endoscopy Lawsuits King Law is Investigating
King Law is investigating several types of endoscopy lawsuits. Endoscopes are used in a variety of procedures, and contaminated endoscopes can present an infection risk in any procedure they are used in. Here is a list of the types of endoscopy lawsuits King Law is investigating:
- Olympus scopes lawsuits
- Colonoscopy infection lawsuits
- Duodenoscope infection lawsuits (including ERCP infections)
- Bronchoscope infection lawsuits
- Other endoscope infection lawsuits
How Can Contaminated Endoscopes Cause Sepsis?
Contaminated endoscopes can cause sepsis by introducing bacteria into sterile tissue or the bloodstream, or by transmitting dangerous bacteria from one patient to another.
Sepsis, as defined by Sepsis-3 guidelines, is life-threatening organ dysfunction caused by a dysregulated response to an infection. Older patients and those with co-morbidities are more likely to experience sepsis from a bacterial infection. Additional complications may include bacteremia, cholangitis, pneumonia, or systemic inflammatory responses.
Although all reusable endoscopes present contamination risks, some types are associated with higher transmission rates than others.
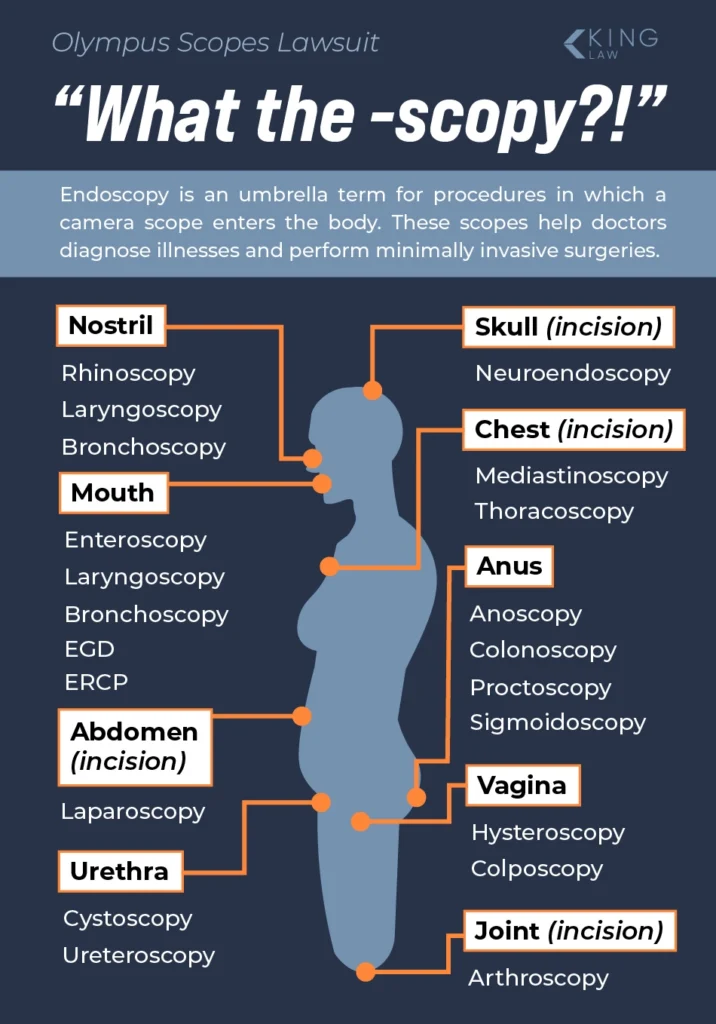
What Are Endoscopes?
According to the National Institutes of Health (NIH), an endoscope is a medical device that is inserted through an opening in the body. An endoscope has a camera attached to view inside the body. Any procedure using an endoscope is a type of endoscopy.
According to the NIH, an endoscope can be used in the following procedures:
- Anoscopy
- Arthroscopy
- Bronchoscopy
- Colonoscopy
- Cystoscopy
- Enteroscopy
- ERCP (endoscopic retrograde cholangiopancreatography)
- Laparoscopy
- Sigmoidoscopy
- Upper endoscopy
Because endoscopes come in contact with the inside of the body, any bacteria, viruses, or fungi on or inside the scope may enter the body. Once a pathogen enters the body, it can cause an infection.
What Role Do Biofilms and Residual Moisture Play?
Biofilm is a slimy, protective layer some bacterial colonies use to anchor themselves to surfaces and repel chemical attacks. Biofilm can form in the small channels of reusable endoscopes, especially when residual moisture is present.
The film allows colonies to resist high-level disinfectants like peracetic acid. Among the bacteria that create biofilm are carbapenem-resistant enterobacterales (CREs), which are resistant to antibiotics of last resort, making such infections very difficult to treat. Mechanical intervention, such as scrubbing and brushing, can provide defense against biofilm formation.
Can Infections Occur Even If Hospitals Follow Endoscope Cleaning Guidelines?
Endoscope infections can occur even in cases where hospitals or surgical centers strictly follow cleaning protocols. That is because the design of some endoscopes can make total disinfection very difficult to achieve.
For example, an
- Long, narrow channels that are too small for brushes to reach
- Hinges
- Sleeves surrounding components
- O-rings
- Valves that regulate fluid flow
- Devices that cannot be disassembled for reprocessing
- Lack of communication between the manufacturer and users
- Access to a manufacturer’s reprocessing instructions
- Quality of reprocessing instructions
These factors have shifted regulatory and legal scrutiny toward device limitations rather than just user error.
Which Endoscope Manufacturers Have Been Implicated in Infection Outbreaks?
Several endoscope manufacturers, including Olympus, Pentax, and Fujifilm, have been linked to infection outbreaks. These outbreaks have led to increased regulatory scrutiny and FDA recalls. Collectively, Olympus, Fujifilm, and Karl Storz are estimated to account for around 90% of the North American endoscope market as of 2024. Olympus, the subject of FDA import alerts in 2025, alone accounts for around 65% of the endoscope’s market global revenue share. Product design flaws and inadequate cleaning protocols produced by these companies remain an ongoing topic of concern.
Olympus Endoscope Infection Reports and FDA Actions
Much of the current concern around endoscopic infections began with adverse events associated with Olympus’s TJF-Q180V. The model would be linked to drug-resistant “superbug” outbreaks around the world between 2012 and 2015, leading to serious illnesses and deaths. FDA recalls would follow, with Olympus later facing an $80 million criminal fine and $5 million forfeiture for failing to report European adverse events involving the device to the FDA.
FDA action against Olympus would continue, revealing a pattern of alleged poor quality control and reporting violations across device categories, and culminating in a broad-reaching import alert in 2025. Infection risks for Olympus scopes remain an ongoing concern, even in newer models, leading the FDA to push for disposable end cap designs.
Pentax and Other Endoscope Models
Other endoscope manufacturers, including Pentax and Fujifilm, have also been linked to infection risks. For example:
- A design flaw in the Pentax ED-3490TK duodenoscope, where cracks and gaps in the distal cap’s adhesive can “lead to microbial and fluid ingress.”
- Pentax agreed to pay a $43 million fine in 2020 for shipping devices without approved instructions or adverse event reports of serious infections.
- Fujifilm, likewise, received an FDA warning letter regarding its failure to report adverse infection events related to its duodenoscopes.
These design flaws and procedural violations have left the companies open to product liability and negligence claims.
What Evidence Links Endoscopes to Patient Infections?
A strong body of medical evidence and research links some models of endoscopes to patient infections. A 2018 comparative study (called Rates of infection after colonoscopy and osophagogastroduodenoscopy in ambulatory surgery centres in the USA) found that, although uncommon, rates of post-endoscopy infections were substantially higher than previously believed.
The study found that infection affected 3 of every 1,000 duodenoscopy patients, 15.6 out of every 1,000 bronchoscopy patients, and 1.1 out of every 1,000 colonoscopy screening patients. Endoscope-related infections may be identified through methods such as:
- Epidemiological linking
- Outbreak clusters of rare, drug-resistant CREs
- Scope cultures
- Genome sequencing
How Lawyers Can Prove Endoscopy Infection Cases
Lawyers can request medical and device records to prove
- Chain of custody forms (scope reprocessing tracking)
- Culture testing
- Genome sequencing linking infections
Your attorney will work to provide proof on your injuries and a possible association with the endoscope used in your procedure.
What Types of Infections Are Associated With Contaminated Endoscopes?
Studies, including a
These organisms can cause bloodstream infections, pneumonia, urinary tract infections, and sepsis. These organisms can remain in the small, moisture-capturing channels of reusable endoscopes and can resist chemical disinfectants through the creation of biofilm.
Some of the organisms of high concern that have been found on endoscopes even after reprocessing include:
- Carbapenem-Resistant Enterobacterales (CRE)
- Klebsiella pneumoniae
- Pseudomonas aeruginosa
- Various multidrug-resistant organisms (MDROs)
Carbapenem-Resistant Enterobacterales (CRE)
Carbapenem-Resistant Enterobacterales (CRE) are so named because they are resistant to the antibiotics of last resort (i.e., carbapenems) and are sometimes referred to as “superbugs.” The classification includes strains of bacteria belonging to the Klebsiella pneumoniae, E. coli, and Enterobacter types. Treating these infections often requires case-by-case discretion and the application of newer drugs or combination therapies.
Klebsiella Pneumoniae and OXA-181 Strains
Several of the CRE outbreaks linked to the Olympus TJF-Q180V involved Klebsiella pneumoniae. Klebsiella pneumoniae is commonly found in human intestinal flora, where it is generally considered harmless in small amounts. However, it can act as an opportunistic pathogen if it spreads outside of the gut. It can show substantial resistance to antibiotics, especially strains with the gene to produce OXA-181, a carbapenemase enzyme that protects it from carbapenem antibiotics.
Pseudomonas Aeruginosa
Another bacterium linked to endoscope infections is Pseudomonas aeruginosa. While P. aeruginosa rarely carries the OXA-181 trait, it is naturally resistant to many drugs and can form hardy biofilm. It is commonly found in water, soil, and other surfaces. Like Klebsiella, it is an opportunistic pathogen.
Multidrug-Resistant Organisms (MDROs)
Multidrug-resistant organisms (MDROS) refer to a wide range of microorganisms that have developed resistance to one or more classes of antimicrobial agents. Most MDROs are bacteria, but the term can also describe organisms like the Candida auris fungus. While these infections are symptomatically similar to those of non-resistant strains, they are more difficult to treat, sometimes leading to higher mortality and longer hospital stays.
What Are the Legal Theories Behind an Endoscopy Infection Lawsuit?
Endoscope infection lawsuits generally rely on existing product liability and/or negligence frameworks to hold device manufacturers accountable for injuries allegedly tied to the company’s devices. These legal theories are used to argue that the infection was in part the result of human activity or negligence.
Here are some of the legal theories that could be applied in endoscopy infection lawsuits:
- Design defect
- Failure to warn
- Manufacturing or quality control failure
- Negligent reprocessing by healthcare facilities
- Wrongful death due to gross negligence
These and other legal theories may be used as allegations in Olympus endoscope lawsuits.
Design Defect
A design defect means that, even when the product is correctly manufactured to its exact intended design, it possesses a fundamental flaw in its design that makes it dangerous to use. The elevator mechanism of the Olympus TJF-Q180V or the gaps in the distal caps of the Pentax ED-3490TK would both be examples of alleged design flaws that raise the risk of infection.
Failure to Warn
Plaintiffs may allege that device manufacturers were aware of the elevated infection risks of using their endoscopes, but they failed to adequately warn medical professionals and patients. Olympus, for example, pleaded guilty to failing to file adverse event reports in the U.S., despite being aware of duodenoscope-related infections in its European markets. Failure to warn deprives consumers of informed consent.
Manufacturing or Quality Control Failures
Manufacturing or quality control failures introduce dangerous flaws into the product that are not a part of its design. These failures may be confined to a particular shipment of the product or be a more persistent feature of the supply chain. The FDA’s import alert on Olympus products was the end result of years of quality control issues at the company’s Aizu manufacturing site, which not only contributed to increased infection rates but also risks of direct tissue injury and burns.
Negligent Reprocessing by Healthcare Facilities
Reusable endoscopic devices require extensive reprocessing to minimize infection risks, beginning with prompt mechanical or manual scrubbing shortly after use to prevent biofilm build-up. If these reprocessing steps are skipped or rushed, hospitals may be liable for the infection. If they are improperly or unclearly documented, the manufacturer may be liable.
Wrongful Death Claims
In instances where the patient died as a result of complications from their endoscope-related infection, such as sepsis, a family member may file a wrongful death claim due to negligence on the part of the manufacturer. Wrongful death claims rely on many of the same legal theories as personal injury cases, but differ with regard to who files the claim and some of the specific damages sought. For example, a wrongful death claim may seek to recoup funeral costs.

Who Can File an Endoscopy Infection Lawsuit?
King Law uses the following criteria to determine eligibility for endoscopy infection lawsuits:
- You had a medical procedure that utilized an endoscope or an endoscopic accessory.
- You developed sepsis or an infection that could be related to a contaminated endoscope.
- You had no history of such an infection within a year prior to the procedure.
You may still qualify even if the cause of your infection is unclear. Our lawyers can review your case and offer clarification.
Additionally, clients need to file their case within their jurisdiction’s statute of limitations. These deadlines can vary significantly, so King Law strongly advises consulting with an attorney promptly after you become aware of your injury.
What Are the Recoverable Damages in an Endoscopy Lawsuit?
Plaintiffs who file endoscopy lawsuits may be eligible for both economic and non-economic damages. While the amount and applicability of each of these examples will vary from case to case, damages may include:
- Medical expenses related to treating the infection
- ICU hospitalization costs
- Long-term organ damage
- Lost income and reduced earning potential
- Pain and suffering
- Funeral costs (wrongful death)
- Loss of companionship (wrongful death)
The amount awarded in each of these categories may vary depending on the extent of the injury and any state limits on damages.
How Is King Law Investigating Endoscopy Infection Claims?
King Law is currently investigating endoscopy infection claims nationwide. Our experienced attorneys are evaluating medical records, procedure logs, manufacturer documentation, and regulatory actions to evaluate whether contaminated endoscopes contributed to clients’ infections or sepsis. A typical investigation will involve the collection and review of:
- The scope model used in the procedure
- Reprocessing documentation
- FDA safety communications involving the device
- Hospital infection control audits
Prospective clients are encouraged to bring any information they have available regarding their endoscope infections to their free consultation. Our team can also help gather applicable medical records on your behalf.
Start a Free Consultation for an Endoscopy Infection Lawsuit
If you or a family member has suffered from a serious infection or sepsis following an endoscopic procedure, contact King Law today at (585) 496-2648 for a free consultation with no obligations. Our attorneys have decades of experience holding medical device manufacturers accountable for negligent practices and work on a contingency basis, so there’s no upfront cost to pursuing compensation for your injuries. You only pay us for our services if you receive compensation for your injuries.
Frequently Asked Questions (FAQs)
Sources and Citations
Here is a list of sources used in this article.
“Infection Prevention and Control in Endoscopy.” Australasian College for Infection Prevention and Control, 2025.
“Human Factors in Endoscope Reprocessing.” Ambu USA, n.d.
ASGE Standards of Practice Committee. “Multisociety Guideline on Reprocessing GI Endoscopes.” American Society for Gastrointestinal Endoscopy, 2021.
Centers for Disease Control and Prevention. “CDC Statement on CRE Outbreak Associated with Duodenoscopes.” 2015.
Centers for Disease Control and Prevention. “About Carbapenem-Resistant Enterobacteriaceae (CRE).” n.d.
Centers for Disease Control and Prevention. “Multidrug-Resistant Organisms (MDRO) Management.” n.d.
Cornell Law School, Legal Information Institute. “Design Defect.” n.d.
Epstein, L., et al. “Transmission of Carbapenem-Resistant Enterobacteriaceae via Duodenoscopes.” Clinical Infectious Diseases, vol. 65, no. 7, 2017, pp. 1159–1165. https://doi.org/10.1093/cid/cix483
Food and Drug Administration. “Duodenoscope Reprocessing and Infection Risk.” 2015.
Food and Drug Administration. “FDA Issues Warning Letters to Endoscope Manufacturer.” n.d.
Food and Drug Administration. “Import Alert for Olympus Medical Devices.” n.d.
Food and Drug Administration. “Endoscopic Aspiration Needle Recall (Olympus).” n.d.
Food and Drug Administration. “Factors Affecting Quality of Reprocessing.” n.d.
GARDP. “Meet Pseudomonas aeruginosa.” Global Antibiotic Research & Development Partnership, n.d.
Kovaleva, J., et al. “Infectious Risks of Duodenoscopes.” Clinical Microbiology Reviews, 2014. https://doi.org/10.1128/CMR.00085-13
LFM Healthcare Solutions. “Proposal on Endoscope-Associated Infections Reporting.” 2025.
Muscarella, L. F. “Biofilm Formation in Endoscopes.” World Journal of Gastrointestinal Endoscopy, 2016. https://doi.org/10.4253/wjge.v8.i7.246
Nanosonics. “Biofilm and Endoscope Reprocessing.” n.d.
Ofstead, C. L., et al. “Transmission of Infection via Endoscopes.” American Journal of Infection Control, 2015. https://doi.org/10.1016/j.ajic.2014.12.003
Olympus Corporation. “Olympus Medical Systems News Release.” 2025.
Rex, D. K., et al. “Duodenoscope-Associated Infection Transmission Study.” JAMA, 2014. https://doi.org/10.1001/jama.2014.1234
Reuters. “Ex-Olympus Executive Avoids Prison in Scope Infection Case.” 2019.
U.S. Department of Justice. “United States v. Olympus Medical Systems Corporation.” 2018.
U.S. Department of Justice. “Olympus Executive Pleads Guilty.” 2019.
U.S. Department of Justice. “Pentax Medical Settlement.” 2023.
van der Starre, C., et al. “Endoscope-Associated Infection Risk Study.” Gastrointestinal Endoscopy, 2018. https://doi.org/10.1016/j.gie.2018.0

