See if You Qualify

While Ozempic has shown promising results for the treatment of diabetes and weight management, it has also gained attention for severe side effects. One of those side effects is a type of vision loss experienced by some patients. A 2024 study published in JAMA Ophthalmology found an increased risk of NAION for those taking semaglutide medications. NAION is a rare eye condition that causes sudden, often irreversible, blindness or vision loss in one eye. As a result, some are pursuing legal action through Ozempic blindness lawsuits.
On December 15, 2025, the Judicial Panel on Multidistrict Litigation centralized lawsuits alleging vision loss from GLP-1 drugs, like Ozempic. The litigation is called: MDL -3163 IN RE: Glucagon-like Peptide-1 Receptor Agonists (GLP-1 RAs) Non-Arteritic Anterior Ischemic Optic Neuropathy Products Liability Litigation. The lawsuits are centralized in Pennsylvania federal court under Judge Karen Spencer Marston. People who were diagnosed with NAION-related vision loss after taking Ozempic can talk to an attorney about filing as part of this federal litigation.
Ozempic Blindness Lawsuit Overview
Ozempic blindness lawsuits claim that the drug’s manufacturer, Novo Nordisk, failed to adequately warn consumers about the risk of vision loss caused by non-arteritic anterior ischemic optic neuropathy (NAION). There are several other eye issues that may warrant legal action, also linked to semaglutide medications like Ozempic. These lawsuits can highlight the impact of blindness and other eye injuries on the consumer’s mental health, ability to work, financial stability, and more. Ozempic lawsuits can hold Novo Nordisk accountable for any wrongdoings and provide plaintiffs with compensation to help with medical bills and other damages.
Ozempic Blindness Lawsuit Updates
Stay up to date on the latest litigation for Ozempic and vision loss/blindness.
April 1, 2026: More Lawsuits Join Vision Loss MDL for GLP-1 Drugs, Like Ozempic
As of April 1, 2026, there are 73 pending lawsuits in the centralized litigation for people alleging NAION from GLP-1 drugs, including Ozempic. This litigation is moving forward. Judge Marston and attorneys are preparing for the Science Day, which is still scheduled for June 2, 2026. Topics to be discussed at this hearing include: background on the diseases treated using GLP-1 drugs; GLP-1 drugs and their labeling and regulatory history relevant to NAION; a background on the alleged in this litigation (NAION); and the science/medical literature related to GLP-1 drugs and NAION. The science behind Ozempic and NAION will be examined by the judge.
March 9, 2026: Man Shares Story About Vision Loss After Taking Ozempic
A man in Maryland named Todd Engel is sharing his story about the vision loss he experienced after he took Ozempic. According to Engel, he started taking Ozempic in 2023, after his doctor prescribed the drug to help him manage his diabetes. A few months later, he lost vision in one of his eyes and was diagnosed with NAION. Ten months later, he lost vision in his other eye, leaving him legally blind. Engel’s neuro-ophthalmologist shared with him that there is a possible connection between semaglutide drugs like Ozempic and an increased risk of NAION. Engel has since filed a lawsuit, alleging that Novo Nordisk was aware of an increased risk of NAION, and he was not warned about this possible association.
March 4, 2026: Science Day Scheduled in GLP-1 Drug MDL Representing Vision Loss Claims
Many people have filed lawsuits alleging that Ozempic or another GLP-1 drug caused their vision loss. A federal MDL has been created for people diagnosed with NAION after taking Ozempic or another GLP-1 drug. Now, Judge Karen Spencer Marston has scheduled a Science Day in this litigation for June 2, 2026. Science Days are an opportunity for attorneys on both sides to educate the judge on the science behind alleged injuries in a civil lawsuit. Attorneys representing plaintiffs will be presenting evidence that says Ozempic and other semaglutide drugs may increase the risk of NAION and associated vision loss. The defense will be able to present opposing evidence in a non-adversarial format. People alleging vision loss from Ozempic can talk to an attorney about potentially joining this MDL.
February 10, 2026: Clinical Study of Diabetic Retinopathy in Patients Taking Ozempic, Other Semaglutide Drugs Underway
A large, 5-year clinical trial is being conducted to study diabetic retinopathy in patients taking semaglutide (like Ozempic) for diabetes. Studies suggest that diabetes may be associated with increased rates of eye diseases and conditions, like retinopathy. The clinical trial study is slated to run until November of 2027. This new study may provide important insights into how a semaglutide like Ozempic can affect vision outcomes in patients with diabetes. Some Ozempic patients have filed legal complaints against Novo Nordisk, claiming that the semaglutide caused them to develop a separate vision disorder called NAION (which can cause sudden blindness).
January 6, 2026: Judge Begins Centralization and MDL Processes for Ozempic Vision Loss Lawsuits
People who took Ozempic and other GLP-1 drugs and experienced NAION (a sudden partial blindness, usually in one eye) can now join a centralized litigation. This MDL is in its very early stages. Ozempic vision loss claims are centralized under U.S. Judge Karen Marston in the Eastern District of Pennsylvania. She will oversee the steps in the MDL process. First, the judge will appoint lead attorneys and set an initial trial schedule. Then, parties on both sides will begin collecting and exchanging information, which is a process known as discovery. Although this litigation is young, people who experienced vision loss after taking Ozempic can talk to an attorney about joining and pursuing a settlement for their injuries.
December 15, 2025: Ozempic Blindness MDL Is Created for Patients Who Suffered Eye Stroke After Taking GLP-1s
Patients diagnosed with non-arteritic anterior ischemic optic neuropathy (NAION) after taking Ozempic, other GLP-1s, can file a lawsuit in the new MDL created for these cases. MDL No. 3163 (IN RE: Glucagon-like Peptide-1 Receptor Agonists (GLP-1 RAs) Non-Arteritic Anterior Ischemic Optic Neuropathy Products Liability Litigation) is active and exclusively for claims involving partial blindness/vision loss caused by GLP-1 use. People taking Ozempic have experienced unexpected vision loss, which can occur suddenly due to a stroke behind one or both eyes. People who suffer from NAION from Ozempic typically cannot restore their vision after they lose their sight. Ozempic’s manufacturer, Novo Nordisk, did not tell people that NAION-related blindness is a potential side effect, and affected patients are filing lawsuits about this. Patients who have not already filed an Ozempic blindness claim are encouraged to do so by contacting a lawyer.
December 4, 2025: JPML Hears Arguments for Creating Federal Group Lawsuit for People Who Lost Their Vision After Taking Ozempic
One of the alleged side effects of Ozempic is a type of vision loss called non-arteritic anterior ischemic optic neuropathy (NAION). On December 4, the Judicial Panel on Multidistrict Litigation (JPML) heard arguments on whether or not to create a group lawsuit for people who say Ozempic and other GLP-1 drugs caused them to develop NAION. If the JPML decides to create this litigation, people who experienced vision loss after taking Ozempic will have new legal options.
December 2, 2025: Another Woman Reports Losing Sight After Taking Ozempic
A woman named Cilette Mitchell lost vision in both of her eyes months after she started taking Ozempic and one other GLP–1 drug. Three months into her Ozempic regimen, she began seeing dark, cloudy spots in her vision. Her eye doctor recommended that she get an MRI immediately, which revealed that a stroke had occurred behind her right eye. Less than a month later, Cilette lost her vision in her left eye, too, from another eye stroke. Cilette believes that Ozempic is the culprit. In her lawsuit, Cilette claims vision loss was not something she was warned about, and that Novo Nordisk should have included this warning on Ozempic’s label.
November 5, 2025: Doctor Who Did Initial Study on Ozempic and Vision Loss Discusses How It Occurs
Dr. Rizzo was one of the physicians and researchers who initially investigated how Ozempic might cause a type of partial blindness called NAION. He and other researchers at Mass General Brigham Hospital went on to publish a study that would spur other investigations. Dr. Rizzo shared information on the ways Ozempic and other GLP-1 drugs might cause NAION and vision loss. He suspects that rapid changes in blood sugar and blood pressure may be contributing factors. He also thinks that prior changes to the eyes caused by diabetes may put people at a higher risk. Dr. Rizzo and fellow team members are conducting a trial to better understand how Ozempic might cause increased rates of NAION.
October 3, 2025: Man in Maryland Becomes Blind After Taking Ozempic to Manage His Diabetes
Like many others, Todd Engel took the semaglutide Ozempic to help him keep his Type 2 diabetes under control. Around four months after he started taking Ozempic, Todd lost his vision. His doctors told him he had developed non-arteritic anterior ischemic optic neuropathy (NAION), which is vision loss that is sudden, painless, and irreversible in most cases. Todd discovered that his Ozempic use may have caused his NAION, and he is filing a lawsuit against Novo Nordisk for compensation.
September 16, 2025: New Possible Lawsuit Option for People Who Experienced Ozempic-Related Vision Loss
More medical studies are finding that people who take semaglutide drugs like Ozempic are at an increased risk of a type of blindness caused by NAION (non-arteritic anterior ischemic optic neuropathy). For example, a 2025 study in the Journal of the American Medical Association (JAMA) found people taking Ozempic were about 1.8 times as likely to experience vision loss from NAION, than those prescribed other diabetes medications. In an unusual move, defendants in the GLP-1 group lawsuit have asked for a consolidation of lawsuits filed by people who have experienced blindness. Defendants in the current group lawsuit against GLP-1 drugs have asked for NAION cases to be consolidated in the same federal court in Pennsylvania and under the same judge. This could point to the defendants’ understanding that their GLP-1 drugs can indeed lead to vision loss in some patients.
August 1, 2025: Possible New Legal Options For People Suffering From Blindness Related to Ozempic
A growing body of research shows that Ozempic and other GLP-1 drugs can increase the risk of a type of vision loss called NAION (non-arteritic anterior ischemic optic neuropathy). Now Ozempic patients who suffer from this condition may have new options for filing lawsuits. A request has been filed in New Jersey state courts to consolidate lawsuits related to NAION and GLP-1 drugs. If approved, a type of group lawsuit called a multicounty litigation (MCL) will be created. This will allow people who took Ozempic and developed NAION to join together in litigation against Novo Nordisk and other GLP-1 manufacturers.
June 6, 2025: Novo Nordisk Asked to Add NAION, Which Can Cause Blindness, to Ozempic Warning Label
The European Medicines Agency has concluded that taking Ozempic and other medications that contain semaglutide may increase someone’s risk of developing NAION. NAION is an eye condition that can develop without warning and cause irreversible blindness in one or both eyes. The EMA has asked Novo Nordisk, which manufactures Ozempic, to add NAION to the list of side effects that can occur with “very rare frequency.” However, the US label for Ozempic does not contain this side effect. Thousands of lawsuits have been filed by Ozempic patients who have experienced a range of complications, including blindness.
September 16, 2024: Blindness and Vision Loss Becoming A Growing Concern in the Ozempic Lawsuits
The American Association of Ophthalmology suggests that Ozempic and other GLP-1 drugs increase the risk of cause blindness or vision loss in several ways. The most common vision problem associated with Ozempic and other semaglutides is diabetic retinopathy. Other, more sudden changes in vision, like eye strokes and NAION, can lead to blindness, particularly in one eye. We expect future studies on vision loss associated with Ozempic use to be an important part of the Ozempic vision loss lawsuits.
July 14, 2024: Ozempic Lawsuit Intensifies with New Studies Linking Drug to Vision Loss
The Ozempic blindness and vision loss lawsuit is full steam ahead following the July 2024 study indicating huge increases in the risk of NAION for users of semaglutide. Another study worth considering is the 2020 study that found over 140 adverse vision effects from the use of semaglutide, the active ingredient in Ozempic. The study was published in Investigative Ophthalmology and Visual Science in June 2020. This study closely assessed the results of the Ozempic clinical trial and was also relied on for a previous lawsuit involving a Northeast Ohio woman who experienced sudden blurred vision after her third dose of Ozempic. We expect an application to include vision loss cases in the federal lawsuit shortly.
July 2024: New Study Connects Ozempic to Blindness
A rare type of partial blindness has emerged as a prominent concern for Ozempic patients following new findings that link semaglutide to sudden, often permanent vision loss in one eye. This study has raised questions about why Novo Nordisk has not warned patients about such serious side effects, similar to concerns regarding stomach paralysis when taking the medication.
January 2024: Researchers Examine Eye Risks Associated with Ozempic
A systematic review of patients taking semaglutide revealed multiple primary and secondary outcomes. Notably, blurred vision, retinopathy, and other macular complications were identified as exploratory outcomes, indicating the need for further research to fully understand all the risks associated with this medication.
August 2021: Ozempic Could Be Linked to Increased Risk of Diabetic Retinopathy
Study results examining diabetic retinopathy (DR) in patients taking semaglutide found “a higher risk for DR complications in patients with proliferative and non-proliferative DR at baseline.” However, researchers acknowledge that worsening DR could be due to rapid drops in blood glucose levels from diabetic medications.
June 2020: Initial Findings Connect Ozempic to Diabetic Retinopathy and Other Ocular Issues
A study found that Ozempic is linked to diabetic retinopathy and adverse ocular events more frequently than other GLP-1 receptor agonists. Reported cases of diabetic retinopathy and adverse ocular events for Ozempic were 16.4% and 1.1%, respectively. In comparison, other diabetic medications showed a range of 2.3%-6.3% for diabetic retinopathy and 0.05%-0.19% for adverse ocular events.
About the Ozempic Blindness Lawsuit:
What Is Blindness From Ozempic?
How Ozempic Causes Blindness and Vision Problems
New Study Links Ozempic and Rare Eye Disease NAION
Percentage of Ozempic Users Reporting Blindness
Ozempic Manufacturer’s Response to NAION Allegations
How to File an Ozempic Blindness Lawsuit
What Is Blindness From Ozempic?
A recent study conducted by the Harvard teaching hospital Mass Eye and Ear found an increased risk of developing non-arteritic anterior ischemic optic neuropathy (NAION) in individuals taking Ozempic. NAION is an eye condition caused by decreased blood supply to the optic nerve. It causes sudden, painless vision loss in one eye with irreparable damage to the nerve and impacts the eye’s connection to the brain. Often referred to as an “eye stroke,” NAION does not currently have effective treatment options.
NAION occurs at an estimated rate of 2.3-10.2 per 100,000 people in the United States above the age of 50. However, it may become more prevalent with the popularity of Ozempic. According to the Mass Eye and Ear study:
- 8.9% of diabetic patients taking semaglutide received a NAION diagnosis.
- 1.8% of diabetic patients taking other medications received a NAION diagnosis.
- 6.7% of overweight or obese patients taking semaglutide received a NAION diagnosis.
- 0.8% of overweight or obese patients taking other medications received a NAION diagnosis.
- Patients with diabetes who took semaglutide were more than four times more likely to develop NAION than those on other medications.
- Patients who were obese and took semaglutide were more than eight times more likely to develop NAION than those on other medications.
Because of how serious NAION is, doctors are encouraged to let their patients know that it’s a potential side effect of Ozempic. Patients should also monitor closely for any potential eye symptoms and seek medical care immediately should they notice anything abnormal. While more research is needed to further understand NAION and Ozempic, the results are strong enough to warrant action and attention.
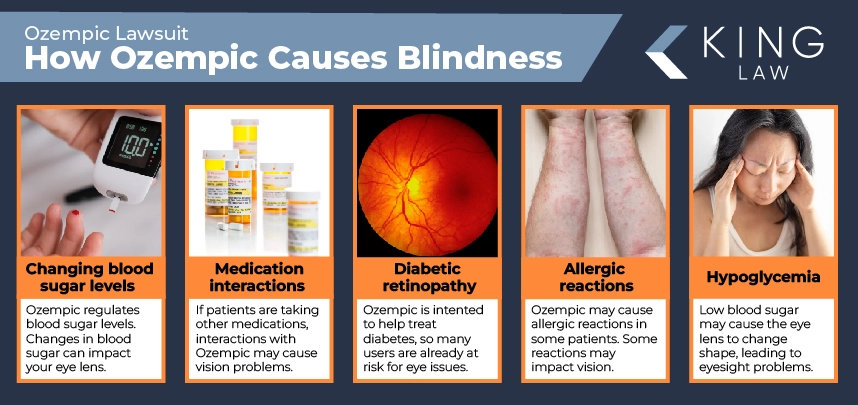
How Ozempic May Increase the Risk Blindness and Vision Problems
Additional research is needed to confirm and expand upon the Mass Eye and Ear study. Right now, it’s unclear how Ozempic may increase the risk of vision loss caused by NAION. Potential causes include:
- Changing blood sugar levels: Ozempic helps regulate blood sugar. Changes in blood sugar can impact your eye lens, causing blurry vision and damage to blood vessels in the retina.
- Diabetic retinopathy: Ozempic is intended to help treat diabetes, so many Ozempic users are already at a heightened risk for some eye conditions, including diabetic retinopathy, which impacts vision.
- Hypoglycemia: Ozempic can help patients with blood sugar issues. If blood sugar is low (hypoglycemia), the eye lens may change shape, leading to blurry vision and other problems with eyesight.
- Medication interactions: If Ozempic patients are taking other medications, which can be coming for diabetes, vision issues could be a side effect of drug interactions.
- Allergic reactions: Ozempic may cause an allergic reaction in some patients. Depending on the type of reaction, vision may be impacted.
To help prevent or treat these issues, patients should let their healthcare provider know that they are taking Ozempic. They should also maintain regular eye examples and monitor blood sugar to identify large fluctuations. Patients should seek medical care as soon as possible if they experience any adverse reactions to Ozempic.
Ozempic Eye Side Effects
Closely managing diabetes and monitoring eye health can help healthcare providers identify any conditions early on. Patients may need to discontinue Ozempic or seek additional treatments. Here is a comprehensive list of potential eye-related side effects for diabetic patients taking Ozempic:
Non-arteritic Anterior Ischemic Optic Neuropathy (NAION)
Non-arteritic anterior ischemic optic neuropathy (NAION) is caused by compromised blood flow to the eye’s optic nerve. This can cause permanent damage to the front of the optic nerve (ischemia), which is responsible for your vision as it communicates between the eye and the brain. Symptoms of NAION can include:
- Sudden, mild to severe vision loss in one eye
- Blurred vision
- Color distortion
- Light sensitivity
- Dark area or shadow in vision
- Loss of peripheral vision
- Disc swelling (visible through eye exam)
Potential risk factors of NAION include:
- Older than 50 years old
- Diabetes
- Hypertension
- High cholesterol
- Sleep apnea
- Some anatomical predispositions
- Medications like semaglutide (Ozempic) and sildenafil (Viagra)
Because Ozempic is intended to help manage blood sugar levels in diabetic patients, changing blood sugar can lead to eye damage and vision changes.

New Study Links Ozempic and Rare Eye Disease NAION
The first study researching Ozempic and blindness looked at the medical records of more than 16,000 patients from 2017 to 2023 after Ozempic was released. Some patients were taking Ozempic for diabetes and others for weight management. Both groups were compared to identify a potential correlation between semaglutide and NAION.
Researchers concluded, “Our main finding is that prescribed semaglutide is associated with an increased risk of NAION.” Diabetic patients who took semaglutide were more than four times more likely to develop NAION than those on non-GLP-1 medications. Obese patients who took semaglutide were more than eight times more likely to develop NAION than those on non-GLP-1 medications.
While the study didn’t confirm that Ozempic directly causes NAION, it was significant enough for researchers to suggest patients be warned about the potential side effect. Researchers also acknowledged the need for more research that could include a larger, randomized sample group for a longer period.
Percentage of Semaglutide Users Reporting Vision Loss or Partial Blindness
Study results indicate an increased risk of non-arteritic anterior ischemic optic neuropathy (NAION) in patients using semaglutide for Type 2 diabetes and obesity. These conclusions are based on a study involving 710 patients with Type 2 diabetes. Here are some of the findings of the JAMA study:
- Taking a semaglutide to treat type 2 diabetes is associated with a four times higher risk of developing NAION compared to other medications.
- Specifically, 8.9% of diabetic patients who were administered semaglutide developed NAION, in contrast to only 1.8% of patients taking other medications.
- Taking semaglutide for obesity treatment has an incidence rate of NAION seven times higher than with other medications.
- Specifically, in the group of patients prescribed semaglutide for weight loss, 6.7% developed NAION compared to just 0.8% of those on other treatments.
These findings showcase the need for careful consideration and monitoring of patients prescribed semaglutide, given the elevated risks of serious eye conditions associated with its use.
Ozempic Manufacturer’s Response to NAION Allegations
Novo Nordisk, like many other pharmaceutical companies that have faced backlash for not disclosing serious health risks, has pushed back against such claims. News outlets have reported that the company’s response has been “unperturbed,” dismissing concerns about possible vision loss after taking Ozempic and reiterating that the data is insufficient to show a strong connection between semaglutide and NAION.
A Novo Nordisk spokesperson pointed out that NAION is not listed on approved labels as an “adverse drug reaction for the marketed formulations of semaglutide.” They also noted limitations in the Mass Eye and Ear study, such as its small and non-randomized sample group. The spokesperson emphasized that “patient safety is a top priority for Novo Nordisk, and we take all reports about adverse events from our medicines very seriously.”
How to File an Ozempic Blindness Lawsuit
To file an Ozempic blindness personal injury lawsuit:
- Set up an initial consultation with an experienced attorney to review your case and confirm eligibility. They will also help you understand the timeline for when you must file to be eligible for compensation.
- Begin gathering evidence to prove you took Ozempic and developed NAION.
- Be available to your attorney as they build and file your case with the appropriate court.
- Follow your attorney’s guidance if presented with an option to reach a settlement, negotiate, or go to trial.
- If a settlement isn’t reached, your case will be presented in trial.
- If the case settles in your favor, you will receive compensation that can be used for medical bills and other damages.
Again, be prepared to collect as much evidence as possible such as medical records, testimonies, doctor’s notes, medical bills, prescription information, and more to detail your experience taking Ozempic and resulting health issues plus their financial impact.
Contact an Ozempic Lawyer for Vision Loss
Reach out to an Ozempic lawyer like King Law as soon as possible. Filing an Ozempic lawsuit requires adherence to state-specific statutes of limitations, which often only allow 2-3 years from the date of your diagnosis or injury to file a claim. To ensure you don’t miss your opportunity for justice and compensation, it is crucial to act promptly.
Contact a lawyer who has extensive experience in handling cases involving Ozempic or other semaglutide drugs, particularly those linked to serious side effects such as blindness/vision loss, stomach paralysis, and intestinal blockages. An experienced lawyer can guide you through the legal process, helping you navigate the complexities of your case and maximize your chances of a successful outcome.
Frequently Asked Questions (FAQs)
Learn more about how Ozempic impacts the eyes with answers to these frequently asked questions.