Complete the form below to see if you qualify for a Olympus Scope Lawsuit

Patients are filing colonoscopy infection lawsuits, alleging that contaminated scopes caused them to develop sepsis or superbug infections. These patients claim a design flaw in the scopes exposed them to potentially dangerous bacteria, leading to serious infections. These infections can occur when bacteria is transferred from one patient to another through a contaminated colonoscope or an attached accessory.
Lawsuits filed against colonoscope manufacturers allege that a design defect kept reusable scopes from being properly disinfected between uses. Without proper disinfection, colonoscopes can transmit pathogens into the body. Additionally, lawsuits accuse medical device makers of not properly disclosing information about this defect or issuing the proper physician and patient warnings.
This page will explain the details of colonoscopy lawsuits, including how scope-linked transmissions occur, regulatory responses to injury reports, and who may qualify to file a lawsuit against scope manufacturers. Many colonoscopy infection lawsuits stem from broader Olympus scope litigation, which includes endoscope claims and duodenoscope claims.
About the Colonoscopy Infection Lawsuit:
Colonoscopy Infection Lawsuit | News and Updates
What Is a Colonoscopy Infection Lawsuit?
How Can Colonoscopes Cause Sepsis or Other Infections?
What Infections Have Been Linked to Scopes Used in Colonoscopies?
CDC and FDA Actions Related to Contaminated Colonoscopes and Endoscopes
Why Does Scope Reprocessing Sometimes Fail?
Are Colonoscope-Linked Infection Outbreaks Rare?
What Are the Symptoms of Infection After a Colonoscopy?
How Do You Prove a Contaminated Olympus Colonoscope Caused Sepsis?
What Legal Theories Apply in a Colonoscopy Infection Lawsuit?
Who Qualifies to File a Colonoscopy Infection Lawsuit?
When Should You Contact a Lawyer After a Colonoscopy Infection?
What Compensation Is Available in a Colonoscopy Infection Lawsuit?
What Should You Do If You Develop an Infection After a Colonoscopy and Want to File a Lawsuit?
King Law Is Investigating Olympus Colonoscopy Infection Claims
Start Your Free Colonoscopy and Sepsis Case Review Today
Colonoscopy Infection Lawsuit | News and Updates
June 2025: FDA Issues Import Alert on Olympus Medical Devices, Scopes
Due to ongoing Quality System regulation violations, the FDA issued an import alert on numerous Olympus medical devices manufactured at the company’s Aizu site in Japan. The import alert gives the FDA the authority to prevent future delivery of the named devices into American markets. While the list of devices mainly includes bronchoscopes, laparoscopes, and ureterorenoscopes, it impacts a broad range of endoscopic products, including those used in colonoscopies.
January 16, 2025: Olympus Accessory Triggers Class I Recall
The FDA posts a Class 1 recall for the Olympus MAJ-891 Forceps/Irrigation Plug, a reusable accessory for the company’s endoscopes, including colonoscopes. The recall was initiated by Olympus on December 18, 2024. The recall warns of the risk of transferring biomaterial between uses when the device is not effectively reprocessed (i.e., sanitized and readied). The device is linked to 120 infection-related injuries, as well as one death. Insufficient reprocessing of the MAJ-891 can “result in infection, urinary tract infection, or sepsis and, in some cases, could result in death.” A Class I recall is the most serious type of FDA recall and is issued when there is a “reasonable probability” that the defective product will cause a serious injury or illness, or death.
January 24, 2024: FDA Report Finds 17 Patients Exposed to Salmonella with Contaminated Colonoscope
An FDA report finds that 17 patients were exposed to potentially dangerous Salmonella bacteria based on more than 30 adverse event reports made to the agency in December 2023. The facility’s infection control department confirmed the suspected colonoscope was contaminated with Salmonella, despite being reprocessed according to manufacturer guidelines after each use. The bacteria carries the risk of gastroenteritis, bacteremia, and septicemia. The Imagina colonoscope was manufactured by Tokyo-based Hoya Corporation Pentax.
January 16, 2018: Judge Orders New Trial in Lawsuit Against Olympus, a Company that Makes Colonoscopy Scopes
A widow in Washington will get a new trial in her wrongful death lawsuit against Olympus, a company that makes scopes used for colonoscopies and other purposes. According to her lawsuit, her husband had a procedure done with an Olympus scope and contracted a severe superbug infection that ended his life. Her original legal complaint alleges that the Olympus scope’s design makes it difficult for medical facilities to clean and disinfect between uses on different patients. Allegedly, after about a seven-week trial, Olympus’s attorneys turned over key documents that suggested Olympus had failed to disclose patient infections and risks associated with its scopes. As a result, the judge ordered a new trial.
April 6, 2016: Group Lawsuit Accuses Massachusetts Hospital of Failing to Sterilize Colonoscopy Equipment
A group of 25 individuals sued Baystate Noble Hospital following news that the hospital improperly sterilized colonoscopy equipment in 2012 and 2013, affecting up to 293 patients. At least one of those patients had tested positive for a colonoscope-transmitted disease. The lawsuit includes patients who have not tested positive for a disease, with plaintiffs claiming the hospital subjected them to negligent infliction of emotional stress. Possible diseases the 293 patients could have been exposed to include hepatitis B, hepatitis C, and HIV.
2012-2015: Deadly Infections Linked to Olympus Scope
Hospitals in Europe, and then the U.S., report outbreaks of drug-resistant bacterial infections in patients after endoscopic procedures utilizing the Olympus TJF-Q180V duodenoscope. Outbreak sites include Erasmus Medical Center in the Netherlands, Virginia Mason Medical Center in Washington, Ronald Reagan UCLA Hospital in California, and Advocate General Hospital in Illinois. Subsequent investigations find a flaw in the TJF-Q180V’s elevator channel, which can trap moisture and provide an effective breeding ground for bacteria. Further, the “sealed” chamber can’t be reached with mechanical brushes, an important first line of defense in fighting such infections.
November 22, 2012: Judge Awards Veteran $1.25 Million In Colonoscopy Lawsuit
A U.S. District judge awarded Robert Metzler, an Air Force veteran, $1.25 million in a nonjury trial. Metzler had contracted hepatitis C during a colonoscopy at the Miami Veterans Administration Medical Center. The court determined that the center’s staff failed to properly clean the colonoscopy equipment, citing an investigation that found over 11,000 veterans received colonoscopies with improperly cleaned equipment between 2004 and 2009 at VA hospitals in Florida, Tennessee, and Georgia. The judge noted that Metzler had no other risks associated with the disease.
July 7, 2006: Review of Medical Literature Links Endoscope Disease Transmission to Reprocessing Errors
A comprehensive review of medical literature finds that endoscopic disease transmission in 48 Salmonella and 216 Pseudomonas cases was due to inadequate cleaning or drying during reprocessing. The review notes the role of biofilm in bacterial transmissions, a protective coating of slime that protects the microorganisms against chemical agents and anchors them to surfaces. The review also suggests that transmissions, though rare, are likely underreported.
October 1997: Evidence Suggests Colonoscopies Can Transmit Hepatitis-C
A French investigation links the transmission of the hepatitis C virus from an infected patient to two other patients by way of a colonoscope. The scope had been treated by immersing it in water containing a detergent and externally scrubbed with swabs. However, the biopsy suction channel had not been thoroughly cleaned with an appropriate brush. The biopsy forceps and diathermic loop, on the other hand, had been mechanically cleaned but not sterilized.
April 1987: Study Links Salmonella Newport Transmission to Colonoscopies
Following the colonoscopy and biopsy of a patient with Salmonella Newport gastroenteritis, the bacteria is later detected in the fecal samples of eight patients also treated with the same biopsy forceps. The study posits that inadequate disinfection allowed the bacteria to survive and cross-contaminate other colonoscopes. Two of the eight patients developed gastroenteritis.
What Are Colonoscopy Infection Lawsuits About?
Type of lawsuit: Personal injury/product liability lawsuit
Basis of lawsuit: People developed serious infections after their colonoscopies because scope manufacturers made a device that was potentially unsafe for patients
Key allegations: Medical device companies knew their colonoscopes could harbor and transmit pathogens to some patients and did not take proper actions
Alleged injury: Complications due to infection after a colonoscopy procedure
Potential defendants in lawsuits: Olympus and Hoya Corporation Pentax
Alleged device malfunction: Scope design makes device sanitization between procedures difficult
What Is a Colonoscopy Infection Lawsuit?
A colonoscopy infection lawsuit is a product liability and/or medical negligence claim made by patients who suffered a serious infection after a colonoscopy. Though these lawsuits include infections other than sepsis, sepsis is among the most immediately life-threatening complications that can result from a contaminated scope.
When patients link these complications to a contaminated colonoscope, they may choose to file a lawsuit against the device manufacturer. The lawsuit may include claims related to design defects, reprocessing failures, or inadequate warnings to patients or medical staff.
According to the Cleveland Clinic,
What Brands of Scopes Are Accused of Causing Infections After a Colonoscopy?
Because Olympus Corporation makes many of the gastrointestinal endoscopes worldwide and some of their scopes have an alleged design defect, many lawsuits have been filed against the company. According to a 2024 investor report from the company, Olympus holds a 70% global market share for gastrointestinal endoscopes.
What Legal Theories Apply in a Colonoscopy and Sepsis Lawsuit?
Even when hospitals and surgical centers disinfected colonoscopes according to regulatory guidelines, the devices and accessories may still have contained potentially dangerous substances. This means that manufacturers could be held responsible through product liability lawsuits. Legal theories that could apply to colonoscopy infection lawsuits include:
- Product liability
- Design defects
- Breach of warranties
- Failure to warn (consumers and physicians)
- Failure to disclose known dangers or defects
How Can Colonoscopes Cause Sepsis or Other Infections?
Colonoscopes can cause sepsis if scopes used during the procedure introduce bacteria into the body or if the intestine is perforated during the procedure. Medical providers use endoscopes called colonoscopes to examine and operate on the intestine. These devices pass through bacteria-rich environments in the body.
The reusable parts of these devices need to undergo reprocessing (i.e., disinfecting) before they can be safely used on the next patient. This involves high-level
Sepsis occurs through one of two pathways, endogenous or exogenous:
- Endogenous: Transmission within a single patient, such as through a perforation or relocation of bacteria from an area that can handle them to one that cannot. For example, if the colon is perforated and bacteria leak out, leading to infection.
- Exogenous: Transmission between patients through contaminated devices. For example, bacteria on a piece of medical equipment introduces bacteria inside the body.
Most of the claims against Olympus and other device manufacturers involve exogenous transmission (i.e., infections originating outside the body).
What Infections Have Been Linked to Scopes Used in Colonoscopies?
Numerous infections have been linked to scopes used during colonoscopies. Colonoscopes have been linked to bacterial infections, though some viral and fungal outbreaks have also been reported. Some of these bacterial infections are highly resistant to antibiotics (e.g., superbug infections), making them challenging to treat.
For example, a study published in the American Journal of Infection Control found that gastrointestinal scopes that were reprocessed in accordance with U.S. government guidelines could still have viable microbes and biologic debris. The study said this suggested that disinfection practices may not be enough to decontaminate some scopes.
Another study published in Clinical Gastroenterology and Hepatology, called “The Study of a Contaminated Colonoscope,” found that some colonoscopes still had pathogens present after decontamination. Organisms of high concern include:
- Staphylococcus aureus
- Pseudomonas aeruginosa
- Mycobacterium avium-intracellulare (MAI)
- Klebsiella pneumonia
People who are infected with these pathogens may experience septic shock, bloodstream infections, liver abscess, CRE colonization, ICU admission, and death.
While duodenoscopes have received the most FDA scrutiny due to device complexity and the more sensitive environments in which they operate, colonoscopes share many of the same risks in the form of narrow channels that can provide an excellent breeding ground for biofilm through moisture retention.
CDC and FDA Actions Related to Contaminated Colonoscopes and Endoscopes
The CDC and FDA have issued guidance, recalls, and import bans in the wake of Olympus scope contamination outbreaks, with recommendations changing as the problems become better understood. Actions taken by the agencies include:
- Warning letters to Olympus
- Encouraging the use of disposable components
- Guidelines for sterilization and disinfection
- Surveillance protocols
- Import bans
The CDC identified prompt manual cleaning as the most critical step in preventing biofilm buildup on endoscopes. However, this protocol can be prone to error due to time constraints and resource constraints. Best practices now recommend that such cleaning take place under 10x magnification to avoid missing biological material that can’t easily be seen with the naked eye.
Nevertheless, a multi-site study found that 71% of endoscopes contained microbial growth despite following recommended guidelines (as cited in: Residual moisture and waterborne pathogens inside flexible endoscopes: Evidence from a multisite study of endoscope drying effectiveness). The regulatory bodies have, as a result, increasingly pushed for the use of disposable components in endoscopes and colonoscopes.
Why Does Scope Reprocessing Sometimes Fail?
There are numerous potential points of failure in scope reprocessing protocols. The first is the design of the device, which may contain narrow channels large enough to trap moisture and biological material but small enough to be difficult to clean with brushes. Additional vulnerabilities in scope disinfecting procedures include:
- Delayed manual cleaning
- Incomplete manual cleaning
- Improper setup of automated endoscope reprocessors (AERs)
- Insufficient drying
- Contaminated rinse water
- Mechanical channel damage
- Biofilm persistence
Most of these processes are designed to combat biofilm, which is a means through which some strains of bacteria protect themselves.

Why Are Some Colonoscopes Difficult to Fully Sterilize?
Research has shown that small channels in endoscope devices and so-called elevator mechanisms may make reusable colonoscopes difficult to reprocess and disinfect. Small compartments within colonoscopes and associated accessories may be difficult to disinfect with chemicals or manual scrubbing. Additionally, some bacteria may be especially resilient to recommended sterilization methods. Some studies have also shown that it can be difficult to verify full sterilization of devices.
What Makes a Colonoscope Contaminated?
Colonscopes and some associated accessories are reusable medical devices. If they are not (or cannot) be properly sanitized, their surfaces may become contaminated. Types of contamination that may affect colonoscopes include
- Biological debris remaining
- Biofilm formation
- Microbial survival in device channels
Pathogens left on devices after contamination are one form of contamination. These pathogens (e.g., bacteria, fungi, or viruses) can make people sick.
What Possible Role Does Biofilm Play in Colonoscopy Infections?
Biofilm is a slimy protective layer some bacteria form to survive chemical assaults, such as from disinfectants. Biofilm can also help physically anchor the bacterial colony to a surface. Biofilm can form quickly in the small channels of the scopes. Preventing the buildup of biofilm involves physically removing biological material and moisture from those channels to prepare them for chemical disinfection.
Biofilm’s resistance to chemical disinfectants is a central theory in many medical device contamination lawsuits involving scopes. This underscores the importance of design defects, which can shield bacteria from mechanical interventions and provide an ideal climate in which biofilm can develop.
Are Colonoscope-Linked Infection Outbreaks Rare?
Colonoscope-linked sepsis outbreaks are not very common, and not every case of bacterial transmission leads to sepsis. Large claims-based studies summarized by Johns Hopkins Medical linked 1.1 infections per 1,000 screening colonoscopies. Non-screening colonoscopies were a little more likely to be linked to infections at a rate of 1.6 per 1,000.
Note that proven rates of transmission are lower, but difficult to pin down due to under-detection. Typically, only MDRO (multidrug-resistant organism) outbreaks tend to trigger investigations..
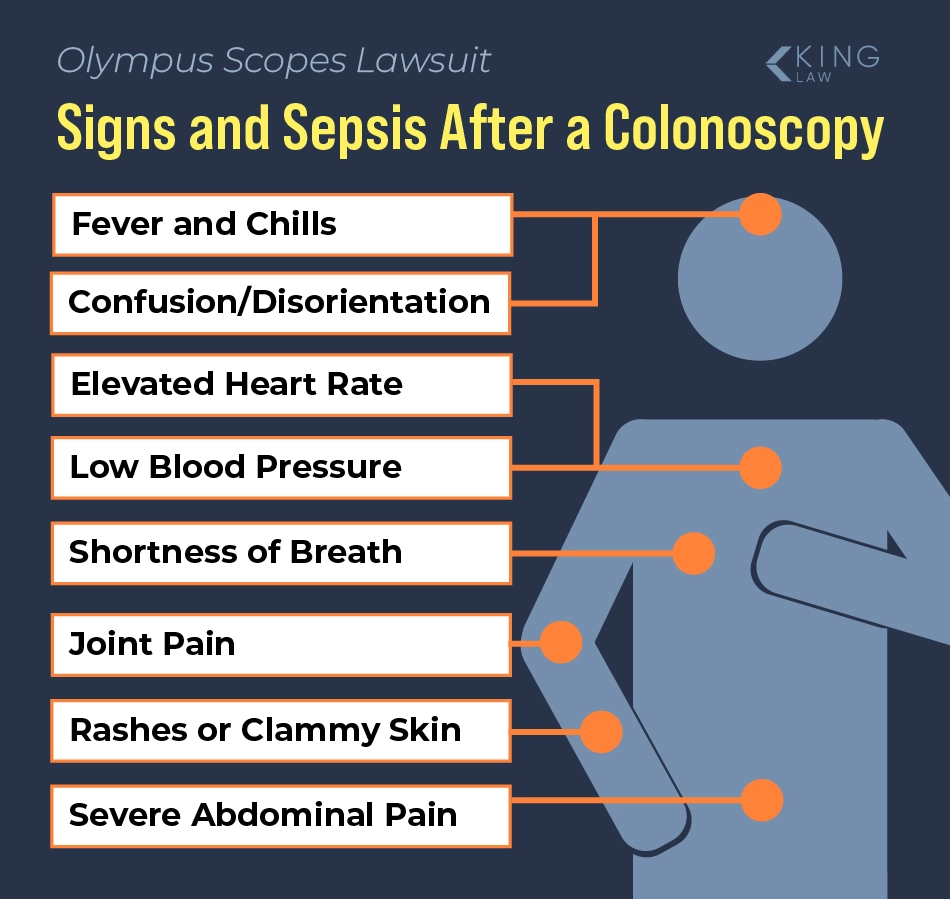
What Are the Symptoms of Infection After a Colonoscopy?
Patients who undergo a colonoscopy should monitor themselves for signs of infection, as some infections can be life-threatening. According to the Cleveland Clinic, sepsis symptoms come on quickly and may include:
- Fever and chills
- Severe abdominal pain
- Rapid heart rate
- Confusion and disorientation
- Low blood pressure
- Rashes or clammy skin
- Joint pain
- Shortness of breath
Sepsis can be extremely dangerous, making early medical intervention critical. The Surviving Sepsis Campaign notes that lactate testing, blood cultures, and rapid antibiotic administration are critical interventions for patient survival.
How Long After a Colonoscopy Can an Infection Appear?
According to Johns Hopkins Medicine, sepsis after a colonoscopy may take up to 30 days to develop. Patients may not have symptoms right away or think that their colonoscopy may have caused their infection. Patients who underwent a colonoscopy and develop an infection should make sure their doctor knows about their recent medical procedure.
How Do You Prove a Contaminated Olympus Colonoscope Caused Sepsis?
Proving an Olympus colonoscope caused sepsis involves linking a specific case to the procedure and medical device. An attorney knowledgeable in colonoscopy infection claims can guide you on building a strong case.
The first consideration is timing proximity. If the case happened within 30 days of a colonoscopy, it may be worth investigating the link. Evidence that can be used in such a case includes:
- Epidemiologic studies and reports
- Bacterial culture data
- Device tracking logs
- Genome sequencing
- Clusters of cases that used the same device
- Reprocessing logs
Your attorney can help you obtain this information. Lawsuits can also use existing regulatory actions, such as FDA device recalls and import alerts, to substantiate their claims.
How Do You Find Out What Colonoscope Was Used During Your Procedure?
Hospitals and out-patient centers track the medical devices their doctors use. Information like serial and lot numbers is especially important if recalls or incident reports arise with a medical device.
Information about the device used in your colonoscopy may be contained in records such as:
- Operative reports
- Endoscopy suite logs
- Sterilization tracking systems
- Electronic medical records
Attorneys can request these logs and records to help build your case. These records can provide essential proof in colonoscope lawsuits.
What Legal Theories Apply in a Colonoscopy Infection Lawsuit?
Legal theories cited in colonoscopy infection lawsuits may vary from case to case. Most lawsuits will center around one or more of the following product liability or negligence justifications:
- Design defects, such as the channel design
- Manufacturing defects, products that were malformed during production
- Failure to warn, such as the risks of using the device
- Negligence, such as providing inadequate reprocessing protocols or failing to report adverse events
- Breach of warranty
- Wrongful death, when applicable
Different theories may apply to different devices. For example, a shipment of an accessory may have a manufacturing flaw, while another device may be flawed at a more fundamental, design level.
Who Qualifies to File a Colonoscopy Infection Lawsuit?
King Law looks for the following qualifications when evaluating colonoscopy infection lawsuits:
- You had a medical procedure that utilized an Olympus colonoscope or accessory.
- You developed sepsis or an infection requiring intravenous antibiotics or a similar intervention.
- You had no history of such an infection within a year prior to the procedure.
Filing deadlines also apply. These deadlines vary by jurisdiction, so it is best to contact an attorney quickly if you have recently suffered a serious infection not long after a colonoscopy.
When Should You Contact a Lawyer After a Colonoscopy Infection?
If you were hospitalized after a colonoscopy, developed sepsis, required IV antibiotics, or were diagnosed with a bloodstream infection, your illness might be related to a contaminated colonoscope. You should speak to an attorney to understand your legal options and potential eligibility for filing a colonoscopy infection lawsuit.
Not all infections are due to device defects or manufacturer negligence. An experienced attorney can determine potential faulty parties after reviewing information in your case.
What Compensation Is Available in a Colonoscopy Infection Lawsuit?
Victims of a colonoscopy-related sepsis injury may be eligible for both economic and non-economic compensation, with the exact amount varying depending upon the severity of the injury. For example, you may be compensated for:
- Direct medical expenses related to the injury, including ICU costs
- Rehabilitation costs
- Lost income or earning potential due to the injury
- Pain and suffering
- The effects of long-term organ damage, like kidney injuries or cognitive impairment
- Wrongful death-related compensation, if applicable, including loss of companionship
In some cases and jurisdictions, punitive damages may also apply. These are not compensation for a specific economic or non-economic injury, but rather are meant to be a penalty for egregious behavior on the part of the defendant.
What Should You Do If You Develop an Infection After a Colonoscopy and Want to File a Lawsuit?
If you developed sepsis after a colonoscopy, it is best to act quickly (as soon as you have recovered enough to do so). Acting quickly can help you gather timely and accurate information, including:
- Your medical records, especially those related to the procedure and sepsis
- The manufacturer and model of the devices used during the colonoscopy
- Hospitalization details
Consult with an attorney as soon as possible. An experienced lawyer can help you track down additional evidence and link your experiences to known outbreak reports and FDA communications, if applicable.
King Law Is Investigating Olympus Colonoscopy and Infection Claims
King Law is currently investigating sepsis claims involving Olympus colonoscopes in all 50 states. Our attorneys have decades of experience handling complex medical device litigation and representing clients against large, corporate defendants. Our partners can leverage their access to medical experts and infection-control consultants to fortify your case and help you seek justice and compensation for your sepsis injuries.
Start Your Free Colonoscopy and Sepsis Case Review Today
If you or a family member suffered sepsis or another serious infection after a colonoscopy, contact King Law today at (585) 496-2648 for a free and confidential case evaluation with no obligations to continue. Our lawyers work on contingency, so there are no upfront costs to pursuing your claim.
Frequently Asked Questions (FAQs)
Source List
Here is a list of sources used in this article.
Centers for Disease Control and Prevention. Essential Elements of a Reprocessing Program for Flexible Endoscopes. CDC, 2016.
Centers for Disease Control and Prevention. Healthcare Equipment Disinfection and Sterilization. CDC, 2023.
Centers for Disease Control and Prevention. MDRO Containment Strategy. CDC, 2022.
Cleveland Clinic. Sepsis. Cleveland Clinic, 2024.
Food and Drug Administration. Update: Alert on Endoscope Accessories – Forceps/Irrigation Plug Issue with Olympus. U.S. Food and Drug Administration, 2024.
Food and Drug Administration. FDA Issues Two Warning Letters to Leading Manufacturer of Endoscopes. U.S. Food and Drug Administration, 2023.
Food and Drug Administration. Import Alerts on Certain Olympus Medical Devices Manufactured in Japan – Letter to Health Care Providers. U.S. Food and Drug Administration, 2025.
Food and Drug Administration. Infections Associated with Reprocessed Duodenoscopes. U.S. Food and Drug Administration, 2022.
Food and Drug Administration. Information About Automated Endoscope Reprocessors (AERs) and FDA’s Evaluation. U.S. Food and Drug Administration, 2021.
Food and Drug Administration. Reprocessing Medical Devices in Health Care Settings: Validation Methods and Labeling Guidance for Industry and FDA Staff. U.S. Food and Drug Administration, 2015.
Goyal, H., et al. Gastrointestinal Endoscope Contamination Rates: A Systematic Review and Meta-Analysis. Endoscopy International Open, 2022.
Hopkins Medicine Newsroom. Infection Rates After Colonoscopy and Endoscopy at U.S. Specialty Centers Are Higher Than Previously Thought. John Hopkins Medicine, 2018.
LFM Healthcare Strategies. U.S. Patients Exposed to Colonoscope Contaminated with Salmonella, FDA Reports. 2024.
Levin, Sam. Deadly Infections Linked to Olympus Duodenoscopes Spur Safety Concerns. Los Angeles Times, 12 May 2015.
Levin, Sam. Timeline: Olympus Endoscope Infections and Investigations. Los Angeles Times, 20 Dec. 2015.
MassLive News Staff. Baystate Noble Hospital Lawsuit Filed After Improper Sterilization of Colonoscopy Equipment. MassLive, 2016.
MedTech Dive Staff. FDA Early Alerts Highlight Concerns with Olympus, Trokamed, Nuwellis, and Baxter Devices. MedTech Dive, 2024.
Muscarella, Lawrence F. The Study of a Contaminated Colonoscope. Clinical Gastroenterology and Hepatology, vol. 8, no. 7, 2010, pp. 571–573
Ofstead, Claire L., et al. Persistent Contamination on Colonoscopes and Gastroscopes Detected by Rapid Indicators Despite Reprocessing. American Journal of Infection Control, vol. 44, no. 6, 2016, pp. 712–714.
Olympus Corporation. Olympus Medical Business. Olympus Investor Relations, 2024.
Olympus Corporation. Olympus Global News Release on Medical Device Safety Measures. Olympus, 2025.
Olympus Corporation. TJF Duodenoscope Reprocessing Customer Letter. Olympus America, 2025.
Petersen, B. T., et al. Transmission of Infection by Gastrointestinal Endoscopy. Gastrointestinal Endoscopy, 2006.
Society of Critical Care Medicine. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock. 2021.
The Ledger Staff. Veteran Awarded $1.25 Million in Colonoscopy Infection Lawsuit. The Ledger, 23 Nov. 2012.
Wang, P., et al. Biofilm Formation and Bacterial Survival in Endoscope Channels. American Journal of Infection Control, 2018.
Zhang, X., et al. Microbial Contamination of Endoscopes Despite Reprocessing. Infection and Drug Resistance, 2022.

